 |
 |
 |
| |
Use of the Elecsys® HBsAg II assay for simple and
accurate quantification of HBsAg levels in sera
of patients infected with HBV
|
| |
| |
Reported by Jules Levin
Bonino F1, Brunetto MR3, Colombatto P3, Moriconi F2, Torresani E2, Lunghi G2, Ofenloch B4,
Melchior W4, Upmeier B4, Roessler D4, Lilienfeld BG4, van der Helm W4
1. Scientifi c Direction and 2. Central Laboratory of the IRCCS Policlinic of Milan, Italy
3. Hepatology Unit Laboratory of the University Hospital of Pisa, Italy
4. Roche Diagnostics AG, Rotkreuz, Switzerland
Background:
An estimated 350 million persons worldwide are chronically infected with HBV resulting in increased risk of developing cirrhosis, hepatic decompensation, and hepatocellular carcinoma1.
After infection, HBsAg is the fi rst immunological marker detectable in serum. Sustained HBsAg loss is considered as the ideal endpoint of therapy in HBeAg-positive and HBeAg-negative patients. This is associated with a complete and definitive remission of the activity of chronic hepatitis B and an improved long-term outcome2.
Recent studies suggest that quantification of HBsAg levels during treatment and at the end of treatment can be used to predict long term post-treatment response to peginterferon alfa-2a3-4 and optimize treatment strategies5.
Roche's Elecsys HBsAg II is a sensitive assay for the detection of HBsAg. Recent studies in Asia have confirmed that the high sensitivity and specificity of Elecsys HBsAg II allows early detection of HBV infection and mutant recognition6, 7.
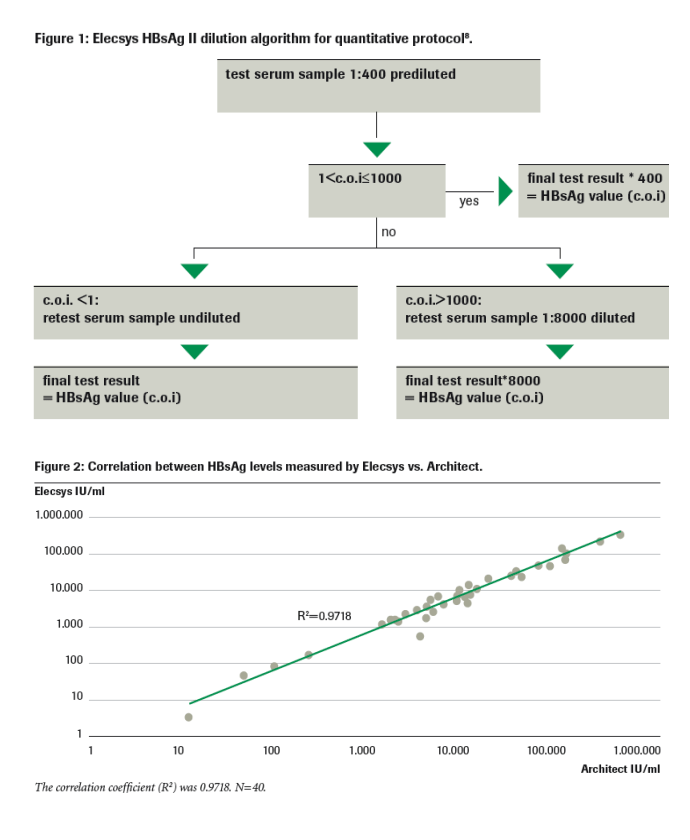
We have previously shown that this assay, currently available as a qualitative assay for HBsAg, can be used effectively for the quantification of HBsAg levels using a simple dilution algorithm (see methods section)8.
Aim:
Comparison of the newly developed Roche Elecsys HBsAg II dilution algorithm with the Abbott Architect HBsAg assay.
Methods:
HBsAg was quantified using the Architect® HBsAg assay (Abbott Laboratories; dynamic range 0.05-250.0 IU/mL, WHO standard) and the Elecsys HBsAg II dilution algorithm (standardized to WHO standard 00-588, conversion factor for estimation of HBsAg units: coi of 1 = 0.064 IU/ml).
The protocol for the Architect assay was as follows: After default 1:100 dilution with the serum diluent provided by the manufacturer, samples with HBsAg levels higher than 250.0 IU/mL were retested at a final dilution of 1:1000. Samples with HBsAg levels <0.05 IU/mL at 1:100 dilution were retested undiluted.
The protocol for the Elecsys HBsAg II dilution algorithm was as follows: All samples were tested at a 1:400 dilution in serum negative for both HBsAg and anti-HBs. If result revealed cut off index (c.o.i.) of >1 then the final result is c.o.i.*400. If c.o.i <1 then the sample is retested undiluted and final result is result of retest (c.o.i). If c.o.i ≥1000 then the sample is retested at a 1:8000 dilution and final result is c.o.i.*8000 (figure 1).
