 |
 |
 |
| |
SIMPLIFICATION WITH FIXED-DOSE TENOFOVIR-EMTRICITABINE OR ABACAVIR-LAMIVUDINE
IN ADULTS WITH SUPPRESSED HIV REPLICATION (THE STEAL STUDY):
A RANDOMIZED, OPEN-LABEL, 96-WEEK, NON-INFERIORITY TRIAL
|
| |
| |
David A Cooper1, Mark Bloch2, Allison Humphries1, Janaki Amin1, David Baker3, Sean Emery1, Andrew Carr on behalf of the STEAL study group
1National Centre in HIV Epidemiology and Clinical Research, University of New South Wales; 2Holdsworth House Medical Practice; 3East Sydney Doctors; 4St Vincent's Hospital, Sydney, NSW, Australia
AUTHOR CONCLUSIONS
In this population, TDF-FTC and ABC-3TC had similar virological efficacy.
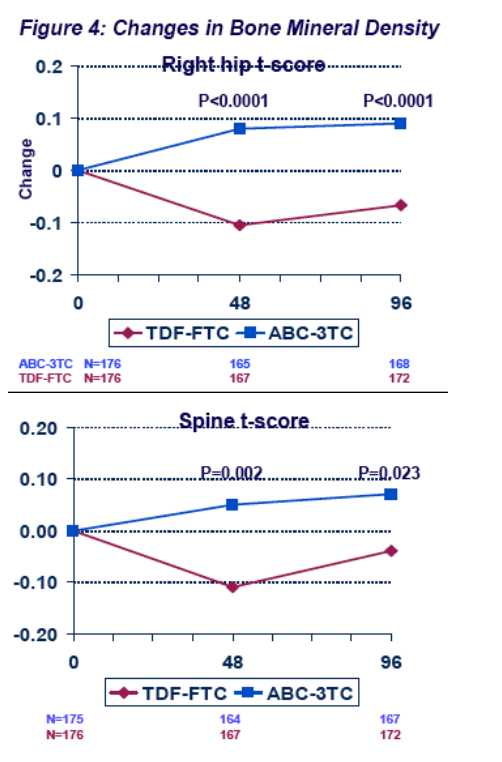
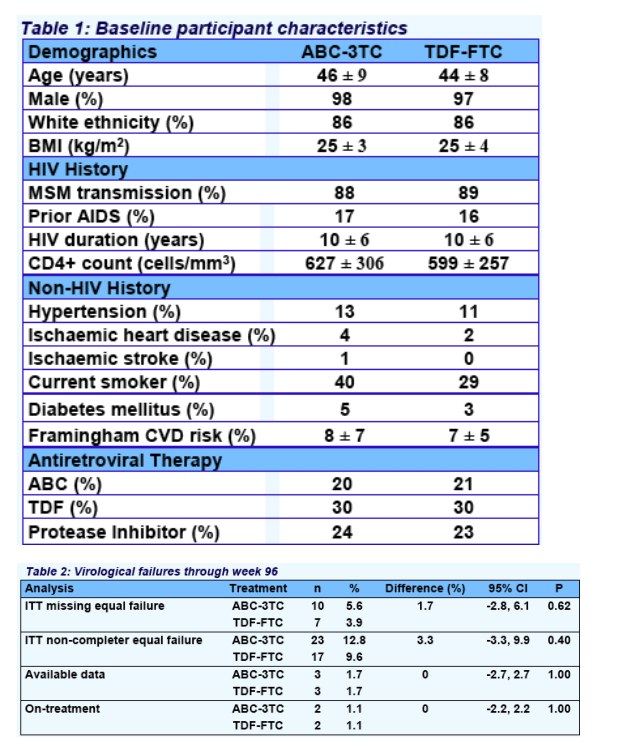
However, ABC-3TC was associated with more SNAEs (particularly cardiovascular disease) and lipid endpoints, and TDF-FTC caused more BMD loss.
Background: Two once-daily, dual-nucleoside, fixed-dose-combination (FDC) tablets are used for adult HIV-1 infection: tenofovir 300mg+emtricitabine 200mg (TDF-FTC); and abacavir 600mg+lamivudine 300mg (ABC-3TC). Which FDC is more effective and safe is uncertain.
