 |
 |
 |
| |
Safety, Tolerability, and Pharmacokinetics of GS-9450 in Healthy Male and Female Volunteers
|
| |
| |
EASL April 23-26 2009 Copenhagen Denmark
Reported by Jules Levin
F. Hoppener1, J.A. Kim2, M.J. Park2, and H.J. Choi2
1KENDLE Clinical Pharmacology Unit, Utrecht, The Netherlands; 2LG Life Sciences, Ltd., Seoul, Republic of Korea

INTRODUCTION
Apoptosis, or programmed cell death, is characterized by distinctive morphological changes such as cell shrinkage and nuclear condensation
Biochemically, a hallmark of apoptosis is the activation of caspases, which are a structurally related family of cysteine aspartyl proteases responsible for cleavage of several critical cellular targets
Caspases can be controlled upstream by the regulation of signals that lead to
zymogen activation, or downstream by inhibitors that prevent them from reaching their substrates
Activation of caspase 8 by apoptotic signaling pathways including Fas and TNF-α, in turn, activates other apical caspases (e.g., caspase 9) or "executioner" caspases (e.g., caspases 3, 6, and 7) that ultimately target a number of cellular components.
This caspase cascade results in the transformation of a functioning cell into an
apoptotic body. Although low levels of apoptosis normally occur to maintain
homeostasis, abnormal amounts of apoptosis occur in disease states, especially in the liver
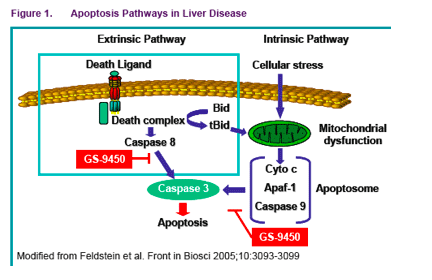
BACKGROUND
Activation of caspases and eventual cell apoptosis has been associated with a
number of liver diseases, including non-alcoholic and alcoholic steatohepatitis,
chronic hepatitis B/C virus infection, and cholestatic liver injury
This suggests that caspase inhibitors may be therapeutically useful in these
