 |
 |
 |
| |
A Finite Course of Peginterferon Alfa-2a Results in Inactive Chronic Hepatitis B and HBsAg Clearance 5 Years Post-Treatment in Patients With HBeAg-Negative Disease: Baseline Characteristics and Predictive Factors of Long-Term Response
|
| |
| |
Reported by Jules Levin
AASLD Nov 3 2009 Boston, MA, USA
Marcellin P,1 Piratvisuth T,2 Brunetto M,3 Bonino F,4 Farci P,5 Yurdaydin C,6 Gurel S,7 Kapprell H-P,8 Messinger D,9 Batrla R10
1Service d'Hepatologie and Centre de Recherches Biologiques Beaujon, Paris, France; 2Department of Internal Medicine, NKC Institute of Gastroenterology and Hepatology, Songklanagarind Hospital, Hat Yai, Thailand; 3UO Gastroenterologia ed Epatologia, Azienda Ospedaliera Universitaria Pisana, Pisa, Italy; 4Scientific Direction, Foundation IRCCS
Policlinico of Milan and University of Pisa, Pisa, Italy; 5Universita di Cagliari, Cagliari, Italy; 6Faculty of Medicine, University of Ankara, Ankara, Turkey; 7Department of Gastroenterology, Uludag University, Uludag, Turkey; 8Abbott GmbH & Co, Wiesbaden, Germany; 9IST GmbH & Co, Mannheim, Germany; 10Hoffmann-La Roche, Basel, Switzerland
Presented at the 60th Annual Meeting of the American Association for the Study of Liver Diseases 30 October - 3 November 2009, Boston, USA
Summary
31% of peginterferon alfa-2a-treated patients achieved sustained immune control 1 year post-treatment and this response was maintained through to year 5 in 88% of patients
A high proportion (28%) of patients with sustained immune control 1 year post-treatment went on to clear HBsAg 5 years post-treatment (compared with 12% of the overall population)
On-treatment HBsAg level was a significant predictor of sustained immune control 1 year post-treatment, and importantly, of HBsAg clearance 5 years post-treatment
Of the patients with a ≥10% decline at week 12 or week 24 and sustained immune control 1 year post-treatment, 40% and 45%, respectively, achieved HBsAg clearance 5 years post-treatment
Patients with a <10% HBsAg decline at week 12 and sustained immune control 1 year post-treatment could also gain long-term benefit from peginterferon alfa-2a
Conclusions
High rates of HBsAg clearance - the closest outcome to clinical cure in CHB - 5 years post-treatment were achieved by patients with low HBsAg levels during peginterferon alfa-2a therapy and in patients who had achieved sustained immune control 1 year post-treatment
Patients with a 10% decline in HBsAg levels during treatment and sustained immune control 1 year post-treatment had particularly high rates of HBsAg clearance (38-40%) 5 years post-treatment
A proportion (up to 38%) of patients not achieving an HBsAg decline ≥10% at week 12 or week 24 achieved HBsAg clearance 5 years post-treatment, therefore, physicians should be cautious when making treatment decisions in these patients
Background
The long-term follow-up of patients enrolled in the large-scale phase 3 study of peginterferon alfa-2a (PEGASYS) ± lamivudine in patients with HBeAg-negative chronic hepatitis B (CHB) has shown that peginterferon alfa-2a can achieve sustained response up to 5 years post-treatment1,2
Of particular significance, the rate of HBsAg clearance continued to increase during the follow-up period, reaching 12% at 5 years.2
We have, therefore, sought to characterize the baseline factors of the patients who achieved this important endpoint - considered by recent guidelines to represent the 'ideal' endpoint of therapy3
