 |
 |
 |
| |
Schering's protease boceprevir phase 2; Schering symposium
|
| |
| |
EASL
Reported by Jules Levin
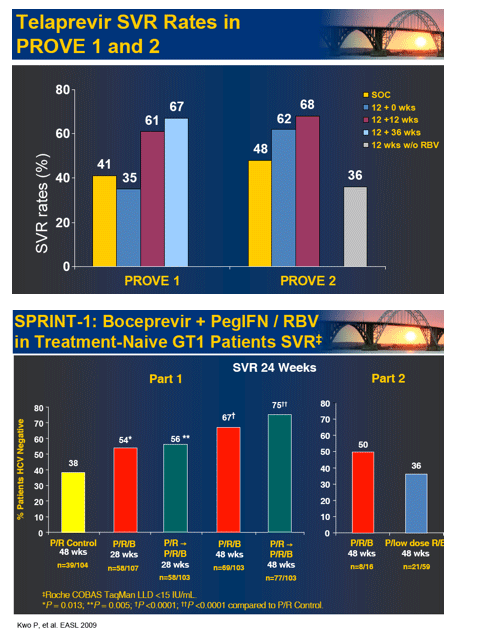
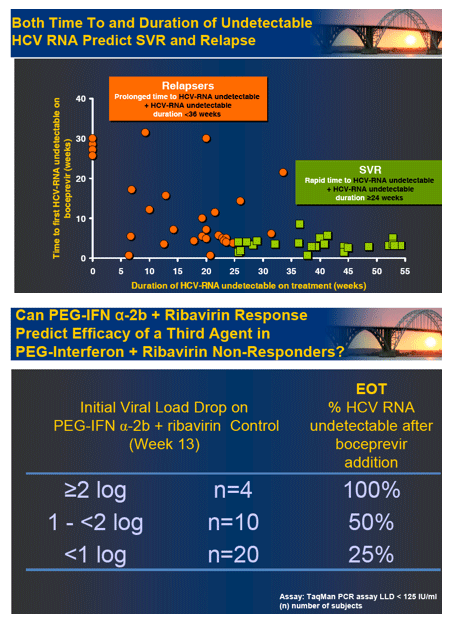
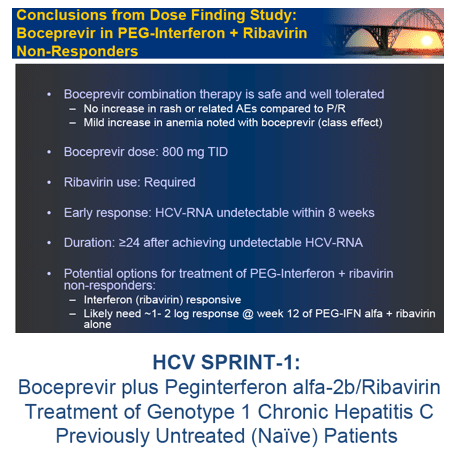
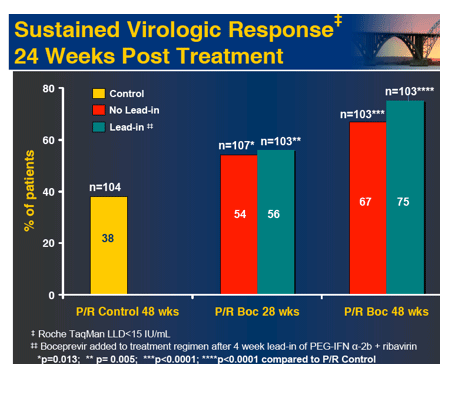
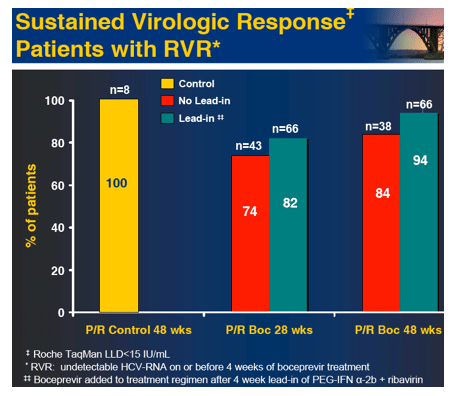
In the early afternoon presentation Schering Plough reported phase II results from the SPRINT Study of their protease inhibitor boceprevir currently in phase III. The data is very interesting. You can see in the table below overall SVR was 55% with 28 weeks of boceprevir+peg/rbv with a 4-week peg/rbv lead-in before starting boceprevir SVr was 56%. Patients with 48 weeks and no lead-in had 67% SVR and patients with 4 week lead-in and 48 weeks therapy had 75% SVR. Of interest they presented patients response who had little response to peg?RBV lead-in. So patients with < .5 log reduction had 44% SVR and patients with .5 to 1 log reduction had 62% SVR. These rates are surprisingly good but of course these patients are susceptible to greater risk for PI resistance. Of note, patients with RVR and lead-in had 82% SVR after 28 weeks and 94% SVR with 48 weeks therapy and lead-in. Without lead-in therapy resulted in 74% and 86% in 28 & 48 weeks, respectively. Still, the most interesting study at this conference will be the INFORM Stud presented saturday which will report the results from combination of 2 orals R7128 the polymerase and ITMN191 the protease being developed by Roche. They are looking at quad therapy combining peg/RBV with these 2 orals. This is the first data of 2 orals in patients. Stay tuned.
