 |
 |
 |
| |
Telbivudine Has Activity against HIV
|
| |
| |
Reported by Jules Levin
CROI 2009 Feb 8-12 Montreal
Emma Low*, A Cox, M Atkins, and M Nelson
Chelsea and Westminster Hosp, London, UK
"LdT may have activity against HIV.
Until further studies have been completed, LdT should not be used as a treatment for hepatitis B in HIV positive individuals who do not require HIV therapy."
Background: Telbivudine (LdT) is an L-nucleoside analogue of thymidine with activity against hepatitis B virus (HBV) DNA polymerase. Despite no studies having investigated the activity and safety of LdT in HIV/HBV co-infection, recommendations from the HIV/HBV International Panel state that LdT has no activity against HIV and it is suggested as a treatment option for HBV infection, when HIV does not require therapy.
Methods: A 45-year-old gentleman with HIV-1/HBV co-infection was noted in February 2006 to have reverted from HBeAg- and anti-HBV+ to HBeAg+ and anti-HBV- with a serum HBV DNA rise from 8,820 copies/mL to 33,600,000 copies/mL. At the time he was ARV and HBV therapy naïve. His HIV disease was stable with a CD4 lymphocyte count of 613 cells/mm3 and an HIV viral load of 14,462 copies/mL.
Reults: In January of 2008, with a serum HBV DNA of 662,000,000 copies/mL, he was offered treatment with either triple therapy for both his HIV and HBV (tenofovir, lamivudine, and efavirenz) or dual therapy for his HBV alone with adefovir (ADV) and LdT. It was decided to opt for ADV and LdT. On review 2 months later he had experienced a fall in his serum HBV DNA to 2782 copies/mL and had suppressed his HIV viral load to < 50 copies/mL. This was confirmed on repeat bloods 4 weeks later. LdT was discontinued 5 months after it was commenced. At the time of discontinuation of LdT his HIV viral load had increased to 127 copies/mL and then rebounded to 3903 copies/mL 1 month later. Three months after discontinuation, the patient agreed to be re-challenged with LdT for a 2-week duration. On the day of restarting LdT his HIV viral load was 1074 copies/mL, 1 week later 177 copies/mL, and 2 weeks later 71 copies/mL (using Roche VL detection assay the last 3 readings were 429, 227, and <47 copies/mL, respectively). The patient has remained on ADV since commencing antiviral therapy in February.
Conclusions: In our opinion the most likely explanation is that LdT has activity against HIV-1. We do not believe that ADV had an effect on the patient's HIV viral load because it has been shown that low dose ADV does not have any effect on HIV viral load. Given that re-challenge with LdT for 2 weeks resulted in suppression of the HIV viral load to 71 copies/mL, we believe that this is strong evidence that LdT is active against HIV-1. Until further trials have been done it should no longer be recommended as a treatment for chronic hepatitis B in HIV+ individuals who do not require treatment for their HIV.
Background
Telbivudine (LdT) is a L-nucleoside analogue of thymidine with activity against hepatitis B virus (HBV) DNA polymerase. Approval by the Food and Drug Administration (FDA) in October 2006 followed the 007 GLOBE trial [1].This phase III trial was designed to compare LdT vs lamivudine (LAM) in individuals with chronic hepatitis B. Despite no studies having investigated the activity and safety of LdT in HIV/HBV co- infection, recommendations from the HIV-HBV International Panel state that LdT has no activity against HIV and it is suggested as a treatment option for HBV infection, when HIV does not require therapy [2]. We report the case of an individual with HIV/HBV co-infection in whom LdT therapy suppressed the HIV viral load (VL) to < 50 copies/ml.
Case Report
A 45 year old individual with HIV-1/HBV co-infection was noted in February 2006 to have reverted hepatitis status from HBeAg-negative and anti-HBV- positive to HBeAg-positive and anti-HBV-negative with a corresponding serum HBV DNA rise from 8,820 copies/ml to 33,600,000 copies/ml. At the time the patient was antiretroviral and HBV therapy naïve. His HIV disease was stable with a CD4 lymphocyte count of 613 cells/mm3 (36.1%) and a HIV VL of 14,462 copies/ml.
