 |
 |
 |
| |
Adherence to AASLD Guideline Recommendations for Laboratory Monitoring of Chronic Hepatitis B Patients Who are Not Receiving Antiviral Treatment
|
| |
| |
Reported by Jules Levin
AASLD Oct 30-Nov 3 2009, Boston, MA, USA
Timothy Juday1, Hong Tang1, Melissa L Harris3, Annette Z Powers2, Edward Kim2, George J Hanna1
1Bristol-Myers Squibb, Plainsboro, NJ, USA; 2Eisai, Inc. Woodcliff Lake, NJ, USA
SUMMARY
The results of this study suggest low rates of guideline-recommended laboratory monitoring of untreated patients with CHB, with high variability in monitoring across specialties, region, and by specific laboratory test
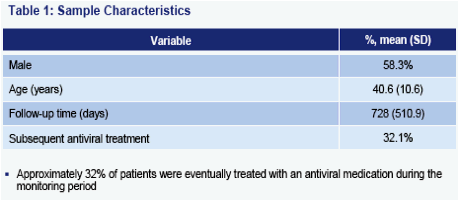
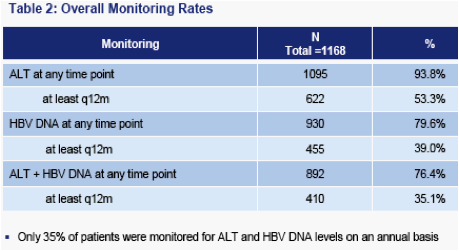
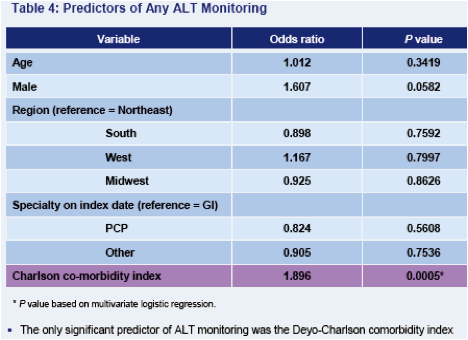
Only about one-third of patients received both ALT and HBV DNA tests on an annual basis. While ALT assessment is the more frequently applied of the two tests, only slightly more than half of all patients receive annual ALT testing
- ALT also appears to be used in a non-specific manner, since the only predictor of monitoring was co-morbidity burden
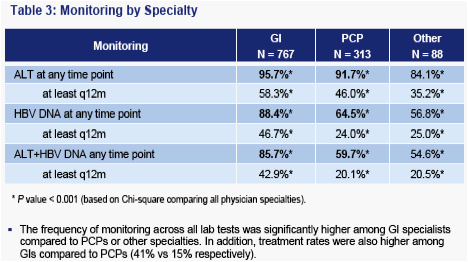
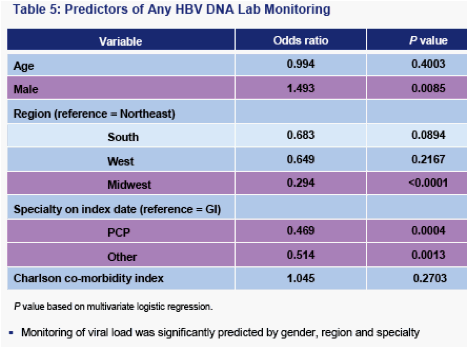
HBV DNA monitoring rates are lower than ALT monitoring; less than half of all patients diagnosed by GIs received annual monitoring
- HBV DNA monitoring appears more specific in that it is not predicted by comorbidities. However, diagnosis by a non-GI or treatment in the Midwest are negative predictors of HBV DNA monitoring
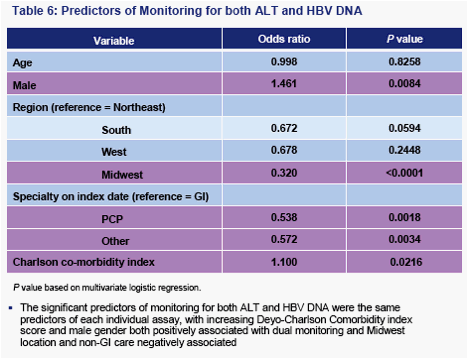
Rates of monitoring for ALT and HBV DNA are similar, suggesting that HBV DNA monitoring is a rate-limiting feature (i.e. most patients who receive HBV DNA monitoring also receive ALT monitoring)
CONCLUSION
Guideline-recommended laboratory monitoring of untreated CHB patients remains low in a commercially insured population, particularly measures of HBV DNA viral load
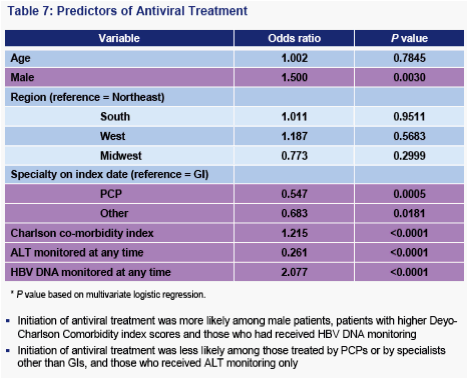
These findings suggest that initiation of appropriate antiviral therapy may be delayed, leaving patients at risk for disease progression
Improving monitoring rates may increase the rates of medically appropriate treatment of CHB, preventing delay of treatment and complications of the disease