HCV SPRINT-1 FINAL RESULTS : SVR 24 FROM A PHASE 2 STUDY OF BOCEPREVIR PLUS PEGINTRON (PEGINTERFERON ALFA-2B)/RIBAVIRIN IN TREATMENT-NAIVE SUBJECTS WITH GENOTYPE-1 CHRONIC HEPATITIS C
P. Kwo1, E. Lawitz2, J. McCone3, E. Schiff4, J. Vierling5, D. Pound6, M. Davis7, J. Galati8, S. Gordon9, N. Ravendhran10, L. Rossaro11, F. Anderson12, I. Jacobson13, R. Rubin14, K. Koury15, C. Brass15, E. Chaudhri15, J. Albrecht15
1Department of Medicine, Indiana University School of Medicine, Indianapolis, IN, 2Alamo Medical Research, San Antonio, TX, 3Mount Vernon Endoscopy Center, Alexandria, VA, 4Center for Liver Diseases, University of Miami Miller School of Medicine, Miami, FL, 5Professor of Medicine and Surgery, Baylor College of Medicine, Houston, TX,6Indianapolis Gastroenterology Research Foundation, Indianapolis, IN, 7Digestive Care/South Florida Center of Gastroenterology, Wellington, FL, 8Liver Specialists of Texas, Houston, TX, 9Department of Hepatology, Henry Ford Health Systems, Detroit, MI, 10Digestive Disease Associates, Baltimore, MD, 11Internal Medicine, University of California-Davis Medical Center, Sacramento, CA, USA, 12The Liver & Intestinal Research Center, Vancouver, BC, Canada, 13Weill Cornell Medical College, New York, NY, 14Digestive Healthcare of Georgia, Atlanta, GA, 15Schering-Plough Research Institute, Kenilworth, NJ, USA
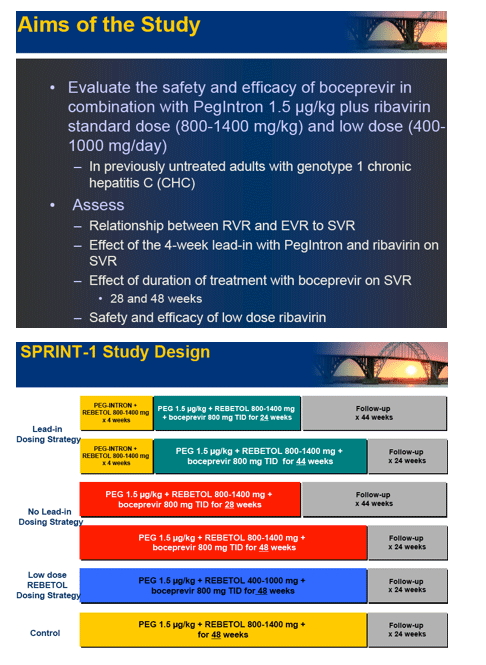
Background: HCV SPRINT-1 assessed the safety and efficacy of boceprevir, an oral inhibitor of HCV-NS3 protease, plus PegIntron (P) (1.5 µg/kg/QW) and ribavirin (R) (400-1400 mg/day).
