 |
 |
 |
| |
HBsAg Clearance Continues to Increase After the End of Treatment With PEGASYS ± Lamivudine: 5-year Follow-up Study in Patients with HBeAg-Negative Disease
|
| |
| |
Reported by Jules Levin
Presented at the 19th Conference of the Asia Pacific Association for the Study of the Liver, 13-16 February 2009, Hong Kong, China
Marcellin P,1 Piratvisuth T,2 Brunetto M,3 Bonino F,4 Lau GKK,5 Farci P,6 Yurdaydin C,7 Gurel S,8 Wu J,9 Popescu M10
1Service d'Hepatologie and Centre de Recherches Biologiques Bichat Beaujon (Inserm CRB3), University of Paris, Clichy, France. 2NKC Institute of Gastroenterology and Hepatology, Department
of Internal Medicine, Songklanagarind Hospital, Prince of Songkla University, Hat Yai 90110, Thailand. 3UO, Gastroenterologia ed Epatologia, Azienda Ospedaliera Universitaria Pisana, Pisa, Italy.
4Foundation IRCCS, Policlinico of Milan and University of Pisa, Italy. 5Department of Medicine, Queen Mary Hospital, The University of Hong Kong, Hong Kong, China. 6Universita di Cagliari,
Cagliari, Italy; 7University of Ankara, Faculty of Medicine, Ankara, Turkey. 8Uludag University, Bursa, Turkey. 9Roche Dee Why, Australia. 10F. Hoffmann-La Roche Ltd, Basel, Switzerland
Author Summary
The improvements in virologic and biochemical response observed previously with PEGASYS-based therapy at 6 months post treatment and at years 1, 2, 3 and 4 post treatment were still apparent 5 years after completion of PEGASYS ± lamivudine therapy
Importantly, the rate of HBsAg clearance in PEGASYS-treated patients increased from 10.9% at year 4 to 12.2% at year 5 highlighting that PEGASYS is associated with a long-term durable post-treatment response
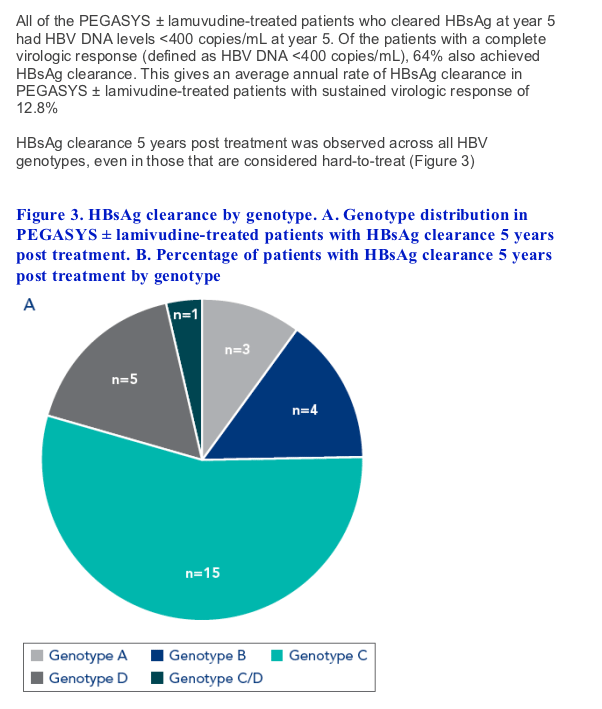
All patients clearing HBsAg at year 5 had HBV DNA <400 copies/mL at year 5 giving an average annual rate of HBsAg clearance of 12.8% in these patients
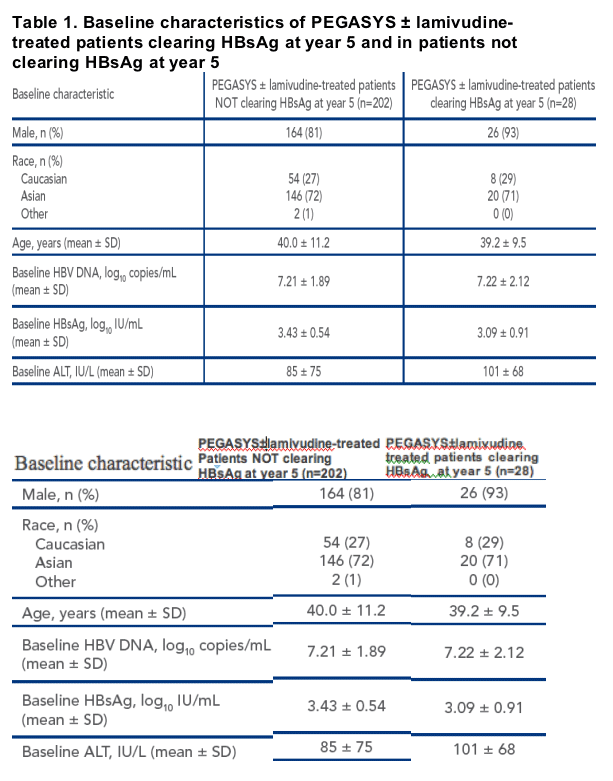
Although most baseline characteristics of patients clearing HBsAg at year 5 were similar to those not achieving this endpoint, baseline HBsAg levels were lower and baseline
ALT levels were higher in patients achieving HBsAg clearance. HBsAg clearance was observed in patients infected with all HBV genotypes including those considered to be hard-to-treat
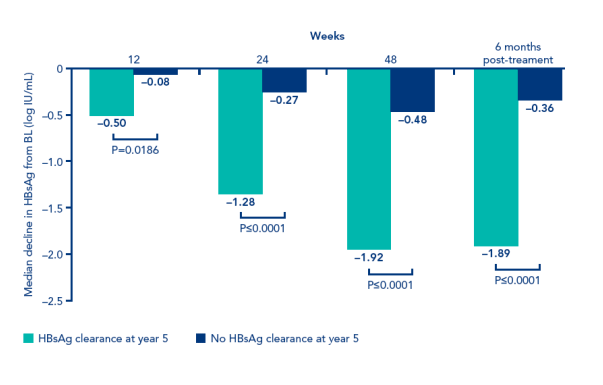
HBsAg decline during treatment was significantly greater in patients with HBsAg clearance, than in non responding patients.
