 |
 |
 |
| |
Factors affecting virologic response to darunavir/ritonavir and lopinavir/ritonavir in treatment-naïve HIV-1-infected patients in ARTEMIS at 96 weeks
|
| |
| |
Reported by Jules Levin
CROI 2009 Feb 8-12 2009, Montreal
Mark Nelson,1 Patrick Yeni,2 Michael Sension,3 Prudence Ive,4 Liddy Chen,5 Andrew Hill,6 Ralph DeMasi,5 Sabrina Spinosa-Guzman7
1Chelsea and Westminster Hospital, London, UK; 2Hopital Bichat and University of Paris, Paris, France; 3Comprehensive Care Center, Fort Lauderdale, Florida, USA; 4Clinical HIV Research Unit, Department of Medicine,
University of the Witwatersrand, Johannesburg, South Africa; 5Tibotec Inc., Yardley, PA, USA; 6University of Liverpool, Liverpool, UK; 7Tibotec BVBA, Mechelen, Belgium
AUTHOR CONCLUSIONS
In ARTEMIS at 96 weeks, significantly more treatment-naïve patients achieved HIV-1 RNA <50 copies/mL with once-daily DRV/r 800/100mg compared with LPV/r 800/200mg total daily dose, even after adjusting for baseline predictors of response (i.e. adherence, age, race and baseline HIV-1 RNA).
These results were also seen when patients who discontinued for adverse events or other reasons were excluded from the analysis (non-VF censored population).
Sub-optimal adherence to DRV/r did not compromise virologic response, whereas patients receiving LPV/r who had sub-optimal adherence were significantly more likely to fail therapy.
The efficacy benefit of DRV/r over LPV/r in the ARTEMIS trial was driven primarily by virologic endpoints. This efficacy benefit was not primarily caused by differences in discontinuations for adverse events or other reasons.
Introduction
ARTEMIS (TMC114-C211; AntiRetroviral Therapy with TMC114 ExaMined In naïve Subjects) is an ongoing, randomized, controlled, Phase III trial evaluating the efficacy and safety of darunavir (DRV;TMC114) with low-dose ritonavir (DRV/r) versus lopinavir (LPV)/r in treatment-naïve HIV-1-infected patients.1
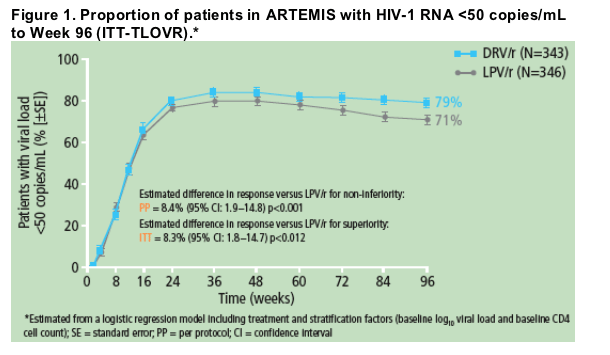
In the 96-week analysis,2 79% of DRV/r compared with 71% of LPV/r patients achieved HIV-1 RNA <50 copies/mL; intent-to-treat/time-to-loss of virologic response (ITT-TLOVR), p value for superiority = 0.012.
Once-daily DRV/r was generally safe and well tolerated in the Week 96 analysis2
-- grade 2-4 diarrhea at least possibly related to treatment occurred less frequently with DRV/r than LPV/r (4% vs 11%; p<0.001)
-- grade 2-4 triglyceride and total cholesterol laboratory abnormalities were reported less frequently with DRV/r than LPV/r (18% vs 28%, p=0.0016 and 4% vs 13%, p<0.0001, respectively).
DRV/r at a dose of 800/100mg qd has been approved in both Europe and the US3 for the treatment of HIV-1 infection in treatment-naïve adult patients.
To determine the factors influencing virologic response to DRV/r in ARTEMIS, we examined the effect of different baseline and treatment factors (such as adherence) on HIV-1 RNA reduction to <50 copies/mL at Week 96 in different subgroups of patients in the trial.
