 |
 |
 |
| |
Safety and pharmacokinetics of etravirine in pregnant HIV-infected women
|
| |
| |
Reported by Jules Levin
EACS Nov 3 2009, Cologne Germany
Patricia Izurieta,1 Thomas N Kakuda,2 Andrew Clark,3 Caroline Feys,1 James Witek2
1Tibotec BVBA, Mechelen, Belgium; 2Tibotec Inc., Yardley, PA, USA; 3Johnson
AUTHOR CONCLUSIONS
ETR PK parameters in five pregnant women were comparable with those of non-pregnant adults, suggesting no ETR dose adjustment is needed during the third trimester
Although clinical data on exposure to ETR during pregnancy is limited, ETR did not have an effect on foetal or neonatal toxicity in this case series
Further evaluation of ETR PK in pregnant women is ongoing (trial NCT00855335)
- this study will investigate the PK parameters of ETR and/or DRV/r during the second and third trimesters of pregnancy and up to 12 weeks post-partum
- changes in ARV activity, safety and tolerability during pregnancy and post-partum will be examined
ABSTRACT
Objectives
Etravirine (ETR; TMC125) is a next-generation NNRTI with demonstrated activity in treatment-experienced, HIV-1-infected adults. As pregnancy data with ETR is limited, an assessment of available pharmacokinetic (PK) and safety data in pregnant women was undertaken.
Methods
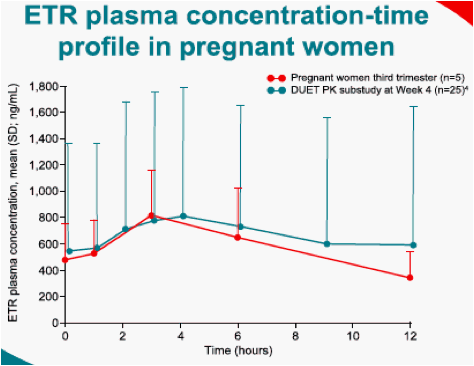
ETR was available via compassionate use to pregnant women in need; PK assessments during the third trimester were requested. Blood samples were collected predose, 1, 3, 6 and 12 hours post-dose. Plasma ETR concentrations were determined using a validated high performance liquid chromatography-mass spectrometry/mass spectrometry (LC-MS/MS) assay. PK parameters were obtained by non-compartmental analysis and compared to historical control. Women were followed until delivery and whenever possible, cord blood samples were obtained.
Results
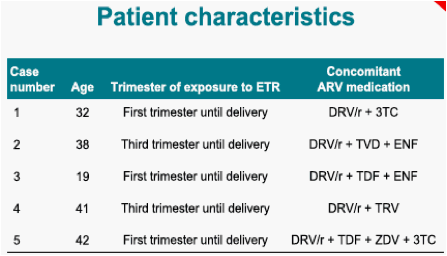
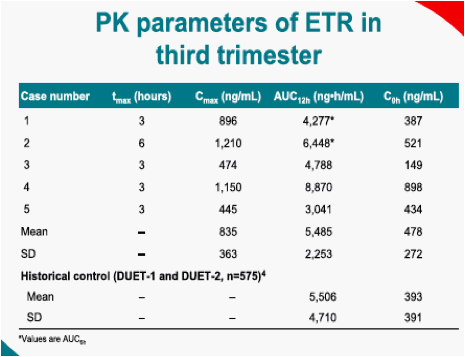
Five women participated in the PK evaluation. Three women were exposed to ETR throughout their pregnancy and two during the third trimester only. Individual PK parameters are as follows: & Johnson, High Wycombe, UK
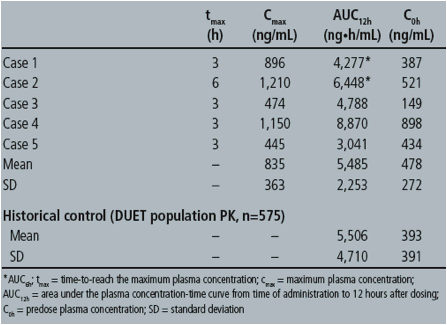
Among the five pregnancies, three Caesarean sections were performed, one pre-term due to twin pregnancy, the remaining two were normal deliveries. All babies were healthy, one baby was born with an accessory auricle, but otherwise normal. There were no other malformations or other abnormal findings. Post-partum ETR cord blood concentration in Case 5 was 112ng/mL, whereas the corresponding plasma concentration was 339ng/mL. Three mothers had undetectable HIV-RNA at delivery; no data was available for the remaining two mothers. Two babies were HIV-DNA negative (two twins) and one baby had undetectable HIV-RNA at delivery, no data is available for the remaining three babies.
Conclusions
ETR pharmacokinetics in five pregnant women were comparable to those of non-pregnant adults suggesting no ETR dose adjustment is needed during the third trimester. Although data on exposure to ETR during pregnancy is limited, this data suggests that ETR may not have an effect on foetal or neonatal toxicity. Further evaluation of ETR pharmacokinetics in pregnant women is ongoing (TMC114-HIV3015: clinicaltrials.gov NCT00855335).





REFERENCES
1. Scholler-Gyure M, et al. Clin Pharmacokinet 2009;48:561-74.
2. Mills A, et al. IAS 2009. Abstract MOPEB036.
3. Campbell T, et al. IAS 2009. Abstract MOPEB038.
4. Kakuda TN, et al. IAC 2008. Abstract TUPE0082
|
| |
|
 |
 |
|
|