 |
 |
 |
| |
Entecavir (ETV) Therapy in Chronic Hepatitis B Patients Previously Treated with Adefovir (ADV) with Incomplete Response On-Treatment or Relapse Off-Treatment
|
| |
| |
44th Annual Meeting of the European Association for Study of the Liver 2009 April 23-26 2009 Copenhagen, Denmark
Reported by Jules Levin
Nancy Leung1, Ching-Lung Lai2, Richard Elion3, Morris Sherman4, Hie-Won Hann5, DJ Lorne Tyrrell6, Chao Wei Hsu7, Chee-Kiat Tan8, Cheng-Yuan Peng9, Kim Mencarini10, Hui Zhang10, Uchenna Iloeje10 and Bruce Kreter11
1Alice Ho Miu Ling Nethersole Hospital, Hong Kong, China; 2Department of Medicine, Queen Mary's Hospital, University of Hong Kong, Hong Kong, China; 3Whitman-Walker Clinic, Washington, WA, USA; 4Toronto General Hospital, Toronto, ON, Canada;
5Liver Disease Prevention Center, Division of Gastroenterology and Hepatology, Thomas Jefferson University Hospital, Philadelphia, PA, USA; 6Health Sciences Center, Edmonton, AB, Canada; 7Chang Gung Memorial Hospital, Chang Gung University College of Medicine, Taipei, Taiwan;
8Department of Gastroenterology and Hepatology, Singapore General Hospital, Singapore; 9China Medical University Hospital, Taichung, Taiwan; 10Research and Development, Bristol-Myers Squibb Company, Wallingford, CT, USA; 11Research and Development, Bristol-Myers Squibb Company, Princeton, NJ, USA
Author Summary of Results
Patients who were suboptimal responders to, or relapsed from ADV during study ETV-079 experienced rapid reductions in HBV DNA when switched to ETV 1.0 mg daily
Mean change in HBV DNA levels were -4.54 log10 at Week 24, and -5.75 log10 at Week 48
HBV DNA levels continued to decline to undetectable levels with extended ETV
treatment
No patients experienced virological rebound during ETV therapy in ETV-901
Author Conclusion
CHB patients switched from ADV to ETV achieve rapid reductions in HBV DNA and increased rates of virological suppression with extended treatment
Introduction
· The primary objective of antiviral treatment for chronic hepatitis B (CHB) is sustained suppression of viral replication and remission of liver disease1
· Many adefovir (ADV) treated patients fail to achieve full viral suppression and ADV resistance is observed more frequently in these patients2,3
· According to CHB treatment guidelines1,4, entecavir (ETV) is recommended for the treatment of patients with ADV-resistant HBV
· Previously presented results of study ETV-079 (E.A.R.L.Y.) demonstrated the superiority of ETV over ADV in HBV DNA reduction as early as treatment day 10, which was maintained through 96 weeks of treatment5, 6
· Patients who did not achieve a response to therapy (HBe seroconversion and
undetectable HBV DNA by PCR), or who relapsed during off-treatment observation were eligible for enrollment into the open-label ETV study AI463-901
Patients and Methods
Study population
· ETV-079 was a randomized, open-label study comparing the early antiviral efficacy, safety and viral kinetics of ETV (0.5 mg daily) with ADV (10 mg daily) in nucleoside-naive HBeAg(+) patients with CHB
· Study ETV-901 is an open-label rollover study of continued ETV treatment for patients previously enrolled in Phase 2/3 ETV clinical studies. Patients were treated with ETV 1.0 mg daily
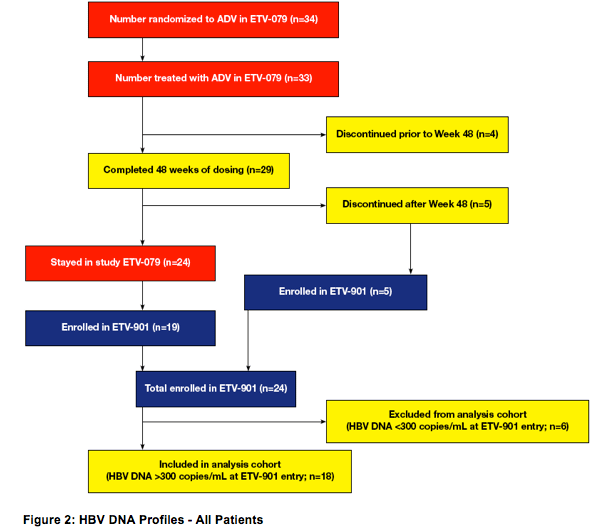
· Twenty-four patients treated with ADV 10 mg daily in ETV-079 enrolled in ETV-901
· This analysis consists of 18 of these 24 patients who at the time of entry into
ETV-901 had detectable serum HBV DNA
- 14/18 patients failed to achieve HBV DNA <300 copies/mL on ADV in ETV-079.
The remaining four patients achieved HBV DNA <300 copies/mL entered postdosing follow-up and experienced virological relapse (treatment gaps from last ADV dose in ETV-079 to first ETV dose in ETV-901: 128, 113, 105 and 98 days)
Study endpoints
· HBV DNA <300 copies/mL
· Serum ALT normalization (≤1 x ULN)
· HBe seroconversion
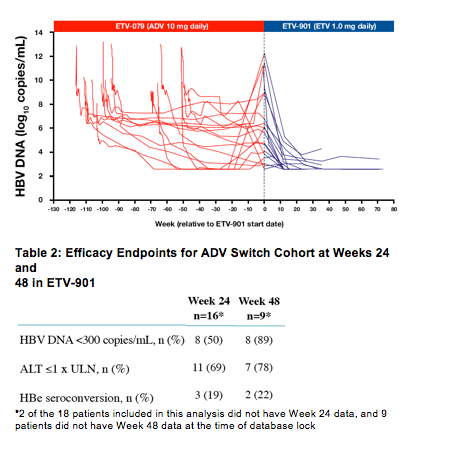
Efficacy assessments were based on available HBV DNA at Weeks 24 (n=16) and 48 (n=9) following switch to ETV in ETV-901 and genotypic resistance was not performed as part of this analysis

HBV DNA
At 24 weeks:
· Mean reduction in HBV DNA was 4.54 log10 copies/mL and 8/16 patients achieved HBV DNA <300 copies/mL
At 48 weeks:
· 8/9 patients (89%) achieved HBV DNA <300 copies/mL
· No patients experienced virological rebound following switch from ADV to ETV in ETV-901
ALT
· At entry into ETV-901, 53% (8/15) of patients had normal ALT. Seven patients who achieved ALT normalization in ETV-079, lost this response by ETV-901 entry
· ALT normalization rates increased to 69% (11/16) and 78% (7/9) at Weeks 24 and 48
respectively in ETV-901
Safety
· The safety profile of ETV in ADV-treated patients remained consistent with previously reported experience5
References
1. Lok AS and McMahon BJ. Hepatology 2007;45:507-539.
2. Lee Y-S, et al. Hepatology 2006;43:1385-1391.
3. Hezode C, et al. J Hepatol 2007;46:791-796.
4. Keeffe EB, et al. Clin Gastroenterol Hepatol 2008;6:1315-1341.
5. Leung N, et al. J Hepatol 2009;42:72-79.
6. Leung N, et al. J Hepatol 2007;46(suppl 1):S24.
|
| |
|
 |
 |
|
|