 |
 |
 |
| |
RDEA427 and RDEA640 are Novel NNRTIs with Potent Anti-HIV Activity Against NNRTI-Resistant Viruses
|
| |
| |
Reported by Jules Levin
CROI 2009 Feb 8-12 Montreal
A. Raney, R. Hamatake, W. Xu, J.- M. Vernier, J.- L. Girardet, P. Weingarten, D. Zhou, H. K. Kim, R. Dick, L.- T. Yeh, B. Quart
Ardea Biosciences, Inc., CA
AUTHOR CONCLUSIONS
RDEA427 and RDEA640 exhibit excellent HIV antiviral activity.
RDEA427 and RDEA640 are comparable or superior to other NNRTIs versus
NNRTI-resistant viruses and clinical isolates.
RDEA427 and RDEA640 show a low potential for CYP induction and a large
selectivity index.
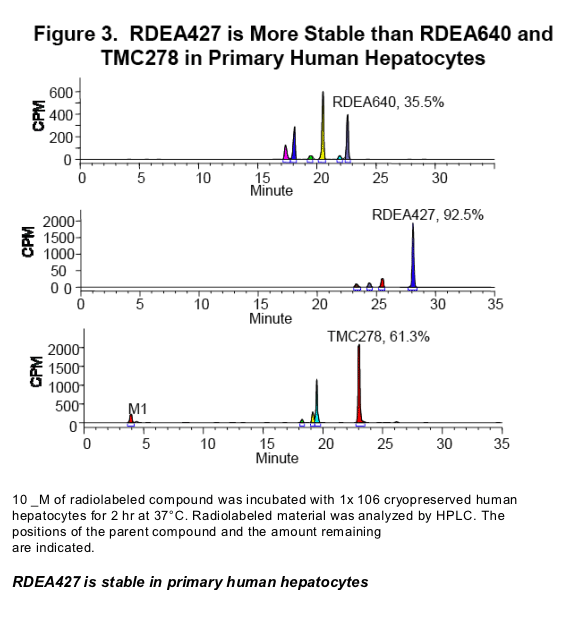
RDEA427 has better metabolic stability and lower covalent binding potential than
RDEA640 or TMC278.
RDEA427 shows strong potential for improved performance over current NNRTIs
and is undergoing preliminary clinical evaluation.
ABSTRACT
Background: Non-nucleoside reverse transcriptase inhibitors (NNRTIs) constitute an important class of drugs used in HIV-1 therapy. Mutant viruses resistant to the most widely used NNRTIs, efavirenz and nevirapine, lead to treatment failure. The characterization of activities against a panel of NNRTI-resistant HIV-1 and in vitro toxicological studies suggest the novel NNRTIs RDEA427 and RDEA640 have the potential to overcome the most prevalent of these resistant strains.
Methods: Antiviral activities of the NNRTIs were determined using VSV-g pseudotyped HIV-1 containing wild-type (wt) and NNRTI-resistant sequences. These assays were also performed with human serum and human serum albumin (HSA) to assess the impact of protein binding on antiviral activity. Cytotoxicity was evaluated in primary human cells and cell lines. Non-linear
regression analysis was used to calculate IC50 values. CYP3A4 induction was determined in vitro using a CYP3A4 promoter-driven luciferase reporter cell line carrying the pregnane X receptor.
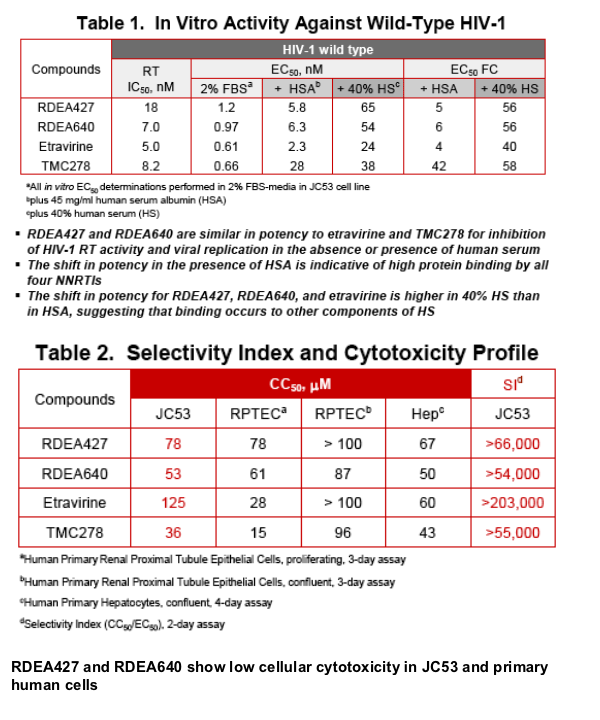
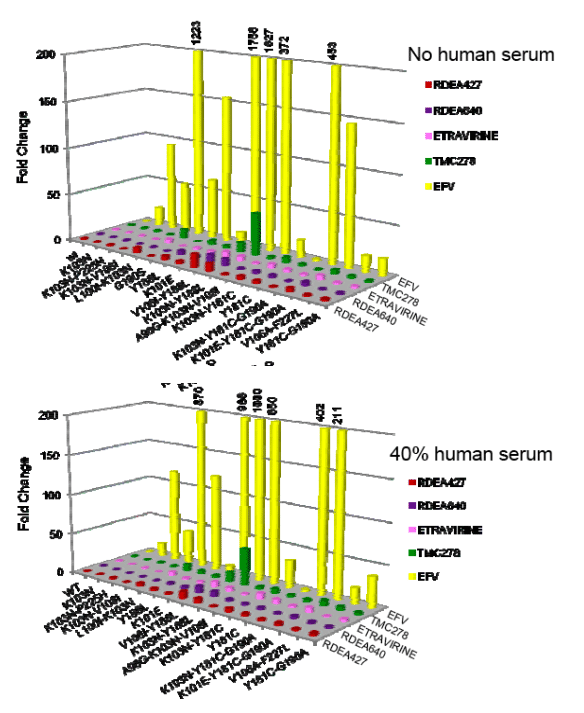
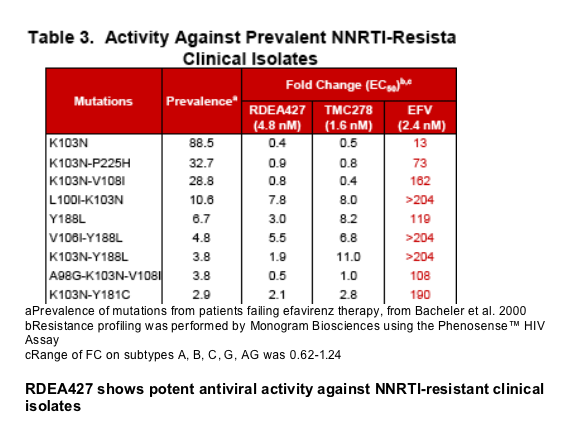
Results: RDEA427 and RDEA640 are potent inhibitors of wt HIV-1 with EC50 values of 0.9 nM and 0.8 nM, respectively, and CC50 values of >50 _M. The fold-changes (FCs) in EC50 against the major NNRTI-resistant viruses found in patients failing efavirenz therapy are significantly lower than those of efavirenz. FCs for both RDEA427 and RDEA640 against the K103N and G190S mutants are 0.9 and 0.3, respectively, versus 16 and 63 for efavirenz. While the K103N/L100I mutant caused a FC of 1700 for efavirenz, and 2.4 and 5.4 for etravirine and TMC278, respectively, the FC of RDEA640 was 0.4 against this mutant. RDEA640 had a 60-fold EC50 shift in the presence of 40% human serum and RDEA427 and RDEA640 had 9-fold shifts in the presence of 45