This is an interesting observation that might allow quantitative HBsAg to be used as an early on-treatment predictor of long-term HBsAg response
AUTHOR CONCLUSIONS
The ability of a finite course of PEGASYS to induce sustained response with increasing HBsAg clearance rates during post-treatment follow up supports its use as first-line therapy in HBeAg-negative patients with CHB. The current analysis has shown that HBsAg decline during treatment is associated with long-term response. Consequently, quantification of HBsAg during treatment may be used to predict long-term response to PEGASYS-based therapy
Background
HBsAg clearance or seroconversion (loss of HBsAg and appearance of anti-HBs) can be considered the closest step we can achieve to clinical cure of chronic hepatitis B (CHB) infection
In a large, multinational phase III study, we demonstrated that HBsAg clearance could be achieved in patients with HBeAg-negative disease following 48 weeks treatment with PEGASYS ± lamivudine, but not in those treated with lamivudine alone1
In a long-term observational follow-up study we found that the rate of HBsAg clearance in the patients treated with PEGASYS ± lamivudine increased from 3% 6 months post treatment to 11% 4 years after the end of treatment2
Recent data have shown that patients treated with a 48-week course of PEGASYS, with or without lamivudine, can induce a marked decline in HBsAg which is sustained 6 months post treatment.3 This decline in HBsAg was not achieved by lamivudine monotherapy
We have also observed previously that HBsAg levels at the end of treatment were associated with HBsAg clearance 3 years post treatment in patients treated with PEGASYS ± lamivudine4
Five-year data from an analysis of the observational follow-up study are now available and should help to identify factors that are associated with favorable outcomes many years after completion of therapy
Objective
To determine 5-year response rates in patients with HBeAg-negative CHB who had received PEGASYS alone or in combination with lamivudine or lamivudine alone as part of the phase III clinical trial and entered the observational follow-up study
Methods
In a randomized multinational trial, HBeAg-negative patients received PEGASYS (180 μg/week, n=177), PEGASYS + lamivudine (100 mg/day, n=179), or lamivudine alone (n=181) for 48 weeks (initial study). Patients were followed up for an additional 24 weeks and the original endpoint of the study was 6 months post treatment
