 |
 |
 |
| |
Absence of a teratogenic potential from a novel
next-generation NNRTI, TMC278
|
| |
| |
Reported by Jules Levin
EACS Nov 13 Cologne Germany
M Desmidt,1 B Willems,1 P Dom,1 G Bailey,1 L De Schaepdrijver,1 L Lammens,1 S Lachau-Durand,2
F van Velsen,2 M Martens,2 W Coussement1
1Johnson & Johnson Pharmaceutical Research and Development, Beerse, Belgium; 2Tibotec BVBA, Mechelen, Belgium
AUTHOR CONCLUSIONS
TMC278 did not show teratogenic potential in rat and rabbit models
at exposures 13- to 80-times higher than those seen in HIV-1-infected
patients receiving 25mg qd TMC278 at steady-state
- AUC0-24h: 2.8µg·h/mL (Phase IIb TMC278-C204 study)
- AUC0- ∞: 37µg·h/mL (NOAEL 40mg/kg/day pregnant rats
and foetuses)
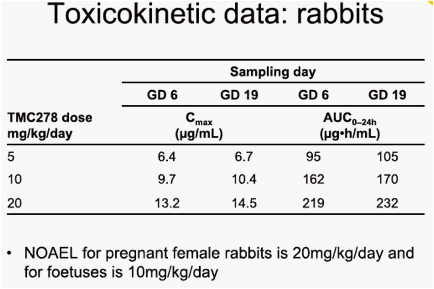
- AUC0-24h: 170-232µg·h/mL (NOAEL 10mg/kg/day rabbit foetuses
and 20mg/kg/day pregnant rabbits).
These animal data suggest that further studies of TMC278 in WOCBP
are warranted.

ABSTRACT
Objective
TMC278 (rilpivirine) is an investigational next-generation NNRTI that has very good anti-HIV-1 activity and a favourable safety/tolerability profile at a convenient once-daily dose of 25mg. The embryo-foetal toxicity of TMC278 was investigated in rats and rabbits.
Methods
TMC278 was administered by oral gavage at doses of up to 400mg/kg/day to pregnant Sprague-Dawley rats and up to 20mg/kg/day in pregnant New Zealand white rabbits during the period of organogenesis (gestation Days 6-17 in rats and Days 6-19 in rabbits).
Results
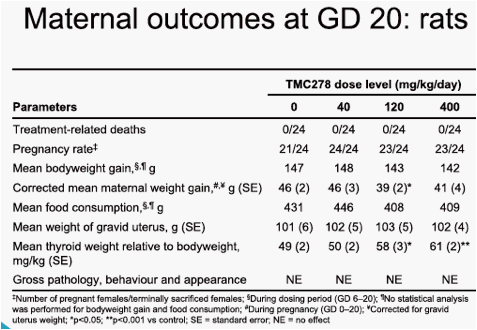
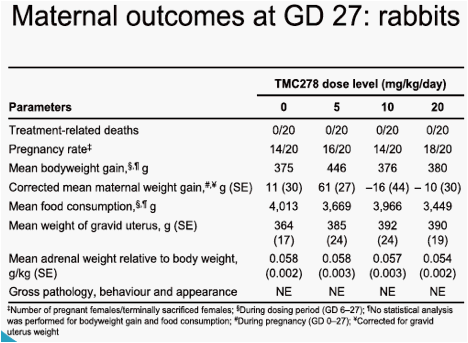
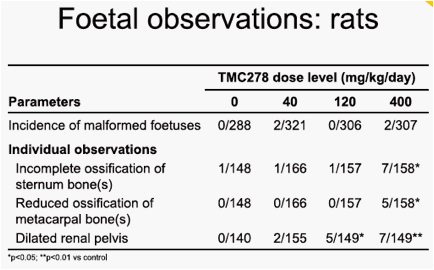
In rats, at the 120mg/kg/day and 400mg/kg/day doses, indications of moderate maternal toxicity were noted (reduced food consumption and bodyweight gain, and increased thyroid weight). In rabbits, there was no maternal toxicity up to 20mg/kg/day. In rats and rabbits, no teratogenic effect was seen. There was no effect of treatment on pregnancy parameters in any group. The only finding on embryo-foetal development in rats was a slight increase in the incidence of a
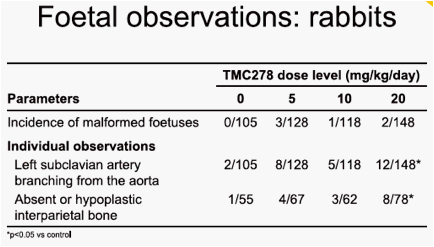
visceral variant, dilated renal pelvis, at the 120mg/kg/day and 400mg/kg/day doses. The maternal and embryo-foetal no observable adverse effect level (NOAEL) was 40mg/kg/day, associated with a maternal area under the plasma concentration-time curve from time of administration to 24 hours after dosing (AUC0-24h) value of 37µg·h/mL. The only finding on embryo-foetal development in rabbits was a slight increase in the incidence of foetuses exhibiting minor variations of the left subclavian artery originating from the aorta and of hypoplastic interparietal bone in the group given 20mg/kg/day. The maternal toxicity and embryo-foetal NOAELs were 20mg/kg/day and 10mg/kg/day, respectively, associated with maternal AUC0-24h values of 232µg·h/mL and 170µg·h/mL. The mean AUC0-24h value in HIV-1-infected patients at 25mg TMC278 qd was 2.8µg·h/mL.
Conclusion
The absence of a teratogenic potential from TMC278 in the rat and rabbit models even at exposures many times higher than those in the clinic is a key element in its safety profile.









Reference
1. Molina J-M, et al. HIV9 2008. Abstract P002.
|
| |
|
 |
 |
|
|