 |
 |
 |
| |
Absorption, metabolism and excretion of TMC278, an NNRTI, after a single oral dose of 150mg in healthy male volunteers
|
| |
| |
Reported by Jules Levin
EACS Nov 13 2009 Cologne Germany
S Lachau-Durand,1 R NVS Mamidi,2 F Cuyckens,2 V Michlova,2 G Mannens,2 A Raoof1
1Tibotec BVBA, Mechelen, Belgium; 2Johnson & Johnson Pharmaceutical Research and Development, Beerse, Belgium

ABSTRACT
Objective
TMC278 (rilpivirine) is a next-generation NNRTI that has demonstrated potent and sustained efficacy through 96 weeks in antiretroviral (ARV)-naïve, HIV-infected patients.1 In the present study, the absorption, metabolism and excretion of TMC278 was evaluated in healthy volunteers.
Methods
A single oral dose of TMC278 (150mg including 1,764kBq of 14C-TMC278) was administered to six healthy male volunteers. Blood and urine were collected until 168 hours and faeces up to 336 hours after administration. Total radioactivity was analysed by liquid scintillation counting, and metabolite profiling in plasma, urine and faeces was performed by radio high-performance liquid chromatography (HPLC) and liquid chromatography-mass spectrometry/mass spectrometry (LC-MS/MS).
Results
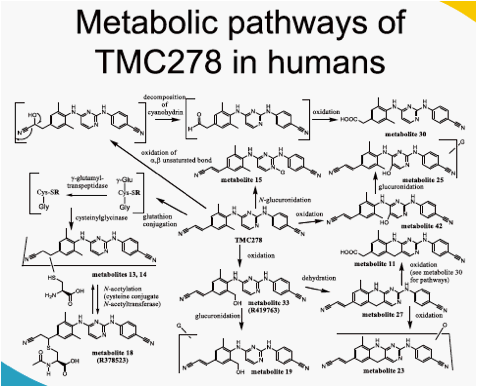
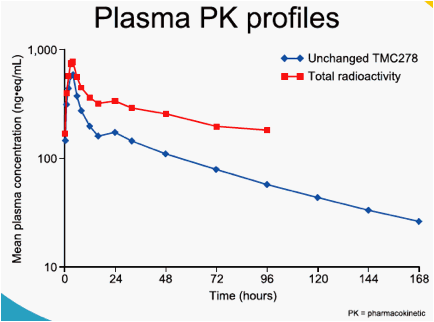
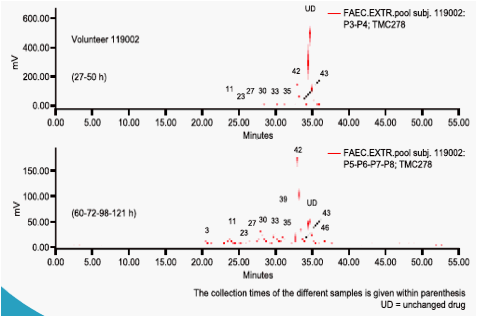
In plasma, unchanged TMC278 represented the major part of the total radioactivity. Minor plasma metabolites included TMC278 N-glucuronide,
the tricyclic metabolite and hydroxymethyl TMC278. The administered
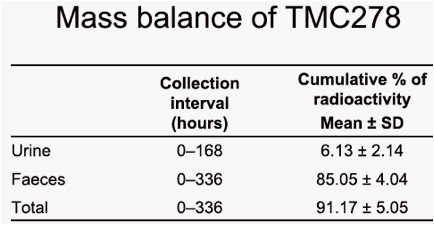
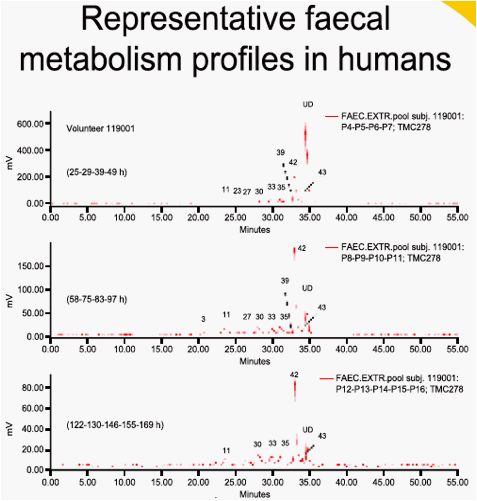
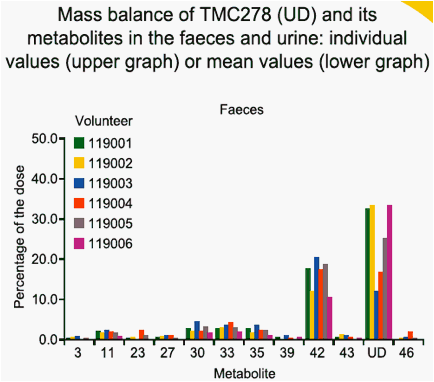
dose was almost completely recovered in faeces (on average 85.1%) of
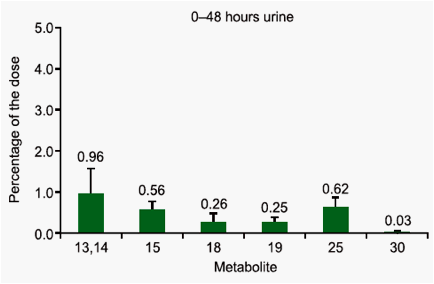
which unchanged TMC278 represented only 25.5%. Major faecal metabolites originated from oxidative pathways, with the most abundant metabolite being derived from oxidation at the 5-position of the pyrimidinyl moiety (16.1%). Three other metabolites (methyl hydroxylation at the dimethylphenyl moiety, a carboxylic acid metabolite on the cyanoethenyl moiety and an unidentified metabolite) each accounted for 2.2-3.0% of the dose. Dehydration of hydroxymethyl TMC278 resulted in a tricyclic metabolite (0.6%), which was further oxidised (0.7%) or transformed to a carboxylic acid metabolite (1.6%). In urine, 6.1% of the dose was recovered and only a small amount of unchanged TMC278 was detected, indicating negligible renal clearance of TMC278. Apart from a carboxylic acid metabolite, Phase II metabolites (glucuronides or glutathione-derived conjugates) were recovered in urine.
Conclusion
TMC278 is eliminated primarily by metabolism and subsequent faecal excretion. No major or unique metabolites were identified in humans.







|
| |
|
 |
 |
|
|