diseases. GS-9450 was found to be a very potent inhibitor of caspase activity during in vitro and in vivo pre-clinical studies. These first-in human clinical studies have been performed to assess safety, tolerability and pharmacokinetics (PK) of GS-9450 in healthy volunteers before exploring its therapeutic value in patient groups
OBJECTIVE
To determine the safety, tolerability and pharmacokinetics of GS-9450 following
administration of single and multiple, escalating oral doses of GS-9450 in healthy male subjects and to assess the safety, tolerability and the pharmacokinetics of GS-9450 following administration of a single oral dose in a single cohort of female subjects
To preliminarily assess the effect of food on the pharmacokinetics of GS-9450 in healthy male subjects
METHODS
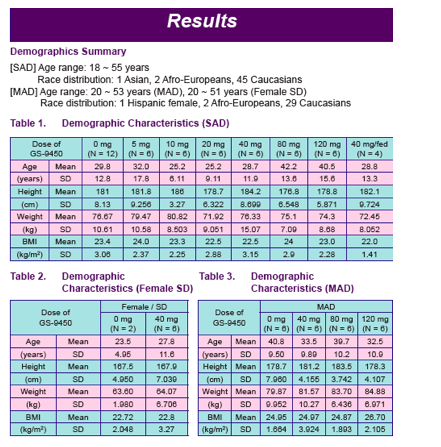
Two, randomized, double-blind, placebo-controlled Phase I studies were performed exposing 54 healthy male subjects and 6 healthy female subjects to GS-9450
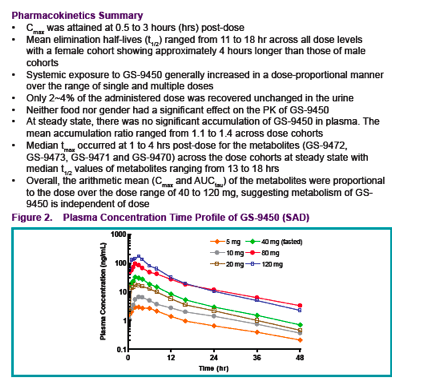
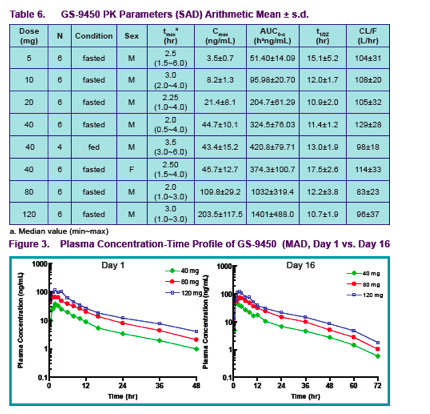
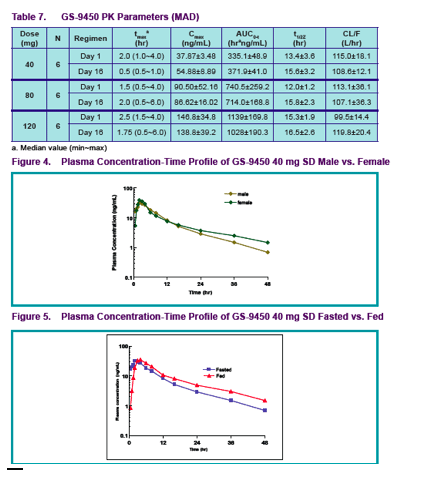
Healthy male subjects received GS-9450 doses of 5 to 120 mg in a single ascending dose (SAD) study and 40 to 120 mg/day for 2 weeks in a multiple ascending dose (MAD) study; female subjects received a single GS-9450 dose (SD) of 40 mg
To evaluate the effect of food intake on the pharmacokinetics of GS-9450, subjects participating in the 40 mg SAD cohort received a second dose of GS-9450 after ingestion of a high fat breakfast
Blood and urine samples were analyzed to determine GS-9450 and metabolite
concentrations in order to calculate single dose and steady state PK parameters
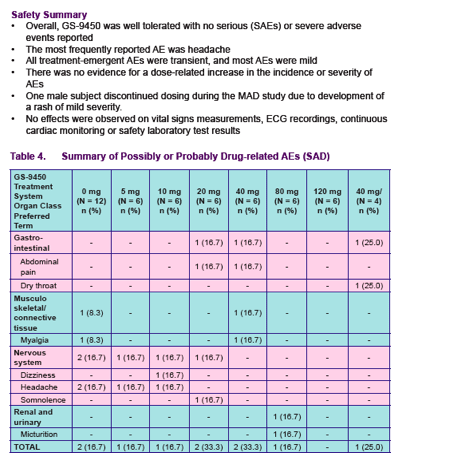
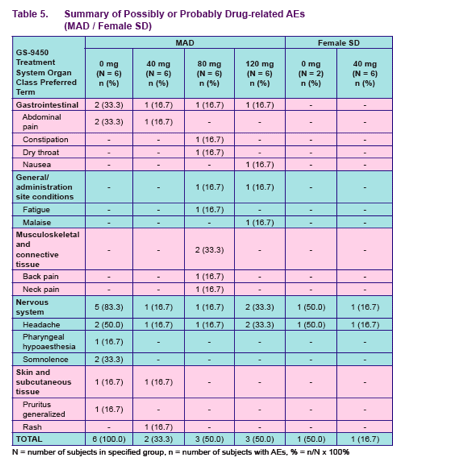
Safety and tolerability were assessed by means of recording of adverse events
(AEs) vital signs measurements, 12-lead ECG recordings, clinical laboratory tests and physical examinations
|
| |
|
 |
 |
|
|