 |
 |
 |
| |
Higher Sustained Post-Treatment Virologic Response Following Peginterferon Alfa-2a ± Adefovir Compared With Adefovir Monotherapy in HBeAg-Positive Patients
|
| |
| |
Reported by Jules Levin
AASLD Nov 3 2009, Boston, MA, USA
Ma W-M,1 Zhou B-P,1,2 Ao F-J,2 Zhou D-Q,3 Hu Y-W,2 He Q,2 Dai W,2 Xu C,2 Chen L-J,4 Liu Y,1 Nie G1
1Shenzhen Municipal Institute of Hepatology, Shenzhen, China; 2Shenzhen Municipal Third People's Hospital, Guangdong, China; 3Shenzhen Traditional Chinese Medicine Hospital, Guangzhou, China; 4Peking University Shenzhen Hospital, Peking, China
Presented at the 60th Annual Meeting of the American Association for the Study of Liver Diseases 30 October - 3 November 2009, Boston, USA
Summary
Peginterferon alfa-2a ± adefovir combination therapy resulted in significantly greater rates of HBeAg clearance and HBeAg seroconversion than adefovir monotherapy
Significantly more patients receiving combination therapy achieved an ontreatment decline in HBV DNA >3 log10 copies/mL during treatment than either monotherapy group
HBsAg clearance and HBsAg seroconversion were achieved post-treatment
by patients in the peginterferon alfa-2a monotherapy and peginterferon alfa-2a + adefovir combination group, but not in the adefovir group
The dose of peginterferon alfa-2a administered was lower than the normal standard dose
Conclusions
Peginterferon alfa-2a + adefovir combination therapy was not significantly superior to peginterferon alfa-2a monotherapy in achieving a post-treatment response. However, the greater HBV DNA suppression observed during combination therapy is encouraging and the potential for combination therapy with other antiviral agents, extended treatment duration and different treatment schedules requires further investigation
Background
Successful treatment of chronic hepatitis B (CHB) results in decreased rates of cirrhosis, liver failure and hepatocellular carcinoma.1,2,3 There is increasing evidence that only a sustained immune response to the hepatitis B virus (HBV) will result in control and elimination of the virus and subsequent prevention of disease progression4,5
A finite course of interferon-based therapy is associated with sustained HBeAg seroconversion post-treatment in approximately one-third of patients5,6
Previous studies in HBeAg-positive patients have speculated that combining the immunomodulatory properties of interferon with the strong antiviral potency of the nucleos(t)ide analogs may increase the rate of sustained response in CHB.5,6
Results from these initial studies did not demonstrate any superiority of combination therapy with lamivudine over pegylated interferon monotherapy in achieving a post-treatment response; however, to date, the potential of combining other nucleos(t)ide analogs with pegylated interferons has not been investigated
In this study we determined the efficacy of peginterferon alfa-2a, adefovir or the combination in patients with HBeAg-positive CHB

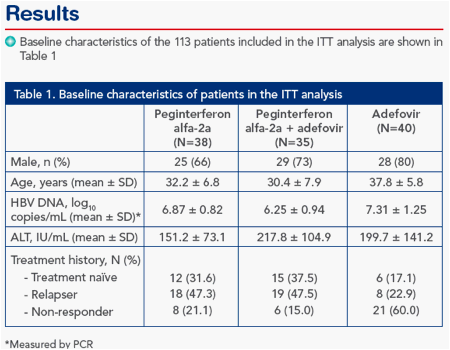
HBeAg response during and post-treatment
Peginterferon therapy, alone or in combination with adefovir, resulted in significantly higher rates of HBeAg clearance and HBeAg seroconversion than adefovir monotherapy (Figure 1)
Figure 1. Peginterferon alfa-2a ± adefovir is associated with significantly higher rates of (A) HBeAg clearance and (B) HBeAg seroconversion on-treatment and 48 weeks post-treatment (week 96) than adefovir monotherapy

