 |
 |
 |
| |
Characterization of virologic failures in the randomized, controlled, Phase III ARTEMIS trial in treatment-naïve patients (Week 96 analysis)
|
| |
| |
Reported by Jules Levin
CROI Feb 2009 Feb 8-12 2009, Montreal
Inge Dierynck,1 Sandra De Meyer,1 Erkki Lathouwers,1 Carline Vanden Abeele,1 Ludo Lavreys,1 Marie-Pierre de Bethune,1 Gaston Picchio2
1Tibotec BVBA, Mechelen, Belgium; 2Tibotec Inc., Yardley, PA, USA
AUTHOR CONCLUSIONS
In this treatment-naïve ARTEMIS population, the VF rate was lower in patients receiving once-daily DRV/r 800/100mg compared with LPV/r 800/200mg total daily dose.
In this Week 96 resistance analysis, no major (primary) PI RAMs developed in VFs with an available genotype at baseline and endpoint. Almost all minor IAS-USA PI RAMs that developed were polymorphic.
All VFs with available phenotypes at baseline and endpoint remained
susceptible at endpoint to all PIs, including the study PI used in the regimen.
These findings confirm the lack of development of major (primary) PI RAMs, and the preservation of phenotypic susceptibility to all PIs in ARTEMIS patients with VF.
Introduction
The emergence of resistance to highly active antiretroviral (ARV) therapy presents a significant challenge to managing HIV-1 infection. Characterizing the development of resistance to new ARVs is needed to help improve treatment decisions.
The HIV-1 protease inhibitor (PI), darunavir (DRV; TMC114), has significant in-vitro ARV activity against both wild-type virus and multidrug-resistant HIV-1 strains.1 DRV has a high genetic barrier, which delays the development of resistance and allows the retention of antiviral activity despite the occurrence of mutations within the target viral protein.
DRV with low-dose ritonavir (DRV/r) at a dose of 600/100mg bid has been approved in the US2 and in Europe3 for the treatment of HIV-1 infection in treatment-experienced adult patients.
DRV/r 800/100mg qd is approved in the US2 and in Europe as part of HIV combination therapy in treatment-naïve adults.
In the ongoing, Phase III, ARTEMIS trial (TMC114-C211; AntiRetroviral Therapy with TMC114 ExaMined In Naïve Subjects), results of the 96-week analysis demonstrated that once-daily DRV/r 800/100mg achieved both primary (non-inferiority) and secondary (superiority) virologic endpoints (HIV-1 RNA <50 copies/mL) versus lopinavir (LPV) with low-dose ritonavir (LPV/r) 800/200mg total daily dose in treatment-naïve patients.4
At Week 96, 79% of the DRV/r patients achieved full virologic suppression (<50 copies/mL) compared with 71% of the LPV/r patients (estimated difference = 8.3%, 95% confidence interval [CI]: 1.8-14.7; intent-to-treat/time-to-loss of virologic response [ITT-TLOVR], p=0.012 for superiority).4
This poster presents the results of a detailed resistance characterization of ARTEMIS patients experiencing virologic failure (VF) with DRV/r or LPV/r, irrespective of HIV-1 RNA levels at endpoint.
Method
Design and patient population
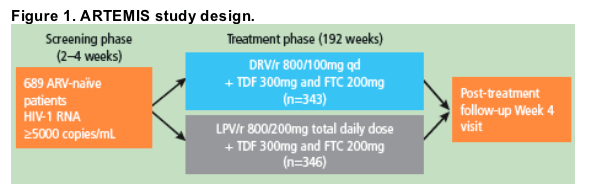
♦ ARTEMIS is an ongoing, randomized, controlled, Phase III trial to compare the efficacy, safety and tolerability, resistance characteristics and pharmacokinetics of DRV/r versus LPV/r in treatment-naïve HIV-1-infected patients.4
♦ Patients were considered treatment-naïve if they had never received treatment with an ARV drug, including both investigational as well as commercially available ARVs indicated for the treatment of HIV-1 infection.
♦ Treatment-naïve, HIV-1-infected patients with HIV-1 RNA 35000 HIV-1 RNA copies/mL were randomized in a 1:1 ratio to DRV/r 800/100mg qd or LPV/r 800/200mg total daily dose combined with a fixed-dose background regimen consisting of tenofovir disoproxil fumarate (TDF) and emtricitabine (FTC; TDF/FTC was provided by Gilead) (Figure 1).