Methods
Patients and study design
♦ Adult, HIV-1-infected, treatment-naïve patients with HIV-1 RNA 35000 copies/mL were randomized to receive DRV/r 800/100mg qd or LPV/r 800/200mg total daily dose
-- all patients receive a fixed-dose background regimen of tenofovir disoproxil fumarate (TDF) 300mg qd and emtricitabine (FTC) 200mg qd (TDF/FTC was provided by Gilead).
♦ The primary objective of the ARTEMIS study was to demonstrate non-inferiority of DRV/r qd versus LPV/r based on the primary endpoint, which was the proportion of patients with confirmed HIV-1 RNA <50 copies/mL.
♦ Detailed methodology of the ARTEMIS study has been reported previously.1
♦ For comparing proportions, unless otherwise stated (eg. using a model with certain covariates), chi-squared tests were used.
Definition of virologic response
ITT-TLOVR was used to define virologic response <50 copies/mL at Week 96
♦ In the TLOVR algorithm, a patient's response is considered to be >50 copies/mL at Week 96 if he/she
-- discontinued randomized treatment before Week 96, for any reason
-- had not achieved HIV-1 RNA levels below 50 copies/mL for at least two consecutive visits before Week 96 (never suppressed)
-- showed a rebound in HIV-1 RNA above 50 copies/mL for two consecutive visits by Week 96, after intitial suppression. Even if this rebound in HIV-1 RNA was temporary, this patient is still a failure by TLOVR.
Confirmed virologic response (CVR) NC=F
♦ CVR is the same as the standard ITT-TLOVR analysis, but does not include any temporary blips in HIV-1 RNA as failures. This method was used in the CASTLE study.4 If a patient shows two consecutive HIV-1 RNA >50 copies/mL values during treatment, but then there is
resuppression to <50 copies/mL on two consecutive visits, the patient is still classified as a success by this method.
Non-VF censored
♦ This analysis excludes patients who discontinued randomized treatment for any reason other than VF.
Multivariate analysis models
♦ Logistic regression models were implemented to investigate the associations between achieving HIV-RNA <50 copies/mL at Week 96 and treatment and prognostic covariates.
♦ Potential prognostic covariates included age, sex, race, region, adherence, baseline log10 HIV-1 RNA and baseline CD4 cell count.
♦ Treatment effect was measured by differences in the unadjusted and model-adjusted responses using TLOVR and TLOVR non-VF censored algorithms (excluding discontinuations for reasons other than VF to determine response).
Adherence
♦ The Modified Medication Adherence Self-Report Inventory (M-MASRI) questionnaire assessed adherence with trial medication by percentages of doses taken from Week 0 to Week 96
-- average adherence from Week 4 to Week 96 was used to assess overall adherence up to Week 96 or time of withdrawal in early terminations (mean adherence >95% [adherent] vs 295% [sub-optimally adherent]).
Results
Patient disposition and baseline characteristics
Demographic data and disease characteristics were well balanced across the treatment arms at baseline (Table 1).
Overall response and response by adherence
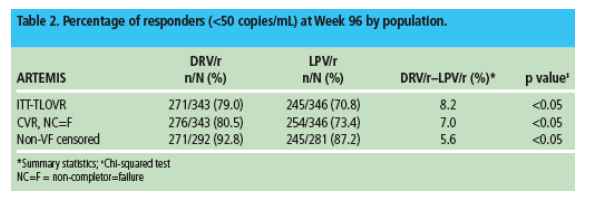
The overall response rate and the percentage of responders are shown in Figure 1 and Table 2, respectively.
When response was assessed by adherence, there was no statistically significant difference in response (HIV-1 RNA <50 copies/mL) at Week 96 between adherent patients in the DRV/r and LPV/r treatment groups.
However, in sub-optimally adherent patients, those receiving DRV/r had a greater response at Week 96 than those receiving LPV/r (Figure 2).