All participating centers in the initial study were invited to participate in a separate observational follow-up protocol (follow-up study), in which patients were followed for up to 5 years after completion of treatment
Parameters assessed at year 5 were:
· HBV DNA <10,000 copies/mL (1785 IU/mL)*
· HBV DNA <20,000 copies/mL (3571 IU/mL)*
· HBV DNA <400 copies/mL (71 IU/mL)*
· HBsAg clearance
· HBsAg seroconversion
· ALT normalization
* HBV DNA levels were measured using the COBAS AMPLICOR HBV MONITOR, which has a conversion factor of 1 IU/mL = 5.6 copies/mL
Quantitative HBsAg in serum was measured pre-treatment, on-treatment (weeks 12, 24 and 48) and 6 months post treatment (week 72) using the Abbott Architect HBsAg assay in available stored sera
Patients receiving either registered or investigational therapy for CHB during the follow-up period were excluded from the follow-up phase and were regarded as non responders. Similarly, patients who had an ALT value >2 times the upper limit of normal or HBV DNA levels >100,000 copies/mL 12 months post treatment discontinued the study and were considered to be non responders in the analyses
Results
Results from the initial study
The effect of PEGASYS ± lamivudine compared with lamivudine alone on virologic and biochemical endpoints has been published previously.1 Briefly, 6 months post treatment PEGASYS treatment was superior to lamivudine and no additional benefits were observed with combination therapy:
The rate of ALT normalization was significantly higher in patients treated with PEGASYS alone (P=0.004) or in combination with lamivudine (P=0.003) compared with that in patients receiving lamivudine alone
The proportion of patients with HBV DNA <20,000 copies/mL 6 months post treatment was higher in PEGASYS ± lamivudine-treated patients (43% for PEGASYS alone and 44% for PEGASYS + lamivudine) compared with lamivudine-treated patients (29%; P=0.007 and P=0.003, respectively)
Rates of sustained HBV DNA suppression to below 400 copies/mL were 19% and 20% for PEGASYS with or without lamivudine, respectively, compared with 7% with lamivudine alone (P<0.001 for both)
HBsAg clearance was only observed in patients receiving PEGASYS either alone or in combination with lamivudine (n=12)
Results from the 5-year analysis
Of the 54 centers involved in the initial study, 42 participated in the long-term study - contributing 315 (59%) of the original 537 patients. Significantly more patients who received PEGASYS ± lamivudine (66% and 64%, respectively) than lamivudine alone (47%; P <0.01 vs PEGASYS ± lamivudine) in the initial study went on to participate in the long-term study