Methods: We compared TDF-FTC and ABC-3TC-based therapy over 96 weeks
when either FDC was substituted for current NRTIs in HLA-B•5701-negative adults with plasma HIV viral load <50 copies/mL. The primary endpoint was virological failure, defined by repeat viral load >400 copies/mL plasma by intention-to-treat, missing=failure (ITTM=F) analysis. Secondary endpoints (ITT) included death, AIDS, serious non-AIDS events, metabolic parameters and body composition (bone/softtissue; ITT-LOCF). We used exact statistics for differences in proportions, T-tests to compare means and Cox regression for hazard ratios for ABC-3TC/TDF-FTC (HR 95%CI).
Results: 360 patients were randomized from January to August 2006. Key baseline characteristics of the 357 treated participants were: male 98%, mean age 45.1 years, prior NRTI therapy 5.8 years, current TDF 30%, current ABC 20%, current PI 24%, mean CD4 count 612 cells/mm3, eGFR 98 mL/min/1.73m2, limb fat 5.5kg, and hip t-score -0.49. Groups were well balanced, except smoking was more prevalent with ABC-3TC (40%) than with TDF-FTC (29%). 1.7% were lost to follow-up with no between-group difference. No patient developed AIDS or renal failure.
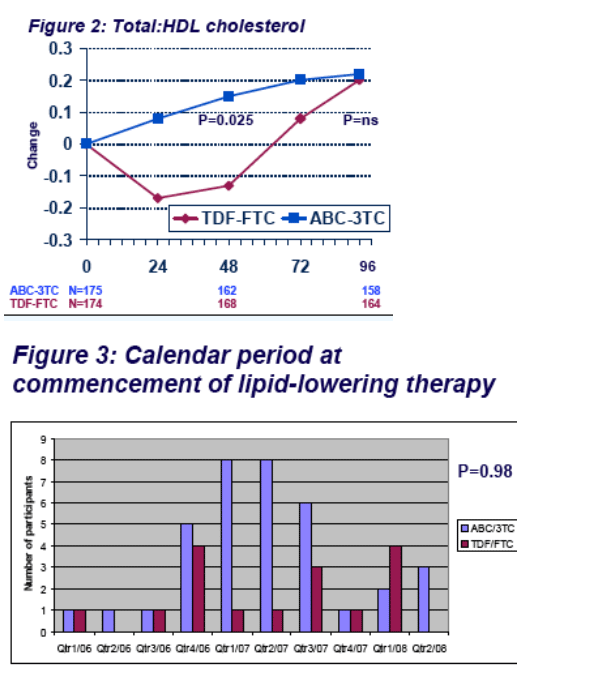
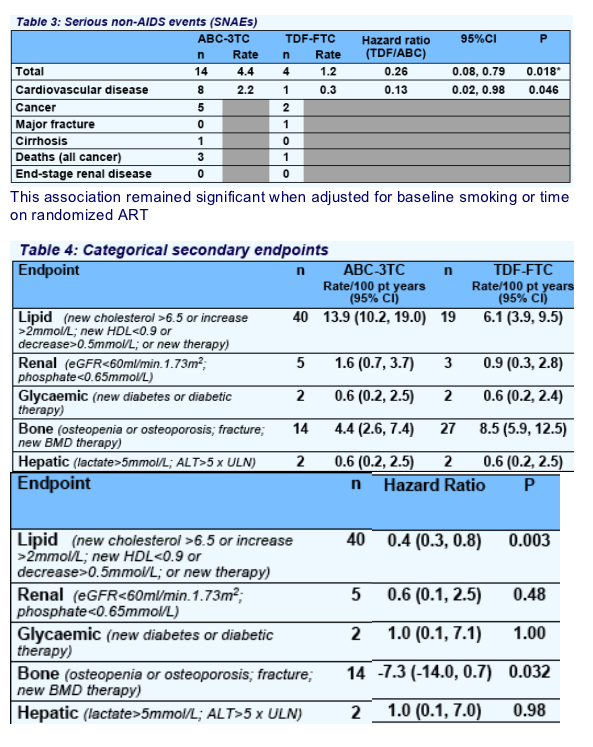
ABC-3TC was associated with more SNAEs (particularly cardiovascular disease) and lipid endpoints, and TDF-FTC caused more BMD loss. There was no significant between group, week-96 difference for limb fat, eGFR, CD4 count, insulin sensitivity, total:HDL cholesterol ratio, or lactate.