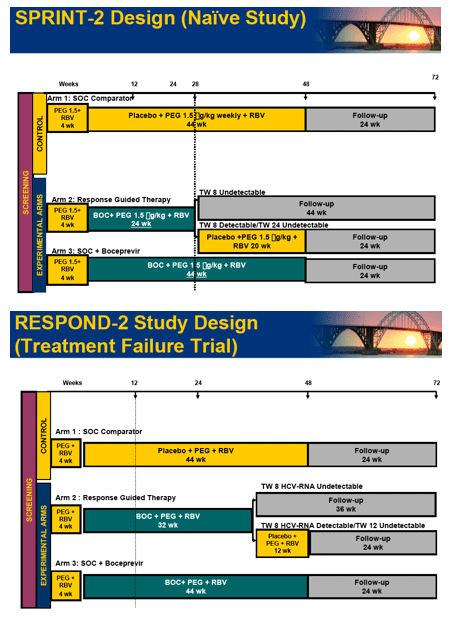
Methods: P/R(800-1400 mg/day) for 48 weeks compared to five boceprevir(800 mg TID) regimens: 4 weeks of P/R lead-in followed by P/R(800-1400 mg/day)/boceprevir for 24 or 44 weeks; P/R(800-1400 mg/day)/boceprevir for 28 or 48 weeks; and P/low-dose R(400-1000 mg/day)/boceprevir for 48 weeks. The primary endpoint was sustained virologic response (SVR) at 24 weeks post-treatment (Roche TaqMan LLD=15 IU/mL).
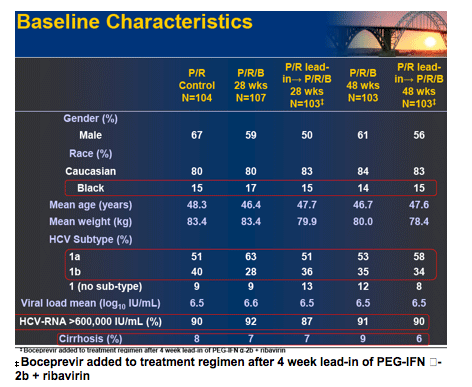
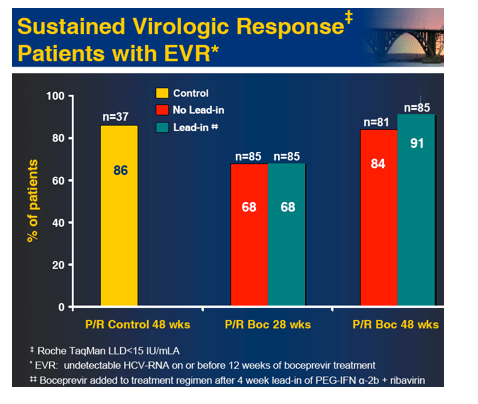
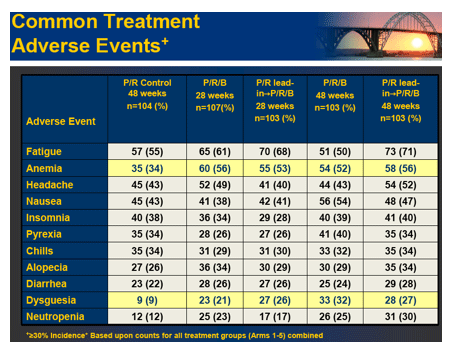
Results: Of 595 patients treated in US (77%), Canada and Europe, 60% were male, 16% Black, 7% cirrhotic, 56% genotype 1a and 89% had high viral load (>600,000 IU/mL). SVR was significantly increased in the 28 and 48 week boceprevir arms compared to P/R Control. RVR and EVR were highly predictive of SVR with boceprevir combinations. Rash-related AEs were similar for boceprevir regimens and P/R Control.