In January of 2008 following a Fibroscan demonstrating significant liver stiffness of 12.8 (IQR 0.9) kPa and a serum HBV DNA of 662,000,000 copies/ml, he was offered treatment with either triple therapy for both his HIV and HBV (tenofovir, LAM/emtricitabine and efavirenz) or dual therapy for his HBV alone (Adefovir [ADV] and LdT). With a CD4 lymphocyte count of 640 cells/mm3 (36.3%) and HIV VL of 8,650 copies/ml it was decided to opt for ADV 10mg od and LdT 600mg od. Dual agent therapy was commenced on 26th February 2008. On review two months later, the individual had experienced a fall in serum HBV DNA to 2,782 copies/ml and had suppressed HIV VL to < 50 copies/ml. This was confirmed on repeat bloods four weeks later. LdT was discontinued five months after commencement, while ADV was continued. At the time of discontinuation of LdT, the HIV VL had increased to 127 copies/ml, rebounding to 3,903 copies/ml one month later. Three months following discontinuation, the patient agreed to be re-challenged with LdT for a two week duration. On the day of restarting LdT his HIV VL was 1074 copies/ml, one week later 177 copies/ml and two weeks later 71 copies/ml. The viral loads were further analysed by Roche VL detection assay with corresponding readings of 429, 227 and <47 copies/ml respectively (Figure 1). The patient had remained on ADV since commencing antiviral therapy in February.
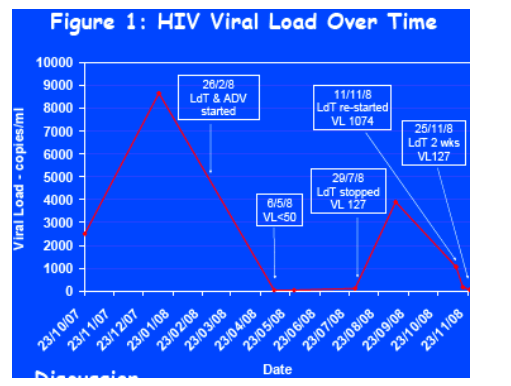
Discussion
In our opinion the most likely explanation for the suppression of HIV VL is LdT activity against HIV-1. It is unlikely that ADV had an effect on the patient's HIV VL as it has been shown that low dose ADV does not have anti-HIV activity [3]. However while on LdT and ADV the patient's HIV VL suppressed to < 50 copies/ml, whereas one month later, while still on ADV monotherapy, it rebounded to 3,903 copies/ml suggesting removal of antiviral pressure. In an attempt to determine whether this was a true effect, the patient underwent re-challenge with LdT. The resultant suppression of the HIV VL to 71 copies/ml represents strong evidence that LdT is active against HIV-1. So far HIV-1 resistance tests have shown no significant mutations.
This is a familiar story. On April 30, 2007 the Department of Health and Human Services (DHHS) Panel on Antiretroviral Guidelines for Adults and Adolescents released a supplement to the Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents concerning the use of entecavir (ETV) in HIV/HBV co-infection. Previously, the guidelines had recommended ETV as an option for patients who required treatment for HBV but not HIV infection. The recommendation had been based on in vitro data showing no significant activity of ETV against HIV- 1[4]. The revision was prompted by a case series of three patients all of whom received ETV without concomitant antiretroviral therapy and experienced a 1log10 decline in HIV-RNA levels [5]. One patient was shown to develop a M184V mutation, indicating that ETV may be active against HIV as well as HBV and if used as monotherapy could promote the development of HIV drug-resistance.
Conclusion
LdT may have activity against HIV.
Until further studies have been completed, LdT should not be used as a treatment for hepatitis B in HIV positive individuals who do not require HIV therapy.
References
[1] Lai CL, Gane E, Liaw YF, Hsu CW, Thongsawat S, Wang Y et al. Telbivudine versus Lamivudine in Patients with Chronic Hepatitis B. The N Engl J Med 2007;357:2576-88.
[2] Soriano V, Puoti M, Peters M, Benhamou Y, Sulkowski M, Zoulim F et al. Care of HIV patients with chronic hepatitis B: update recommendations from the HIV-Hepatitis B Virus International Panel. AIDS 2008;22:1399-1410.
[3] Benhamou Y, Bochet M, Thibault V, Calvez V, Fievet MH, Vig P et al. Safetly and efficacy of adefovir dipivoxil in patients co-infected with HIV-1 and lamivudine-resistant hepatitis B virus: an open-label pilot study. Lancet 2001;358:718-723
[4] BARACLUDE (Product Labelling, Bristol-Meyers Squibb). March 2005.
[5] McMahon M, Jilek B, Brennan T, Shen L, Zhou Y, Bhat S et al. The anti-hepatitis B drug entecavir inhibits HIV-1 replication and selects HIV-1 variants resistant to antiretroviral drugs. 14th Conference on Retroviruses and Opportunistic Infections; Feb 25-28, 2007; Los Angeles, CA. Abstract 136LB.
|
| |
|
 |
 |
|
|