 |
 |
 |
| |
HBsAg Clearance Continues to Increase Post-Treatment in Patients with HCV/HBV Coinfection Treated With Peginterferon Alfa-2a Plus Ribavirin: 1.5 Year Follow up
|
| |
| |
Reported by Jules Levin
AASLD Nov 3 2009, Boston, MA, USA
Liu C-J1, Chuang W-L2, Lee C-M3, Yu M-L2, Lu S-N3, Dai C-Y2, Huang J-F2, Hu T-H3, Chen C-H3, Hung C-H3, Wang J-H3, Wu S-S4, Liao L-Y5, Kuo H-T6, Chao Y-C7, Tung S-Y8, Yang S-S9, Su W-W4, Lin C-L5, Kao J-H1, Liu C-H1, Chen P-J1, Chen D-S1
1National Taiwan University College of Medicine, Taipei, Taiwan; 2Kaohsiung Medical University Hospital, Kaohsiung, Taiwan; 3Chang Gung Memorial Hospital-Kaohsiung, Kaohsiung, Taiwan; 4Changhua Christian Hospital, Changhua, Taiwan; 5Taipei City Hospital, Ren-Ai Branch, Taipei, Taiwan.
6Chi Mei Medical Center, Tainan, Taiwan; 7Tri-Service General Hospital, Taipei, Taiwan; 8Chai-Yi Chang Gung Memorial Hospital, Chiayi, Taiwan; 9Cathay General Hospital, Taipei, Taiwan
Presented at the 60th Annual Meeting of the American Association for the Study of Liver Diseases 30 October - 3 November 2009, Boston, USA
Summary
A finite course of treatment with PEGASYS/ribavirin resulted in undetectable HBV DNA in 47% of patients 1.5 years post-treatment and undetectable HCV RNA (SVR) in 84% of patients - overall 34% of patients achieved undetectable levels of both viruses
Rates of HBsAg clearance increased post-treatment from 12% at 6 months post-treatment to 16% at 1.5 years post-treatment
HBsAg clearance was achieved by patients infected with either genotype B or C
All patients achieving HBsAg clearance post-treatment also had HBV DNA <2000 IU/mL
Conclusion
Combination therapy of PEGASYS and ribavirin is effective for treatment of patients dually infected with HCV and HBV and can result in successful treatment of both chronic viral infections
The high rates of HBsAg clearance - the closest outcome to cure in chronic hepatitis B - that increase post-treatment, coupled with the similar SVR rates in HCV/HBV-coinfection and HCV-monoinfection, suggest that PEGASYS and ribavirin combination therapy is an effective first-line treatment strategy for this patient group
Background
Patients with hepatitis C virus (HCV) and hepatitis B virus (HBV) coinfection are at high risk of progressive disease and, therefore, need effective treatment strategies
In some coinfected patients, presence of HCV suppresses HBV - this results in the low HBV DNA levels, which are a characteristic of HCV/HBV-coinfection. Effective suppression of HCV following treatment can, therefore, sometimes result in reappearance of HBV DNA1
We previously reported a high overall rate of HBsAg clearance (12%) 6 months post-treatment in HCV/HBV-coinfected patients treated with peginterferon alfa-2a and ribavirin as part of a study that demonstrated similar efficacy in terms of sustained virologic response (SVR) rates in HCV/HBV-coinfected patients and HCV- monoinfected patients1,2
In the current study we investigated whether the rate of HBsAg clearance increases during long-term follow-up of patients coinfected with HCV/HBV
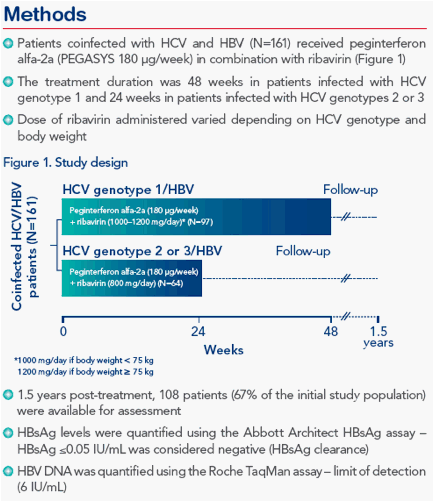
RESULTS
Baseline characteristics of the initial population
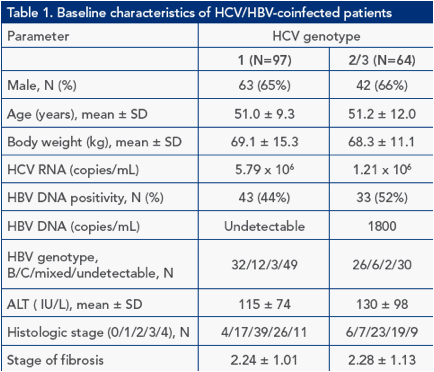
Table 1 shows the baseline characteristics of HCV/HBV patients included in the initial study (N=161)
HBV DNA was detectable in 76 patients (47%)
The mean levels of HBV DNA at baseline were low - a characteristic of patients with HCV/HBV-coinfection
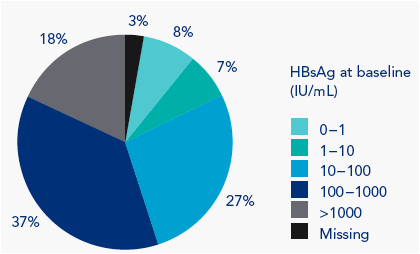
The majority of patients had baseline HBsAg levels in the range 100-1000 IU/mL (Figure 2)
Figure 2. Distribution of HBsAg levels at baseline
Virologic response 1.5 years post-treatment
Overall HBV DNA was undetectable in 51 HCV/HBV-coinfected patients (47%) 1.5 years post-treatment
-- Of the 51 patients with detectable HBV DNA levels at baseline, 73% had undetectable HBV DNA levels 1.5 years post-treatment
Overall, HCV RNA was undetectable (SVR) in 91 patients (84%) 1.5 years post-treatment
A total of 41 patients (38%) had undetectable levels of both viruses 1.5 years post-treatment
Figure 3. HBsAg clearance continues to increase post-treatment
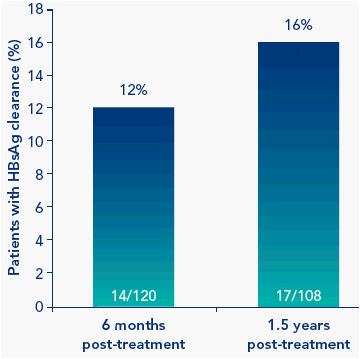
All patients with HBsAg clearance 1.5 years post-treatment had HBV DNA <2000 IU/mL with 53% of patients (9/17) having undetectable HBV DNA levels 1.5 years post-treatment
Eight patients also had undetectable HCV RNA (SVR)
HBsAg clearance was seen in patients infected with HBV genotypes B and C - four patients with genotype B and four patients with genotype C achieved HBsAg clearance (genotype was not available for the remaining patients)
References
1. Liu CJ et al. Peginterferon alfa-2a plus ribavirin for the treatment of dual chronic infection with hepatitis B and C viruses. Gastroenterology
2009; 136(2):496-504
2. Liu CJ et al. Analysis of HBsAg levels in patients coinfected with hepatitis
B virus and hepatitis C virus treated with peginterferon alfa-2a and ribavirin. J Hepatol 2009; 50 (suppl 1):S9
Disclosure information:
Editorial support for the development of this poster was provided by Elements
Communications and funded by F. Hoffmann-La Roche, Basel, Switzerland
|
| |
|
 |
 |
|
|