♦ Use of LPV/r qd or bid (total daily dose of 800/200mg) dosing was based on regulatory approval for naïve patients and investigator/patient preference; a switch from capsule to tablet (Meltrex) was made according to local regulatory approval and drug availability.
Virologic analysis
♦ Analyses were performed on the dataset of the 96-week analyses, at which time all patients had reached Week 96 of treatment or discontinued earlier. All VFs in the dataset were included in the analysis, including those occurring prior to or after the Week 96 timepoint.
♦ VFs were defined as 'rebounders' (loss of HIV-1 RNA <50 copies/mL) and 'never suppressed' (never achieved HIV-1 RNA <50 copies/mL).
♦ The TLOVR (non-VF censored) imputation method was used for the identification of VFs, meaning that data were not imputed at timepoints after discontinuation for patients who discontinued for reasons other than VF (non-VF). Moreover, patients who discontinued before Week 12 were not taken into account to determine VF.
♦ Initially, phenotypic (Antivirogram) and genotypic determinations were only performed on plasma samples with HIV-1 RNA 31000 copies/mL. In order to better assess the relationship between VF and resistance in the ARTEMIS study, additional testing was performed on samples from VFs with HIV-1 RNA 350 and <1000 copies/mL. This resulted in more geno-/phenotypic data at endpoint (endpoint was defined as the last on-treatment visit with a geno- and/or phenotype).
♦ Phenotypic resistance was defined as having a fold-change in 50% effective concentration (FC) above the biologic/clinical cut-off (Antivirogram). The clinical cut-off of 10 was used for both DRV5 and LPV;6 a clinical cut-off of three was used for tipranavir (TPV),7 and 1.4 for TDF.8 The biologic cut-offs 2.2, 2.1, 2.3, 2.2, 1.8
and 3.1 were used for amprenavir (APV), atazanavir (ATV), indinavir (IDV), nelfinavir (NFV), saquinavir (SQV), and FTC, respectively.
♦ Lists of PI resistance-associated mutations (RAMs) were based on the IAS-USA list9 (major [also known as primary] PI RAMs and minor PI RAMs). The IAS-USA PI RAMs were further classified as non-polymorphic major [D30N, V32I, M46I/L, I47A/V, G48V, I50L/V, I54A/L/M/S/T/V, L76V, V82A/F/L/S/T, I84V, N88D/S and L90M] and
minor [L10F/I/R/V, V11I, L24I, L33F, E35G, K43T, F53L/Y, Q58E, A71I/T/V, G73A/C/S/T, T74P, N83D and L89V] according to the CASTLE 96-week analyses.10 Polymorphic mutations that were excluded from the IAS-USA list9 were defined as PI RAMs listed as minor in IAS-USA that had mutation scores of zero for all PIs,
according to values assigned by the Standford database. The remaining nonpolymorphic mutations listed in IAS-USA were further categorized as either major or minor using both IAS-USA and Stanford classifications, with the Stanford database taking precedence because of its mutation scoring algorithm. DRV RAMs were based on the 2007 DRV RAMs list.11
♦ The development of resistance at endpoint, compared to baseline, was studied in patients who experienced VF
-- development of a mutation: defined as the detection of a mutation by population sequencing at endpoint, but not at baseline or pre-baseline
-- loss of susceptibility to an ARV: defined as having a FC above the
biological/clinical cut-off at endpoint while not at baseline.
♦ Sample HIV-1 subtype was derived as the best match in-between the determined protease-reverse transcriptase nucleotide sequence (determined by Virco BVBA, Mechelen, Belgium) and the corresponding ones from the Los Alamos National Laboratory subtype reference subset (2005).12
Results
Baseline characteristics
The baseline characteristics of the patients showed a population of ARV-naïve patients, that included a majority (64.3%) with Centers for Disease Control and Prevention (CDC) category A HIV infection and 8.7% with CDC category C disease
-- baseline characteristics were similar between the two treatment groups. The mean baseline HIV-1 RNA was 4.85 log10 copies/mL and the median CD4 cell count was 225 cells/mm3.