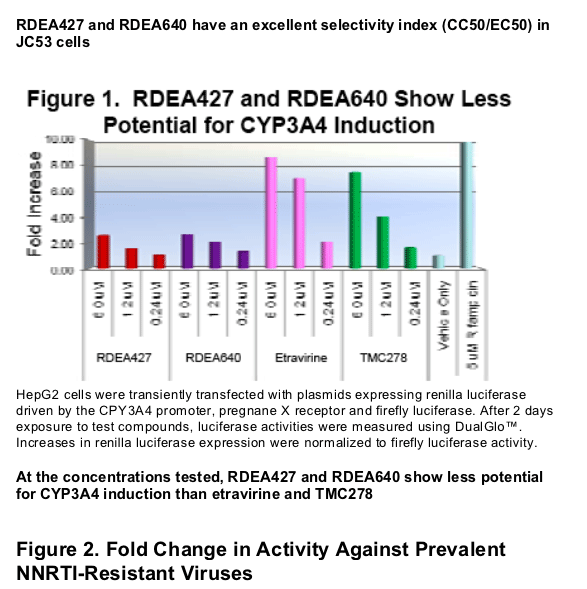
mg/ml HSA, values similar to the FCs for etravirine and better than those for efavirenz and TMC278. At 6 _M, RDEA427 and RDEA640 caused 2.6-fold increases in CYP3A4 promoter-driven luciferase activity compared with 8.7-fold and 7.5-fold increases for etravirine and TMC278, respectively.
Conclusions: RDEA427 and RDEA640 are superior to efavirenz against a panel of NNRTI-resistant viruses and are less affected by binding to serum proteins than efavirenz and TMC278. RDEA427 and RDEA640 also show a low potential for CYP induction and a large selectivity index. The in vitro characterization of these novel NNRTIs shows strong potential for improved performance over current NNRTIs and warrants evaluation in humans
INTRODUCTION
The NNRTI class of compounds is a critical component of current HIV therapies. Although NNRTIs are generally effective and have resulted in reduced AIDS-related morbidity and mortality, none of them is curative. Mutant viruses resistant to NNRTIs emerge rapidly and are cross-resistant to the two extensively used drugs, efavirenz and nevirapine. Although there are newer NNRTIs available or in development that are active against the NNRTI-resistant viruses, they have tolerability issues and significant drug interaction potential. NNRTIs with activity against resistant virus, excellent tolerability and reduced potential for drug interactions are needed to expand the NNRTI armamentarium. The novel NNRTIs under development by Ardea have the potential to meet these unmet medical needs.
METHODS
Antiviral activities of the NNRTIs were determined using VSV-g pseudotyped HIV-1 containing wild-type (wt) and NNRTI-resistant sequences. These assays were also performed with human serum and human serum albumin (HSA) to assess the impact of protein binding on antiviral activity. Inhibition of HIV-1 RT was determined by measurement of radioactive dATP incorporation into activated calf thymus DNA. Cytotoxicity was evaluated in primary human cells and a cell line using CellTiterGlo. Non-linear regression analysis was used to calculate IC50 values. CYP3A4 induction was determined in vitro using a CYP3A4 promoter-driven luciferase reporter cell line carrying the pregnane X receptor. Metabolic stability was measured in primary human hepatocytes, and covalent binding was measured in human microsomes.

|
| |
|
 |
 |
|
|