Results:
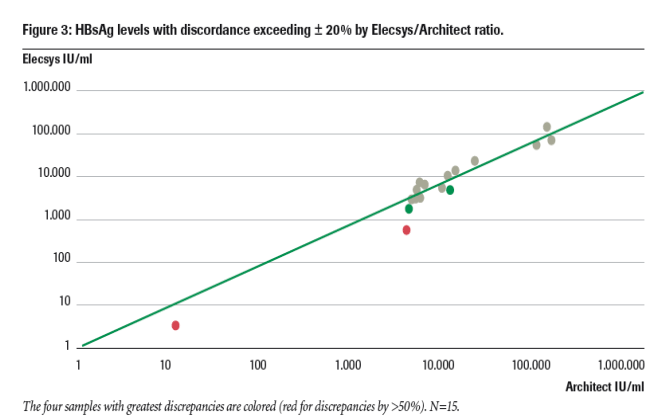
As shown in figure 2, HBsAg levels measured with the two assays correlated well (R2=0.9718, n=40) over a 5 log range (3 - 3x105 IU/mL).
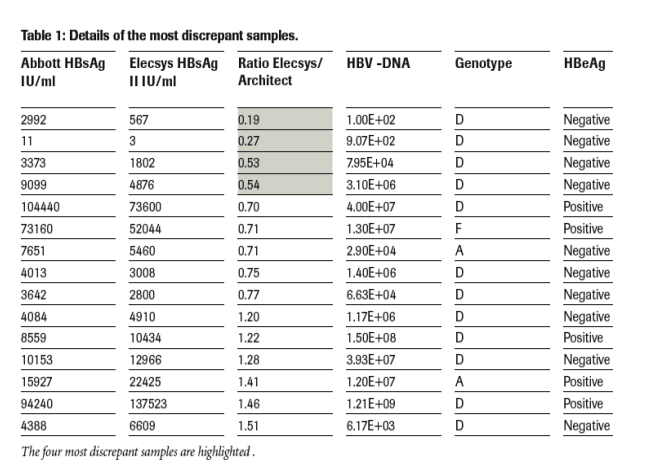
Discrepancies in HBsAg levels >±20% were reported for a minority of the samples (n=15), mainly distributed evenly above and below the ideal correlation line (figure 3 and table 1).
In the four samples with greatest discrepancy (all of them with low titre, range 3 - 9x103 IU/mL), Elecsys showed lower values (in two cases by >50%, figure 3).
Conclusions:
The Roche Elecsys HBsAg II assay provides a simple and reliable means for determining HBsAg levels and shows a high correlation to the Abbott Architect HBsAg assay.
Only a minor portion of the samples tested showed discrepancies of >±20%. It is worth mentioning that the four samples with greatest discrepancies were of low viral load titre (3 - 9x103 IU/mL) and might be overestimated by the Architect HBsAg assay.
This simple assay protocol could be used to provide useful information during on-treatment monitoring of HBsAg levels to predict the long term outcome of HBV infec tion in patients with chronic hepatitis B undergoing peginterferon alfa-2a therapy and optimized treatment strategies.
References:
1. Chronic hepatitis B, Anna S. F. Lok and Brian J. McMahon, Hepatology 2007;45:507-539
2. EASL Clinical Practice Guidelines: Management of chronic hepatitis B, EASL, Journal of Hepatology 50, 2009
3. On-treatment HBsAg decline during peginterferon alfa-2a (40KD) ± lamivudine in patients with HBeAg-positive CHB as a potential predictor of durable off-treatment response. Lau GKK et al., AASLD 2008 abstract, poster 910
4. In patients with HBeAg-negative chronic hepatitis B HBsAg serum levels early during treatment with peginterferon alfa-2a
predict HBsAg clearance 4 years post-treatment. Marcellin P et al., AASLD 2008 abstract, poster 919
5. On-treatment quantifi cation of HBsAg in patients with HBeAg-negative chronic hepatitis B treated with peginterferon alfa-2a may help identify those likely to be cured by this therapy and optimize treatment strategies Brunetto MR et al., Hepatitis B virus surface antigen levels - a guide to sustained response to peginterferon alfa-2a in HBeAg negative chronic hepatitis B: Hepatology 2009, in press
6. Comparison of the sensitivity and specifi city of the Elecsys HBsAg II assay with other available assays in China for detection of HBsAg. Jia J et al. APASL 2009 poster PE073.
7. Comparison of the technical and clinical performance of the Elecsys HBsAg II assay with the Architect, AxSym and Advia
Centaur HBsAg screening assays. Louisirirotchanakul S et al. APASL 2009 poster PE123
8. Quantitative Assessment Of Serum HBsAg Levels Using The Elecsys HBsAg II Screening Assay: Results Of A Feasibility Study.
Bonino F et al., AASLD 2008 abstract, poster 934
COBAS, ELECSYS, LIFE NEEDS ANSWERS and MODULAR are trademarks of Roche.
All other trademarks are the property of their respective owners.
2009 Roche
|
| |
|
 |
 |
|
|