The majority of patients (60.8%) harbored HIV-1 subtype B; 13.1% harbored HIV-1 subtype C; 17.0% harbored HIV-1 subtype CRF01_AE, and 9.2% harbored another HIV-1 subtype.
A low level of transmitted (primary) drug resistance was observed in this ARV-naïve population. At screening and/or baseline 1.6% harbored at least one PI resistant mutation and 3.6% harbored at least one NRTI resistant mutation according to the list of transmitted drug resistant mutations proposed by Shafer, et al.13
Virologic failures
The VF rate was lower in the DRV/r group (11.7%) than in the LPV/r group (17.1%); more LPV/r than DRV/r patients rebounded or were never suppressed (Figure 2).
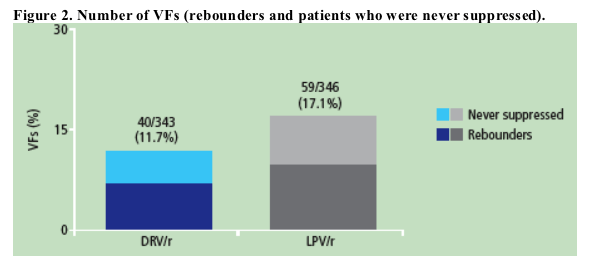
Twelve out of the 24 rebounders (50%) in the DRV/r group and 16 out of 33 rebounders (48%) in the LPV/r group had transient HIV-1 RNA elevations (confirmed HIV-1 RNA >50 copies/mL), but subsequently returned to undetectable at the final viral load measurement without any changes to their study regimen.
The subtype distribution of both the DRV/r and LPV/r VFs was similar to the subtype distribution of the overall population: 57.5% DRV/r and 57.6% LPV/r VFs harbored HIV-1 subtype B; 20.0% DRV/r and 16.9% LPV/r VFs harbored HIV-1 subtype C; 12.5% DRV/r and 15.3% LPV/r VFs harbored HIV-1 subtype CRF01_AE, and 10.0% DRV/r and 10.2% LPV/r VFs harbored another HIV-1 subtype (see baseline characteristics).
Transmitted drug resistance was rarely observed among VFs. At baseline and/or screening, only one DRV/r VF harbored PI-resistant mutations (I84V and L90M), and four DRV/r VFs and one LPV/r VF harbored NRTI resistant mutations (M41L, D67G, L210W, and T215D/S/Y and T69D, respectively) according to the list of transmitted drug resistant mutations proposed by Shafer, et al.13
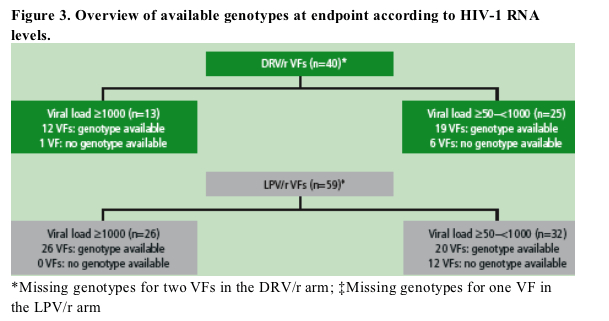
Figure 3 presents an overview of available endpoint genotypes obtained from all VFs according to the HIV-1 RNA levels. In the DRV/r group 31 out of 40 VFs had paired baseline/endpoint genotypes and 46 out of 59 VFs had paired baseline/endpoint genotypes in the LPV/r group.