It has been established previously that patients who achieve a sustained response following interferon-based therapy have an increasing chance of HBsAg clearance when they are followed up longer term. Sustained suppression of HBV DNA to below 10,000 copies/mL in patients treated with interferon-based
therapy can be considered to represent the attainment of sustained immune control (as in patients with inactive disease). We have, therefore, characterized patients who achieved this 1 year post-treatment and investigated the rate of HBsAg clearance 5 years post-treatment in this group
Recent analyses have shown that patients achieving a response to peginterferon alfa-2a have significantly greater decreases in HBsAg during treatment compared with patients who do not.4-6
We also sought to determine the chance of sustained immune control and subsequent HBsAg clearance among patients with an on-treatment HBsAg decline

RESULTS
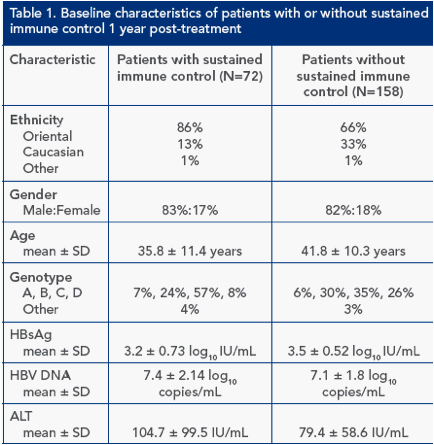
Baseline characteristics of patients with sustained immune control 1 year post-treatment (Table 1)
Of the 230 patients included in the long-term follow-up study, 72 (31%) achieved sustained immune control 1 year post-treatment
Predictors of sustained immune control 1 year post-treatment
Univariate logistic regression analysis identified baseline HBsAg, ethnicity, genotype, age and baseline alanine aminotransferase (ALT) to be significant predictors of response (Table 1)
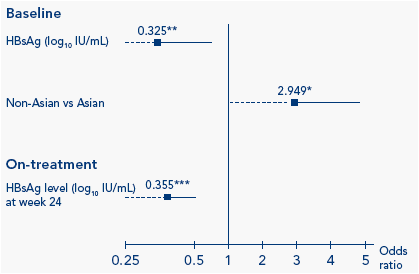
On-treatment, HBsAg was identified as a significant predictor of sustained immune control 1 year post-treatment using univariate analysis (Table 2). The odds ratio (OR) for HBsAg level on-treatment was substantially higher than that at baseline. Multivariate analysis identified age, baseline HBsAg level, ethnicity and genotype to be predictors at baseline and absolute HBsAg level (log10 IU/mL) at week 24 and log10 HBsAg drop at week 12 as on-treatment predictors (Figure 1).
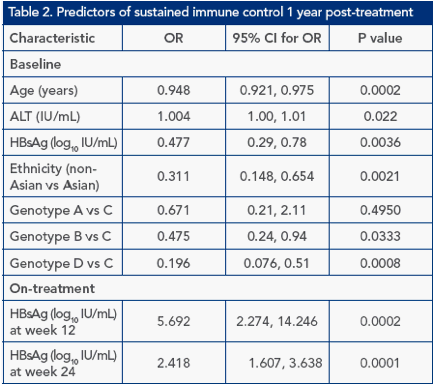
Figure 1. Multivariate analysis of sustained immune control 1 year
post-treatment
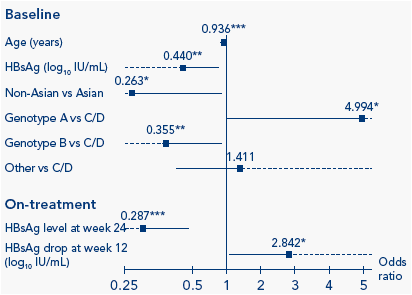
Explanatory factors at baseline considered in stepwise selection procedure: Age (1 year decrease), ALT ratio (ALT/ULN), HBsAg level (log10 IU/mL), ethnicity, gender, fibrosis score, genotype. Explanatory factors on-treatment considered in stepwise selection procedure: HBsAg at week 12, HBsAg at week 24, log drop in HBsAg from baseline to week 12; log drop in HBsAg from baseline to week 24; HBV DNA at week 12, HBV DNA at week 24, log drop in HBV DNA from baseline to week 12; log drop in HBV DNA from baseline to week 24. Boxes: odds ratios; lines: confidence intervals (dotted lines represent confidence intervals
<0.25 or >5). *P<0.05; ** P<0.01; *** P<0.001
Of the 53 patients who achieved an HBsAg decline ≥10% from baseline at week 12, 47% (25/53) achieved sustained immune control 1-year post-treatment compared with 16% (11/67) of patients with an HBsAg decline <10%
Similarly, of the 67 patients who achieved an HBsAg decline ≥10% from baseline at week 24, 43% (29/67) achieved sustained immune control 1 year post-treatment compared with 13% (7/53) of patients with an HBsAg decline <10%
Baseline characteristics of patients with HBsAg clearance 5 years post-treatment (Table 3)
A total of 28 patients (12%) achieved HBsAg clearance 5 years post-treatment
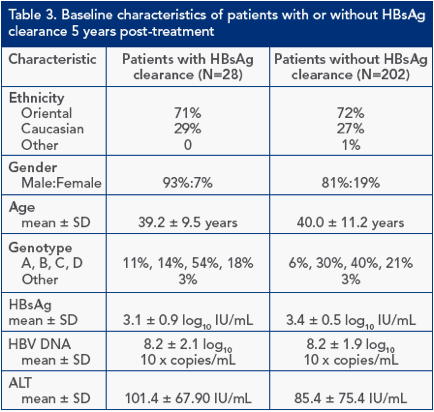
Univariate logistic regression analysis identified baseline HBsAg level to be a significant predictor of HBsAg clearance 5 years post-treatment (Table 4). Unlike for 1-year post-treatment response, age, ethnicity and genotype did not predict post-treatment response