Conclusions: In this population, TDF-FTC and ABC-3TC had similar virological
efficacy and protection against AIDS. ABC-3TC was associated with more serious non-AIDS events.
Introduction
Two once-daily, dual nucleoside analogue, reverse transcriptase inhibitor (NRTI), fixed-dose-combination (FDC) tablets available:
-- tenofovir 300mg-emtricitabine 200mg (TDF-FTC)
-- abacavir 600mg-lamivudine 300mg (ABC-3TC)
-- Which FDC is more effective and safe is uncertain.
-- We hypothesized that switching to TDF-FTC would be virologically non-inferior to ABC-3TC over 96 weeks in HIV-infected adults with sustained suppression of HIV replication, but that TDF-FTC and ABC-3TC would have different safety profiles.
METHODS
Eligible participants randomly allocated 1:1 to continue their current NNRTI and/or PI and switch their NRTIs to either TDF-FTC or ABC-3TC.
Key eligibility criteria:
• Age ≥ 18 years
• on stable 2NRTI + NNRTI or PI ART ≥ 12 weeks
• HIV RNA <50 copies/mL plasma ≥12 weeks
• glomerular filtration rate (GFR) ≥ 70mL/min/1.73m2
• creatinine clearance ≥ 50 mL/min
• HLA-B•5701 negative (unless already on ABC)
• no prior hypersensitivity, intolerance or failure to study drugs
• no prior exposure to either study FDC drugs
• not on un-boosted atazanavir
• no previous non-traumatic fracture
Study visits at 0, 4, 12, 24, 36, 48, 60, 72, 84 and 96 weeks.
At each visit adverse events, concomitant medications, adherence, weight, biochemistry and HIV viral load were assessed; every 12 weeks blood count, liver function tests and CD4 count performed and blood stored; every 24 weeks quality of life (SF-8) and fasting metabolic measures conducted; every 48 weeks body composition measured by dual-energy x-ray absorptiometry
Primary endpoint was virological failure, defined by repeat viral load >400 copies/mL by intention-to-treat, missing=failure (ITTM=F) analysis. Secondary endpoints (ITT) included death, AIDS, serious
non-AIDS events, metabolic parameters and body composition (bone/soft-tissue; ITT-LOCF).
Exact statistics were used for differences in proportions, T-tests to compare means and Cox regression for hazard ratios. A sample of 175 participants per group yielded a 90% probability to detect a two-tailed 95% confidence interval of 15% around a 0% difference between treatment arms in virological failure rates.
RESULTS
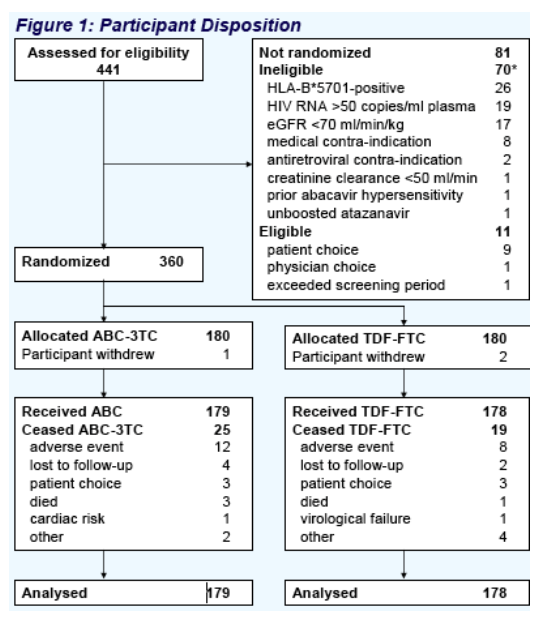
|
| |
|
 |
 |
|
|