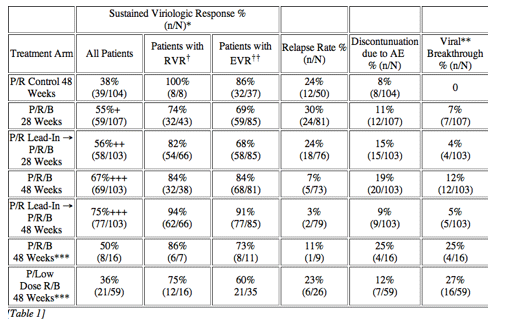
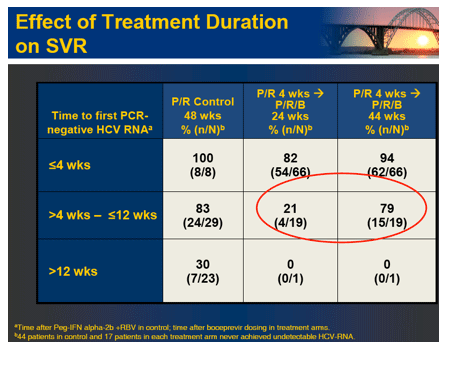
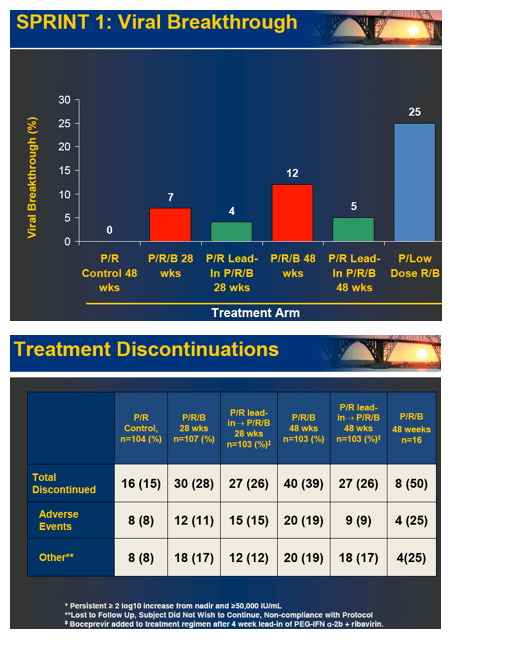
Conclusions: Both 28 and 48 week boceprevir regimens significantly increased SVR with very low relapse rates in 48 week regimens. However, low dose ribavirin with PegIntron and boceprevir was associated with increased viral breakthrough, relapse and lower efficacy. In contrast, P/R lead-in prior to boceprevir substantially increased SVR and reduced viral breakthrough.