Multivariate analysis identified HBsAg level (log10 IU/mL) and ethnicity at baseline and HBsAg level at week 24 as predictors of HBsAg clearance 5 years post-treatment (Figure 2)
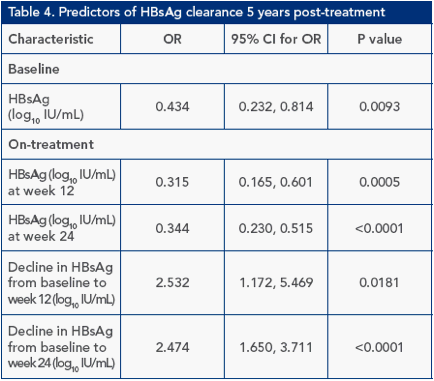
Figure 2. Multivariate analysis of HBsAg clearance 1 years post-treatment
Explanatory factors at baseline considered in stepwise selection procedure: Age (1 year decrease), ALT ratio (ALT/ULN), HBsAg level (log10 IU/mL), ethnicity, gender, fibrosis score, genotype. Explanatory factors on-treatment considered in stepwise selection procedure: HBsAg at week 12, HBsAg at week 24, log drop in HBsAg from baseline to week 12; log drop in HBsAg from baseline to week 24; HBV DNA at week 12, HBV DNA at week 24, log drop in HBV DNA from baseline to week 12; log drop in HBV DNA from baseline to week 24. Boxes: odds ratios; lines: confidence intervals (dotted lines represent confidence intervals
<0.25 or >5). *P<0.05; ** P<0.01; *** P<0.001
Association between sustained immune control 1 year post-treatment and HBsAg clearance 5 years post-treatment
A total of 88% (36/41)* of patients who achieved sustained immune control 1 year post-treatment maintained this response through to year 5 post-treatment
Of the 72 patients achieving sustained immune control 1 year post-treatment, 20 (28%) went on to achieve HBsAg clearance 5 years post-treatment
Rates of HBsAg clearance were high in patients with a 10% decline in HBsAg level from baseline at either week 12 or 24 AND sustained immune control 1 year post-treatment
· 40% (10/25) of patients with an HBsAg decline ≥10% at week 12 AND sustained immune control 1 year post-treatment achieved HBsAg clearance 5 years post-treatment compared with 36% (4/11) of patients with an HBsAg decline <10% at week 12 and sustained immune control 1 year post-treatment
· 45% (13/29) of patients with an HBsAg decline ≥10% at week 24 AND sustained immune control 1 year post-treatment achieved HBsAg clearance 5 years post-treatment compared with 14% (1/7) of patients with an HBsAg decline <10% at week 24 and sustained immune control 1 year post-treatment
*In patients with data available 1 and 5 years post-treatment
References
1. Marcellin P et al. Quantitative HBsAg for early prediction of response to PEGASYS therapy in patients with early HBeAg-negative disease. Hepatol Int 2009; 3(1):27
2. Marcellin P et al. Increasing rates of HbsAg clearance and seroconversion in patients with HBeAg-negative disease treated with peginterferon alfa-2a ± lamivudine: results of 5-year post-treatment follow-up. J Hepatol 2009; 50(suppl 1):S336
3. EASL. EASL clinical practice guideline: management of chronic hepatitis B. J Hepatol 2009; 50:227-242
4. Marcellin P et al. Sustained response of hepatitis B e antigen-negative patients 3 years after treatment with peginterferon alfa-2a. Gastroenterol 2009; 136:2169-79
5. Moucari R et al. Early serum HBsAg drop: a strong predictor of sustained virological response to pegylated interferon alfa-2a in HBeAg-negative patients. Hepatology 2009; 49:1151-57
6. Moucari R et al. High rates of HBsAg seroconversion in HBeAg-positive chronic hepatitis B patients responding to interferon: a long-term follow-up study. J Hepatol 2009; 50:1084-92
7. Marcellin P et al. Peginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-negative chronic hepatitis B. New Engl J Med 2004; 351:1206-1217
Disclosure information:
Editorial support for the development of this poster was provided by Elements Communications and was funded by F.Hoffmann-La Roche, Basel, Switzerland
|
| |
|
 |
 |
|
|