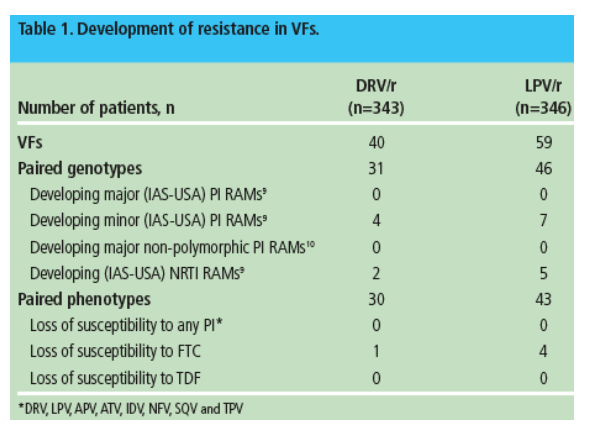
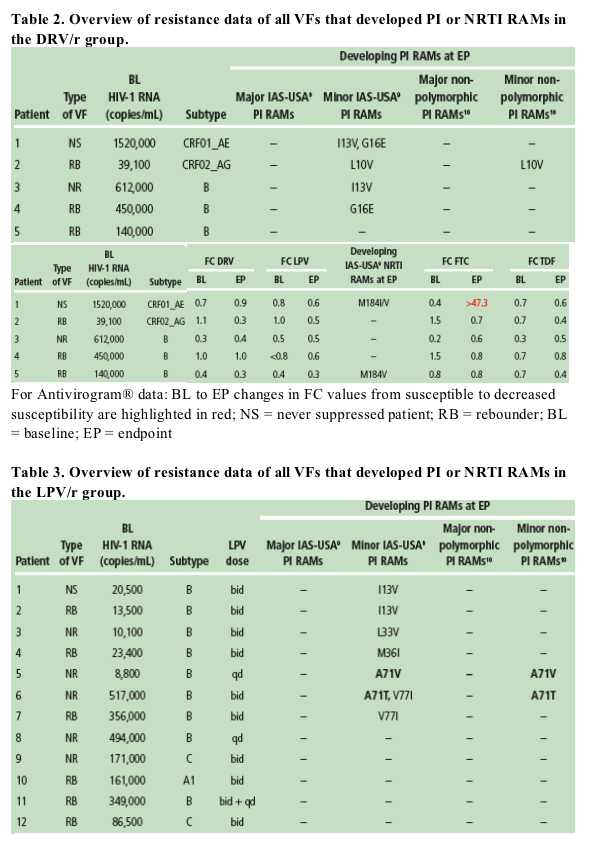
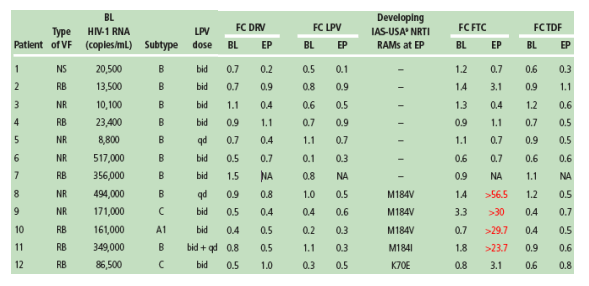
Among all the VFs with an available genotype at baseline and endpoint (31 for DRV/r and 46 for LPV/r), no major (or primary) IAS-USA PI RAMs developed (Tables 1-3)
-- in the DRV/r arm, four VFs developed one or two IAS-USA minor PI RAMs9 at endpoint as compared with baseline and pre-baseline: L10V, I13V, G16E, and I13V+G16E. After exclusion of polymorphic mutations, only one VF developed one minor non-polymorphic PI RAM10 (Tables 1 and 2). None of the developing mutations correspond to DRV RAMs11
-- in the LPV/r group, seven VFs developed one or two IAS-USA minor PI RAMs9 at endpoint as compared to baseline and pre-baseline: I13V (n=2), L33V, M36I, A71V, V77I, and A71T+V77I. After exclusion of polymorphic mutations, two VFs developed one minor non-polymorphic PI RAM10 (Tables 1 and 3). Of the mutations that developed only A71T/V are described as LPV RAMs.9
Based on phenotype (Antivirogram), all HIV isolates from VFs with available phenotypes at baseline and endpoint (30 for DRV/r and 43 for LPV/r) remained susceptible at endpoint to all PIs including the regimen PI (Table 1).
Two VFs in the DRV/r group developed a NRTI RAM:M184V or M184I/V (FTC RAMs). In one patient the development of M184I/V was associated with decreased susceptibility to FTC included in the background regimen (Tables 1 and 2).
Five VFs in the LPV/r group developed a NRTI RAM: K70E (TDF RAM), M184I and M184V (n=3). Development of the FTC RAMs M184I and M184V mutation was associated with decreased susceptibility to FTC included in the background regimen (Tables 1 and 3).
No VFs developed the K65R mutation and no phenotypic resistance to TDF was noted.
|
| |
|
 |
 |
|
|