 |
 |
 |
| |
"Deep" Sequencing to Identify Treatment-Experienced Patients Who Respond to Maraviroc (MVC)
|
| |
| |
Reported by Jules Levin
EACS Nov 13 2009 Cologne Germany
Luke C. Swenson1, Winnie Dong1, Theresa Mo1, Conan Woods1, Xiaoyin Zhong1, Alexander Thielen2, Mark Jensen3, Jayvant Heera4, Pinaki Biswas6, Suzanne Ellery4, Marilyn Lewis5, Ian James5, Doug Chapman6, Hernan Valdez6 and Richard Harrigan1
1BC Centre for Excellence in HIV/AIDS, Vancouver, Canada; 2Max Planck Institute for Bioinformatics, Saarbrucken, Germany; 3Fortinbras Consulting, Buford, GA, USA; 4Pfizer Global Research and Development, New London, CT, USA, 5Pfizer Global Research and Development, Sandwich UK, 6Pfizer, Inc., New York, NY, USA
AUTHOR CONCLUSION
We examined and optimized the performance of a genotypic tropism assay based on next generation "deep" sequencing of the HIV env V3 loop at predicting outcomes after starting maraviroc-based regimens
This large study establishes "deep" sequence analysis of the HIV V3 as an extremely promising tool for discriminating between treatment-experienced individuals who do and do not respond to maraviroc. Approximately twice as many individuals were detected with X4 virus as by the original Trofile assay, with similarly suboptimal response to therapy
BACKGROUND
The MOTIVATE 1 & 2 studies compared maraviroc (MVC) + optimized background therapy (OBT) vs placebo + OBT in therapy-experienced patients with R5 HIV, with tropism determined using the original Trofile assay (Monogram Biosciences). Patients with non-R5 HIV received MVC in the A4001029 safety study
As per standard of care, tropism testing is required prior to treatment with CCR5 antagonists such as MVC. Currently, the recombinant phenotypic Trofile assay is the most commonly used method to determine HIV tropism/coreceptor usage. Screening in MOTIVATE was performed with the original Trofile assay, which has since been replaced by the enhanced sensitivity Trofile assay.
Genotypic testing using either standard, population-based sequencing or novel 'deep' 454 sequencing of the V3 loop of HIV env may offer alternative approaches to determine tropism
Population-based sequencing has recently been shown to be comparable to the original Trofile assay in predicting clinical response to MVC in the MOTIVATE trials (IAS 2009, Cape Town, Abstract WELBA101)
Here we compare "deep" sequence analysis using a 454/Roche GS-FLX instrument with original Trofile results on a large number of screening samples from MOTIVATE and A4001029 studies of MVC in treatment-experienced individuals
A variety of potential cut-off values for declaring a viral variant "X4" were examined using the geno2pheno (co-receptor) algorithm, as well as the proportion of X4 variants present within a sample to assign a "non-R5" call
Outcomes examined included viral load change (Fig 3), percent of patients with a plasma viral load <50 HIV RNA copies/mL (Fig 4), time to tropism change from R5 using Trofile over 48 weeks (Fig 5), pVL change with different active treatment backgrounds (Fig 6), as well as CD4 cell count change and time to therapy discontinuation (not shown). Optimized values were established at a 3.5% false positive rate (FPR) in the g2P algorithm at a prevalence of 2% of the viral population
Outcomes are indicated for each endpoint for the training dataset of the combined maraviroc arms (75%; panel a), the validation set (25%; panel b), all patients receiving MVC (panel c), a bootstrapped analysis of the MVC arm (panel d), and the placebo arm (panel e). Results were compared against the original Trofile assay. Trofile DM and X4 results were combined into a single "Trofile X4" category
Results
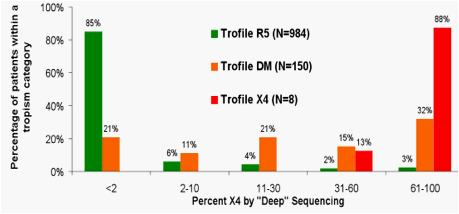
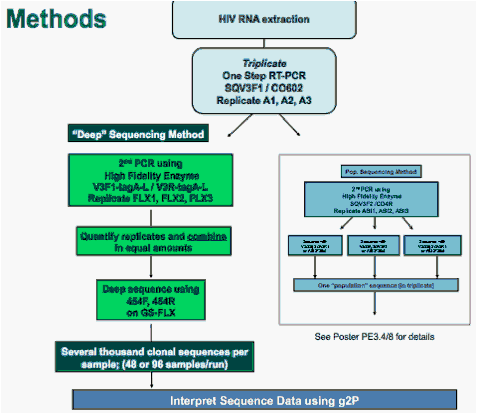
Figure 1. Most patients designated R5 by Trofile had <2% X4 by "deep" sequencing.
