 |
 |
 |
| |
The Long-term Use of Tenofovir in HIV/HBV Co-infection Induces a Marked Decrease in Liver Fibrosis
|
| |
| |
Reported by Jules Levin CROI 2009 Feb 8-12
Karine Lacombe*1,2,3, A Boyd1, J Guechot2, D Wendum2,3, J-M Molina2, L Serfaty2, C Lascoux-Combe2, P Bonnard1,2, P Miailhes4, and P-M Girard1,2,3
1INSERM UMR-S707, Paris, France; 2AP-HP, Paris, France; 3UPMC, Paris VI, France; and 4Hospices Civils de Lyon, France
Background: The long-term effect of tenofovir (TDF) on liver fibrosis evolution has not been well characterized.
Methods: TDF-treated, co-infected patients from the French HIV/hepatitis B virus (HBV) cohort were included in the present analysis. The level of fibrosis was estimated using a biochemical score (Fibrometre). Time-weighted change from baseline (DAVG) was modelled at every 12 months after treatment initiation using GEE models adjusted for serum HBV DNA, age, CD4+ T cell count, body mass index, alcohol consumption, and hepatitis C or D co-infection. Crude change in METAVIR stage was also analysed on available pairs of liver biopsies performed at baseline and during follow-up.
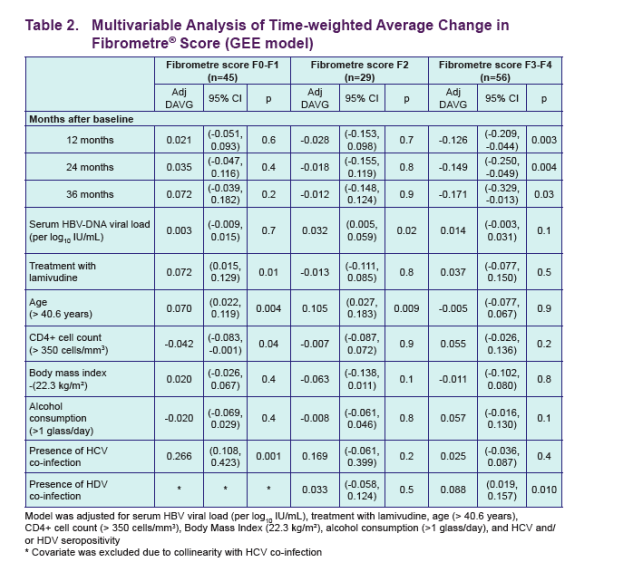
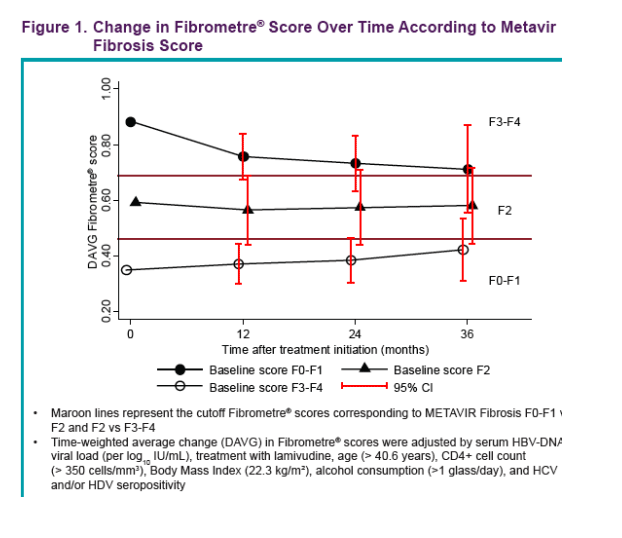
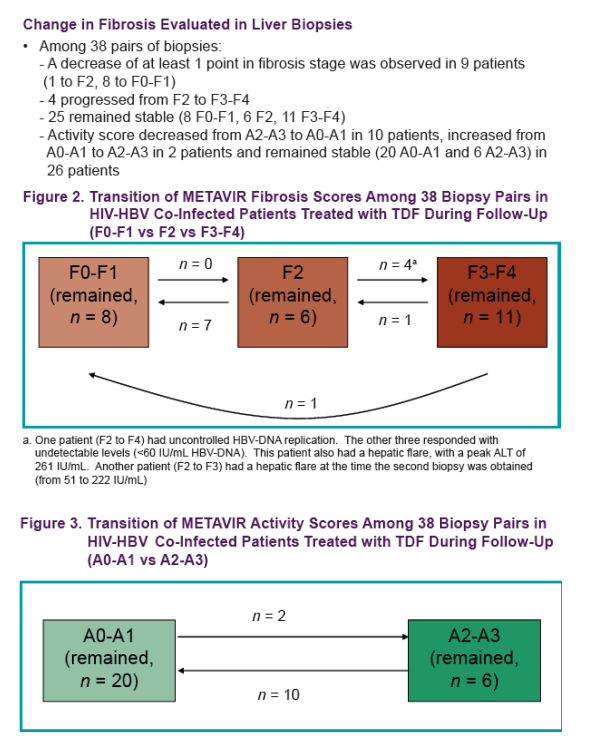
Results: We treated 130 co-infected patients (mean age years [SD] 40.9 [7.7]) with TDF and followed them for a median of 29.5 months (IQR 16.7). At baseline, 45 patients presented with Fibrometre stage F0-F1, 29 with stage F2, and 56 patients with stage F3-F4. The majority of patients were exposed to lamivudine before inclusion (mean duration 28.6 months [30.0]), and 76% were still co-treated with lamivudine/emtricitabine during follow-up. Overall, transaminases significantly decreased over time (mean ALT/AST [SD] at baseline, 24 months, and 36 months: 76 [90]/57 [46], 47 [40]/41 [30], and 39 [29]/33 [15], p = 0.002/<0.0001). Among patients with F3-F4 baseline fibrosis stages, there was a steep decline in fibrosis score at 12 months (adj DAVG = -0.126, 95%CI -0.209 to -0.044, p = 0.003) followed by a slow and stable decline at 24 and 36 months of treatment (adj DAVG = -0.149, 95%CI -0.250 to -0..049, p = 0.004 and adj DAVG = -0.171, 95%CI -0..329 to -0.013, p = 0.03, respectively). Patients with F0-F1 and F2 baseline fibrosis remained consistently stable over follow-up (adj DAVG at 36 months: 0.072, 95%CI -0.039 to 0.182, p = 0.2 and -0.012, 95%CI -0.148 to 0..124, p = 0.9, respectively) (figure1). Among 38 pairs of biopsies, a decrease of at least 1 point in fibrosis stage was observed in 9 patients (1 to F2, 8 to F0-F1) while 4 progressed from F2 to F3-F4 and 25 remained stable (8 F0-F1, 6 F2, 11 F3-F4). Activity score decreased from A2-A3 to A0-A1 in 10 patients, increased from A0-A1 to A2-A3 in 2 patients and remained stable (20 A0-A1 and 6 A2-A3) in 26 patients.
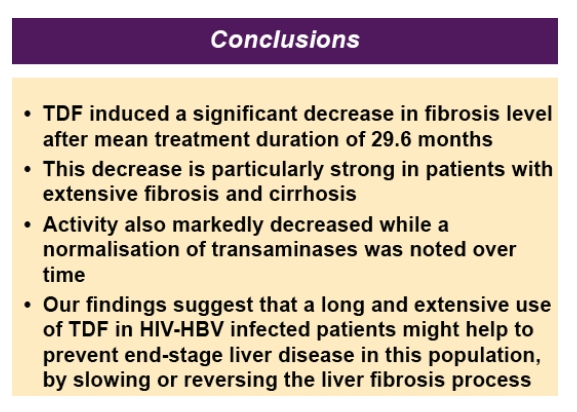
Conclusions: TDF induced a significant decrease in fibrosis level after mean treatment duration of 29.6 months. Our findings suggest extending use of TDF in HBV-infected patients.
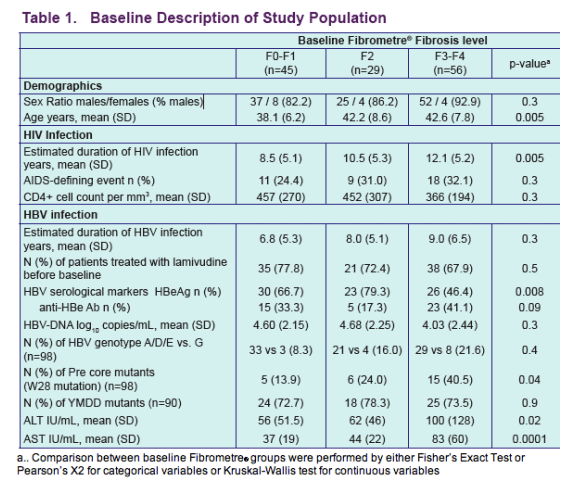
· 130 co-infected patients [mean age years (SD): 40.9 (7.7)] were treated with TDF and followed for a median of 29..5 months (IQR 16.7)
· At baseline, 45 patients presented with Fibrometer stage F0-F1, 29 with stage F2, and 56 patients with stage F3-F4
· The majority of patients were exposed to lamivudine before inclusion [mean duration: 28.6 months, SD (30.0)], and 76% were still co-treated with lamivudine/ emtricitabine during follow-up
· Transaminases significantly decreased over time [mean ALT/AST (SD) at baseline, 24 months, and 36 months: 76(90) / 57(46), 47(40)/41(30), and 39(29) / 33(15), p=0.002/<0.0001]
· Among patients with F3-F4 baseline fi brosis stages, there was a steep decline in fibrosis score at 12 months [adj DAVG=-0.126 (unadj DAVG= -0.139); 95% CI: -0.209, -0.044; p=0.003) followed by a slow and stable decline at 24 and 36 months of treatment [adj DAVG=-0.149 (unadj DAVG=-0.166); 95% CI: -0.250, -0.049; p=0.004 and adj DAVG=-0.171 (unadj=-0.169); 95% CI: -0.329, -0.013; p=0.03 respectively]
· Patients with F0-F1 and F2 baseline fi brosis remained consistently stable over follow-up [adj DAVG at 36-months: 0.072 (unadj DAVG=0.068); 95% CI: -0.039, 0.182; p=0.2 and -0.012 (unadj DAVG=-0.064); 95% CI (-0.148, 0.124); p=0.9 respectively]
|
| |
|
 |
 |
|
|