230 patients who had received PEGASYS either alone or in combination with lamivudine and 85 patients who had received lamivudine alone participated
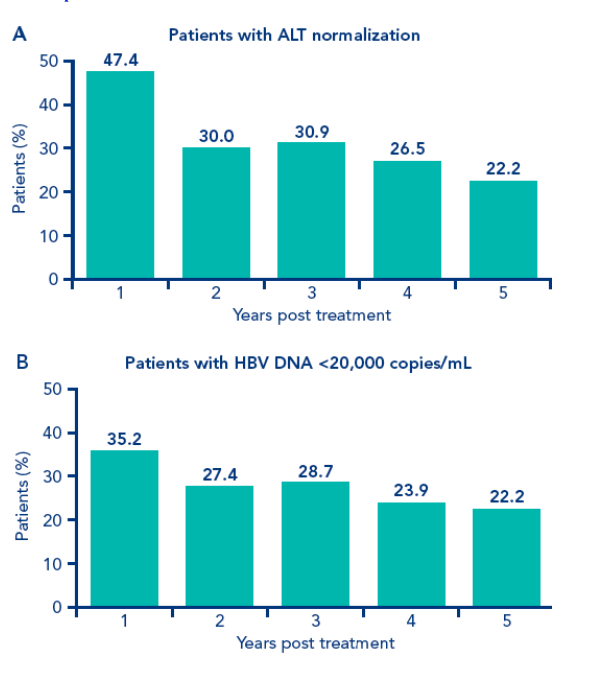
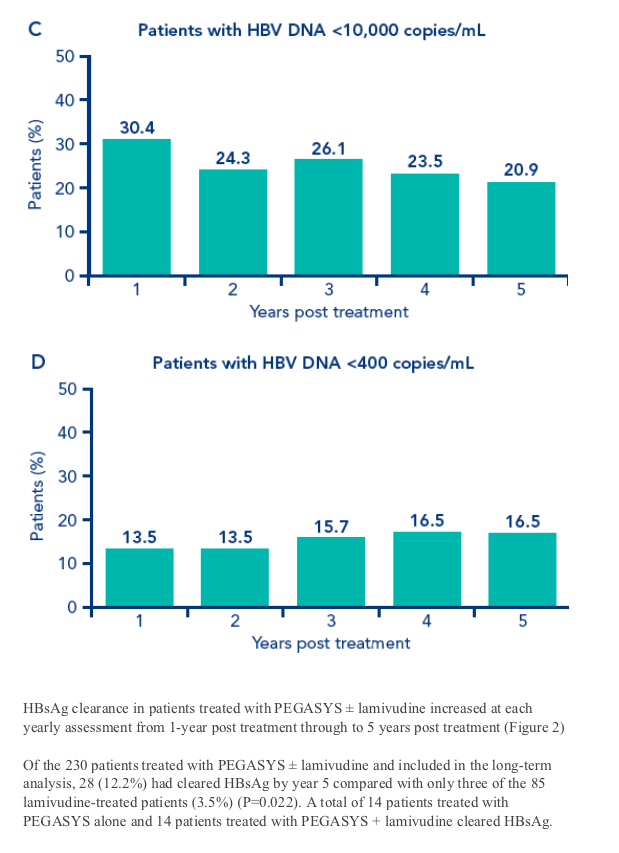
The long-term virologic and biochemical effects over time of a 48-week course of treatment with PEGASYS ± lamivudine in patients participating in the long-term study are shown in Figure 1
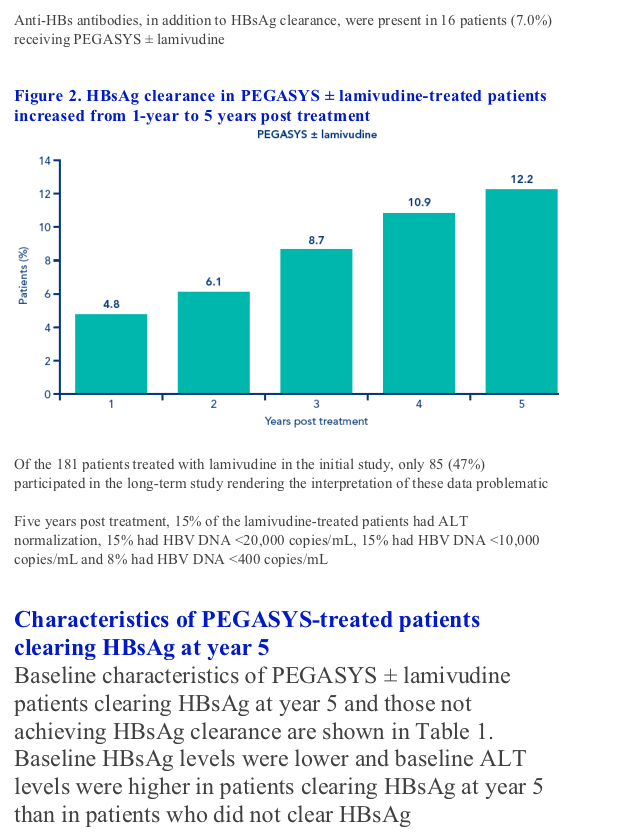
Figure 1. Percentage of patients treated with PEGASYS ± lamivudine in the long-term study who achieved biochemical and virologic responses 1, 2, 3, 4 and 5 years post treatment (n=230). A. Patients with ALT normalization; B. Patients with HBV DNA <20,000 copies/mL; C. Patients with HBV DNA <10,000 copies/mL; D. Patients with HBV DNA <400 copies/mL
HBsAg clearance in patients treated with PEGASYS ± lamivudine increased at each yearly assessment from 1-year post treatment through to 5 years post treatment (Figure 2)
Of the 230 patients treated with PEGASYS ± lamivudine and included in the long-term analysis, 28 (12.2%) had cleared HBsAg by year 5 compared with only three of the 85 lamivudine-treated patients (3.5%) (P=0.022). A total of 14 patients treated with PEGASYS alone and 14 patients treated with PEGASYS + lamivudine cleared HBsAg. Anti-HBs antibodies, in addition to HBsAg clearance, were present in 16 patients (7.0%) receiving PEGASYS ± lamivudine

References
1. Marcellin P et al. Peginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-1. negative chronic hepatitis B. N Engl J Med 2004;351:1206-1217
2. Marcellin P et al. In patients with HBeAg-negative chronic hepatitis B, HBsAg serum levels early during treatment with peginterferon alfa-2a predict HBsAg clearance 4 years post treatment. Hepatology 2008;48(suppl 1):919A
3. Brunetto M et al. HBV DNA suppression induced by peginterferon alfa-2A (40KD), but not by lamivudine, results in HBsAg loss and seroconversion at 3 years post treatment. J Hepatol (EASL 2008)
4. Marcellin P et al. Virological and biochemical response in patients with HBeAg-negative chronic hepatitis B treated with peginterferon alfa-2a (40KD) with or without lamivudine: results of 4-year follow-up. J Hepatol 2008;28(suppl2):S46
Disclosure information: Editorial support for the development of this poster was funded by F. Hoffmann-La Roche, Basel, Switzerland
|
| |
|
 |
 |
|
|