The distribution of percent X4 virus was determined by "deep" sequencing and compared with tropism assigned by original Trofile. Using optimized cut-off values (3.5% FPR at 2% prevalence), almost all samples called "X4" by Trofile consisted of >60% X4 virus according to "deep" sequencing, and 85% of Trofile R5 samples had <2% X4 by "deep" sequencing
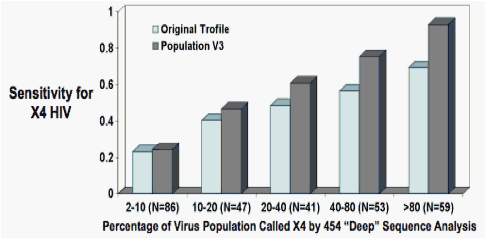
Figure 2. Sensitivity of Original Trofile and Population V3 Sequencing compared to "Deep" Sequence Analysis
Using the 454 method as the "gold standard", original Trofile and population-based V3 sequencing had lower sensitivity for detecting CXCR4-using virus at low %X4 prevalence than at higher %X4
Labeling: R5 (green) or X4 (red) by "deep sequencing", and R5 (black line) or X4 (dotted line) by Trofile. Values are indicated for (left to right) the training set; the validation set; all MVC patients, a bootstrapped analysis of the entire dataset, and all placebo recipients. Cut-off used for the 454 data was 3.5 FPR using g2P, above a prevalence of 2%
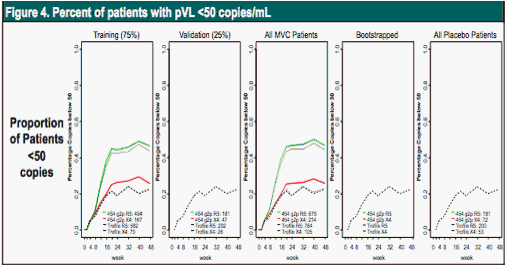
Labeled as above. Patients screened as R5 by 454 sequencing were much more likely to have pVL <50 c/mL at week 48 after MVC than patients screened as X4.

Table 1. Percentage of patients with pVL <50 c/mL at week 48.
More patients screened as R5 by 454 sequencing achieved pVL <50 c/mL at week 48 than those called R5 by Trofile.
Figure 5. Time to change of tropism from R5 to non-R5 after receiving MVC
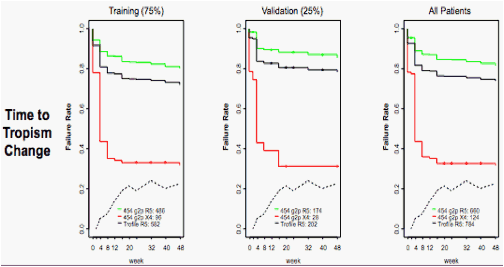
Stratified by 454 status, as above. Samples called R5 by original Trofile but X4 by 454 sequencing were more likely to "switch" tropism over the course of the study period.
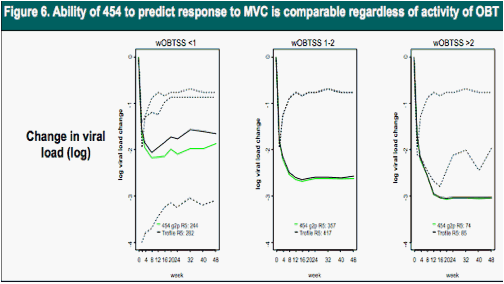
wOBTSS dependent pVL decline in MVC arms in patients with R5 HIV by Trofile or "deep" sequencing. Where the number of active drugs in the background regimen, i.e., the weighted optimized background therapy susceptibility score (wOBTSS) is <1, 1-2 or >2.
|
| |
|
 |
 |
|
|