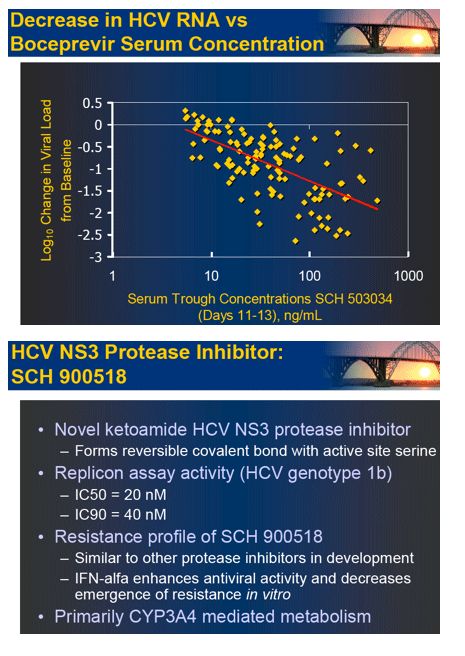
Here are selected slides from Schering symposium with first set below presented by from Stefan Zeuzem, and second slideset by Paul Kwo on SPRINT-1 boceprevir results.


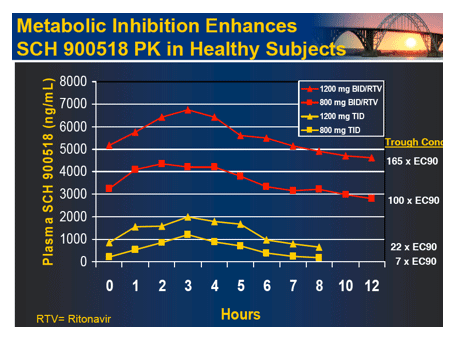





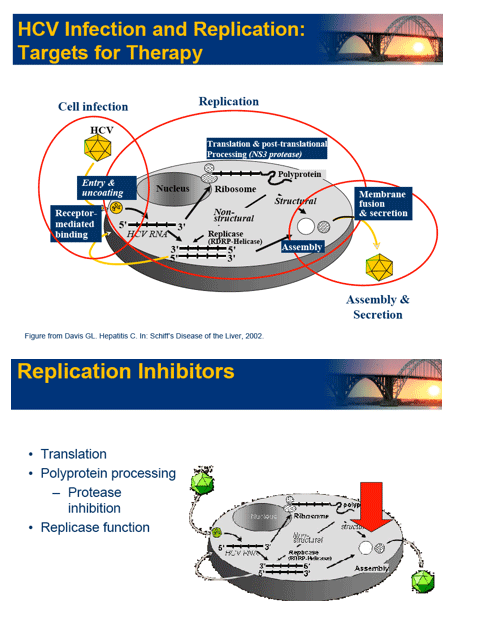
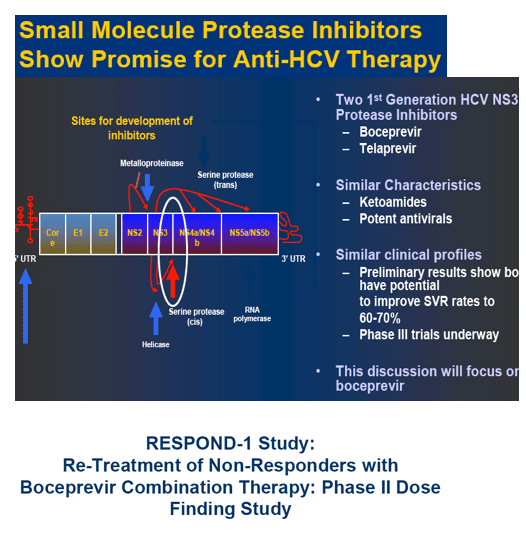

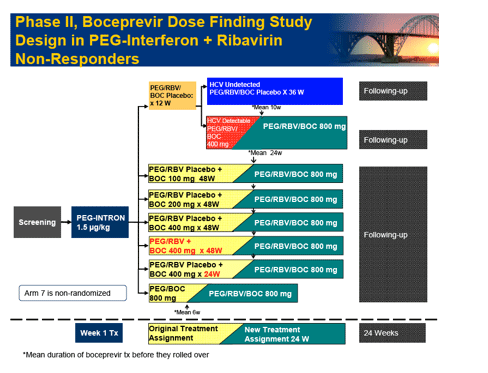

|
| |
|
 |
 |
|
|