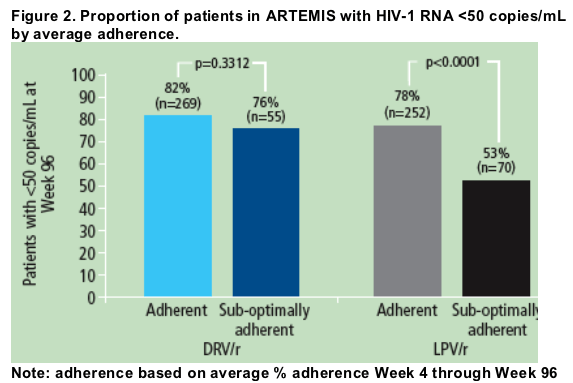
In the DRV/r group, sub-optimally adherent patients had similar rates of response (76%) compared with adherent patients (82%; p=0.3312).
In the LPV/r group, sub-optimally adherent patients had statistically lower rates of response (53%) than adherent patients (78%; p<0.0001).
Multivariate analysis
In the multivariate analyses, the difference in response (HIV-1 RNA <50 copies/mL) favoring DRV/r was maintained after adjusting for baseline and treatment factors (Figure 3).
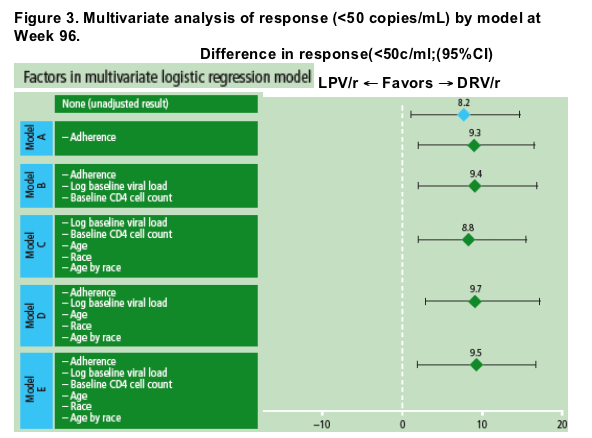
In the multivariate analysis, the full model (N=643) included treatment, adherence, age, race, baseline log10 HIV-1 RNA, and baseline CD4 cell count
-- region was initially included in the model, but was found to be significantly correlated with race, and therefore was removed.
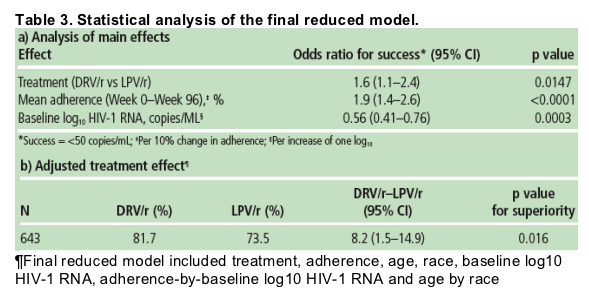
In the final reduced model, treatment effect was also examined; the analysis of main effects is shown (Table 3a). The response and difference in response was calculated for the final reduced model (Table 3b)
-- significantly more treatment-naïve patients achieved HIV-1 RNA <50 copies/mL with once-daily DRV/r compared with LPV/r.
Repeat analysis in the non-VF censored population
Multivariate analysis
The analysis of main effects was performed in non-VF censored patients (excludes all patients who discontinued for reasons other than true VF), and the reduced model results are shown (Table 4a)
-- significantly more treatment-naïve patients achieved HIV-1 RNA <50 copies/mL with once-daily DRV/r compared with LPV/r (Table 4b).

REFERENCE
1. Ortiz R, et al. AIDS 2008;22:1389-97.
2. Mills A, et al. 48th ICAAC/46th IDSA, Washington, DC, USA, 25-28 October 2008. Abstract H-1250c.
3. Tibotec Inc. PREZISTATM (darunavir) Prescribing Information. Revised October 2008 [accessed 22 December 2008]. Available from: http://www.prezista.com.
4. Molina J-M, et al. 48th Annual ICAAC/IDSA 46th Annual Meeting, Washington, DC, USA, 25-28 October 2008. Abstract H-1250d.
|
| |
|
 |
 |
|
|