A
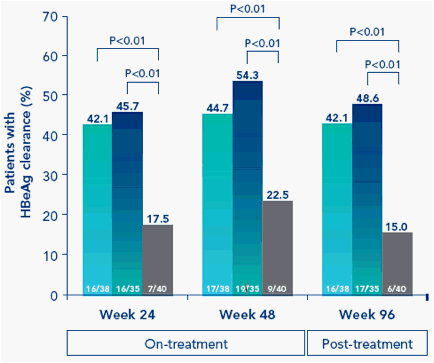
B
Virologic response during and post-treatment
Peginterferon alfa-2a + adefovir combination therapy resulted in substantial HBV DNA suppression on-treatment (Figure 2)
Figure 2. Significantly more combination therapy patients achieved a >3 log10 decline in HBV DNA from baseline during treatment than either monotherapy group

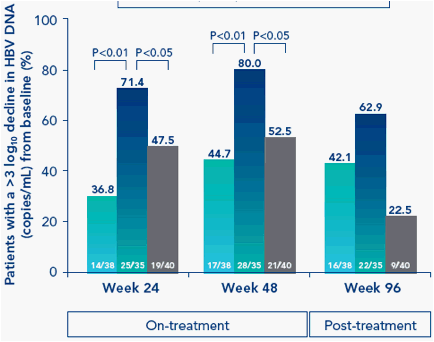
Substantially more of the combination therapy group had HBV DNA levels below 500 copies/mL both on-treatment and post-treatment; however, this did not reach statistical significance (Table 2)
Rate of ALT normalization continued to increase to week 48 and to week 96 in peginterferon alfa-2a monotherapy group, but decreased between weeks 48 and 96 in the combination therapy and adefovir monotherapy group
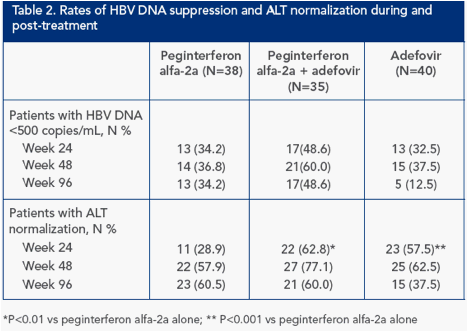
HBsAg response post-treatment
One patient (2.6%) in the peginterferon alfa-2a monotherapy group and one patient in the combination therapy group (2.9%) achieved HBsAg clearance at week 96 (48 weeks post-treatment) compared with none of the patients in the adefovir monotherapy group
Similarly, one patient (2.6%) in the peginterferon alfa-2a monotherapy group and one patient (2.6%) in the combination therapy group (2.9%) achieved HBsAg seroconversion at week 96 (48 weeks post-treatment) compared with none of the
patients in the adefovir monotherapy group
References
1. Niederau C et al. Long-term follow-up of HBeAg-positive patients treated with interferon alfa for chronic hepatitis B. New Engl J Med 1996; 334:1422-1427
2. Lin SM et al. Interferon therapy in HBeAg-positive chronic hepatitis reduces progression to cirrhosis and hepatocellular carcinoma. J Hepatol 2007; 46:45-52
3. Sung JJ et al. Meta-analysis: treatment of hepatitis B infection reduces risk of
hepatocellular carcinoma. Aliment Pharmacol Ther 2008; 28:1067-2077
4. Chisari FV, Ferrari C. Hepatitis B virus immunopathogenesis. Annu Rev Immunol
1995; 13:29-60
5. Janssen H et al. Pegylated interferon alfa-2b alone or in combination with
lamivudine for HBeAg-positive chronic hepatitis B: a randomized trial. Lancet
2005; 365:123-129
6. Lau GKK et al. Peginterferon alfa-2a, lamivudine, and the combination for
HBeAg-positive chronic hepatitis B. New Engl J Med 2005; 352:2682-2695
Disclosure information:
Editorial support for the development of this poster was provided by Elements
Communications and funded by F. Hoffmann-La Roche, Basel, Switzerland
|
| |
|
 |
 |
|
|