 |
 |
 |
| |
Preclinical Potency, Pharmacokinetic and ADME Characterization of ABT-333, A Novel Non-Nucleoside HCV Polymerase Inhibitor
|
| |
| |
44th Annual Meeting of the European Association for the Study of the Liver ( EASL), Copenhagen, Denmark, 22-26 April 2009
Reported by Jules Levin
C. Maring*, R. Wagner, D. Hutchinson, C. Flentge, W. Kati, G. Koev , Y. Liu, D. Beno, J. Shen, Y. Lau, Y. Gao, J. Fischer, S. Vaidyanathan, H. Lim, J. Beyer, R. Mondal, A. Molla
Abbott Laboratories, Abbott Park, IL, USA
from Jules: Abbott has an ambitious new drug HCV program. The protease program was reported at EASL by their partner Enanta where they reported preclinical data on 2 HCV protease inhibitors, a report on which I emailed out earlier today; all NATAP email reports are posted on the NATAP website within usually the next day. At EASL Abbott also reported preclinical data on their 2 HCV NNRTI inhibitors, ABT-072 and ABT-333, which are starting in clinical studies.
ABBOTT AND ENANTA INITIATE PHASE 1 CLINICAL TRIAL ON ABT-450 HCV ...
Feb 18, 2009 ... Compounds in various states of development include protease inhibitors (ABT-450) and polymerase inhibitors (ABT-333 andABT-072). ...
www.natap.org/2009/HCV/022709_01.htm
Abbott Acquires HCV Protease Inhibitors from Enanta Pharma
Dec 12, 2006 ... Note from Jules Levin: Abbott is developing HCV polymerase inhibitors in-house so this new development allows them to pursue the development ...
www.natap.org/2006/HCV/121206_02.htm
AUTHOR CONCLUSIONS
ABT-333 is a potent inhibitor of genotype 1a and 1b HCV polymerases with no inhibition of human polymerases.
ABT-333 is a potent inhibitor of genotype 1a and 1b HCV replicons that is attenuated by 12-13 fold in the presence of human plasma.
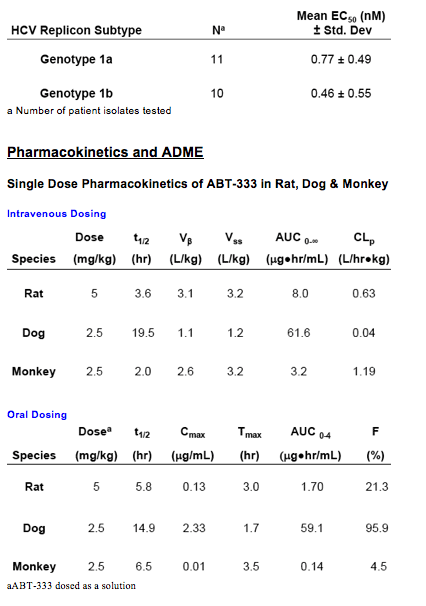
The pharmacokinetic profile of ABT-333 in rat, monkey and dog is characterized by a wide range of plasma clearance values with high volumes of distribution in all species.
Oral doses of ABT-333 distribute preferentially to the liver in rat and monkey at significant multiples over that observed in plasma and at relatively lower multiples in dog.
The CYP metabolism and inhibition profile of ABT-333 together with a low potential for CYP induction suggest a low potential for clinically significant drug-drug interactions.
BACKGROUND
HCV is an enveloped positive-strand RNA virus that replicates primarily in the cytoplasm of hepatocytes. The RNA polymerase (NS5B) encoded by the HCV genome has been shown to be indispensable for HCV replication, making this key viral enzyme an attractive drug target. The structure of HCV polymerase contains the canonical structural features found in most polynucleotide polymerases. However, HCV polymerase has a unique structural architecture and two noteworthy enzymatic properties that distinguish
it from mammalian polymerases: 1) it synthesizes RNA using an RNA template, whereas virtually all mammalian polymerases use DNA as the template source, and 2) unlike mammalian polymerases, HCV polymerase has the ability to initiate RNA synthesis without using an RNA oligonucleotide primer.
Consequently, it has been possible to identify potent and selective inhibitors of HCV polymerase. Nucleoside and non-nucleoside HCV polymerase inhibitors are typically differentiated by their respective binding sites and mechanisms of inhibition. In general, nucleoside inhibitors act as substrates and inhibit
elongation by chain termination, whereas non-nucleoside inhibitors more typically inhibit HCV polymerase at the initiation phase of RNA synthesis. Non-nucleoside inhibitors that bind to each of the known inhibitor binding sites have advanced to human clinical studies and exhibit in vivo activity in HCV-infected patients.
ABT-333 is a novel non-nucleoside HCV polymerase inhibitor with potent activity against genotype 1a and 1b HCV replicons. It is currently being evaluated for safety, pharmacokinetics and antiviral efficacy in chronically-infected HCV patients. This report describes the potency, pharmacokinetic and ADME profile of ABT-333 in preclinical model systems.
METHODS
The polymerase inhibition and selectivity profiles of ABT-333 were determined with a panel of selected HCV NS5B and human polymerase enzymes. Polymerases from HCV genotypes 1-4 were cloned from plasmid DNA or from existing replicon cells and amplified, transfected, expressed and purified using
methods described previously . IC50s were determined by methods described previously. Inhibition of human DNA polymerases by ABT-333 was determined by Replizyme Ltd. Cell culture potency was assessed in HCV replicons with and without 40% human plasma. Transient replicons with polymerases isolated from the serum of HCV-infected humans employed the use of a shuttle vector system. ADME studies with 3H-ABT-333 were conducted in vitro with human microsomes and hepatocytes and in vivo in rats.

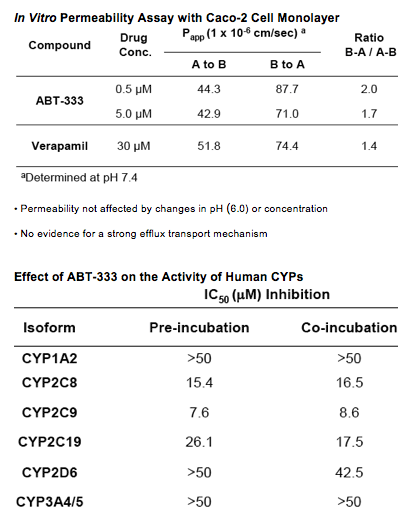

Drug-Drug Interaction Potential of ABT-333
· CYP2C8, 3A4 and 2D6 contribute ~60, 30 and 10% respectively of the control activity to ABT-333 metabolism in human liver microsomes.
· ABT-333 is not an inducer of CYP3A4/5 mRNA in human hepatocytes (incubation at 10 µM, produced <20% of the induction signal of a rifampin positive control.
|
| |
|
 |
 |
|
|