More education is needed about importance of appropriate CHB monitoring as recommended by the guideline
BACKGROUND
Over 1 million US residents have chronic hepatitis B (CHB) infection, with an estimated prevalence of 0.3%-0.5%. In spite of the availability of an effective vaccine, the number of US residents with CHB infection remains high, primarily due to the slow progression of CHB and the influx of immigrants with chronic infection.1
Approximately 15% to 40% of untreated CHB patients will develop serious hepatitic sequelae during their lifetime, including cirrhosis, hepatic decompensation, hepatocellular carcinoma (HCC), and death.2-6
Regular laboratory monitoring of untreated patients is recommended in order to determine when to initiate antiviral therapy.2
American Association for the Study of Liver Diseases (AASLD) guidelines recommend monitoring of ALT and HBV DNA levels at least annually, although based on laboratory results frequency of monitoring may be higher.
OBJECTIVES
To assess adherence to AASLD guideline-recommended laboratory monitoring of CHB patients not receiving antiviral treatment:
- identify predictors of monitoring
-- identify predictors of initiation of antiviral treatment (i.e. oral
nucleos(t)ide analogues or interferon)

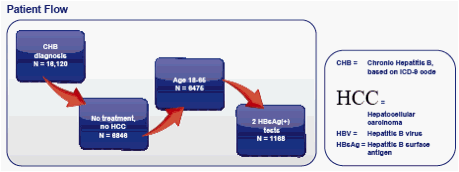

RESULTS
REFERENCES
1. Belongia EA, Costa J, Gareen IF, et al. National Institutes of Health Consensus Development Conference
Statement: management of hepatitis B. Ann Intern Med. 2009;150:104-10.
2. Lok ASF, McMahon BJ. Chronic hepatitis B: update 2009. Hepatology. 2009;50:661-662.
3. McMahon BJ. Epidemiology and natural history of hepatitis B. Semin Liver Dis. 2005;25: Suppl 1:3-8.
4. Yim HJ, Lok ASF. Natural history of chronic hepatitis B virus infection: what we knew in 1981 and what we know in 2005. Hepatology. 2006:S173-81.
5. Chen CJ, Yang HI, Su J, et al. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA. 2006:65-73.
6. Hadziyannis SJ, Tassopoulos NC, Heathcote EJ, et al. Long-term therapy with adefovir dipivoxil for HBeAg-negative chronic hepatitis B for up to 5 years. i. 2006;131:1743-1751.
|
| |
|
 |
 |
|
|