 |
 |
 |
| |
Albuferon vs Pegasys Genotype 1
|
| |
| |
Slides follow abstract below
EFFICACY AND SAFETY OF ALBINTERFERON ALFA-2B IN COMBINATION WITH RIBAVIRIN IN TREATMENT-NAÏVE, CHRONIC HEPATITIS C GENOTYPE 1 (CHC G1) PATIENTS
Reported by Jules Levin
S. Zeuzem1, M. Sulkowski2, E. Lawitz3, V. Rustgi4, Y. Lurie5, M. Grigorescu6, A. Muir7, P. Cronin8, E. Pulkstenis8, M. Subramanian8, J. McHutchison7
1W. Goethe-University Hospital, Frankfurt, Germany, 2Johns Hopkins University, Baltimore, MD, 3Alamo Medical Research, San Antonio, TX, 4Metropolitan Research, New York, NY, USA, 5Sourasky Medical Centeris, Tel-Aviv, Israel, 6Clinica Medicala III, Cluj-Napoca, Romania, 7Duke Clinical Research Institute, Chapel Hill, CA, 8Human Genome Sciences, Rockville, IN, USA
Background: A phase 3, randomized, active controlled, multi-center study evaluated the efficacy and safety of albinterferon alfa-2b (albIFN), a genetic fusion polypeptide of albumin and interferon alfa-2b in CHC G1 patients.
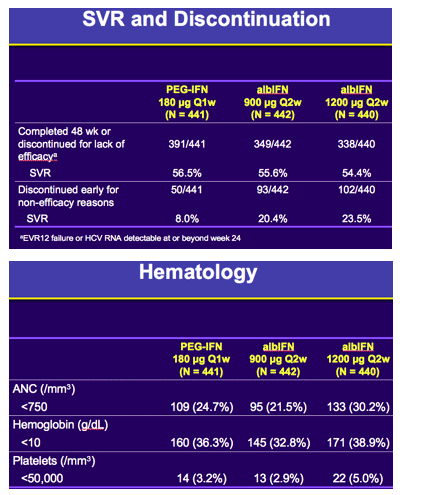
Methods: In total, 1331 patients were randomized 1:1:1 to one of the 3 treatment groups: alb-IFN900µg q2weeks; alb-IFN1200 µg q2weeks; PEG-IFNα2a 180 µg q1week for 48 weeks, in combination with weight-based oral ribavirin 1000-1200 mg/day. Randomization was stratified by baseline HCV RNA levels (≥ or < 800,000 IU/mL), BMI (≥ or < 25 kg/m2) and race (African-American or other). The primary end point was sustained virologic response (SVR), defined as serum HCV RNA < 10 IU/mL at week 72. The sample size provided 90% power to demonstrate non-inferiority with a margin of 12%. According to Data Monitoring Committee recommendation, all patients receiving alb-IFN 1200µg were reduced to 900µg q2weeks, which impacted 51% of this group.
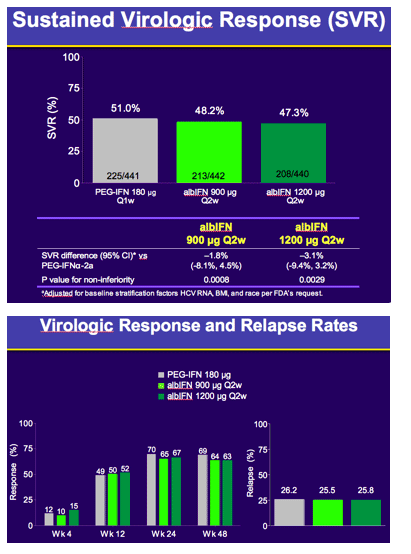
Results: The study achieved the primary objective of demonstrating non-inferiority of albIFN 900µg (p=0.0008) and albIFN 1200µg (p=0.0029) versus PEG-IFNα2a for SVR.
In the intention-to-treat population, SVR rates were 51.0% (225/441), 48.2% (213/442), and 47.3% (208/440) in the PEG-IFNα2a, albIFN900 and albIFN1200 groups respectively. Multivariate analysis identified the following predictors of SVR: HCV RNA < 400,000 IU/ml, age < 45, normal GGT, high ALT, F0-2 fibrosis, and race, consistent with previous studies.
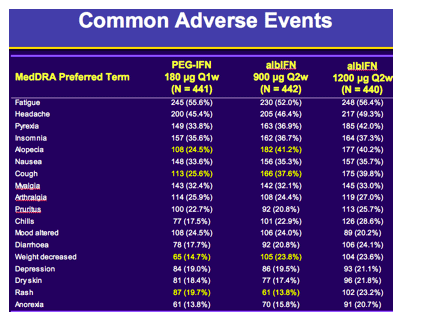
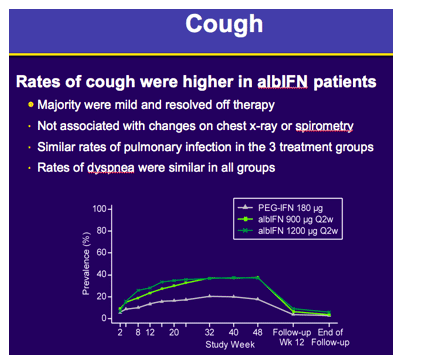
Safety analyses showed: 1) Rates of serious and/or severe adverse events (AE) were 23.1%, 24.0% and 28.2%; 2) Treatment discontinuations due to AEs were 4.1%, 10.4% and 10.0%; 3) Severe or serious pulmonary events were rare and blinded central review of Chest X-rays (n=753) during treatment showed comparable rates of interstitial lung findings: 4.2%, 2.4% and 4.5%; 4) Mortality rates of 1/441, 0/442 and 2/440 in the PEG-IFNα2a, albIFN900 and albIFN1200 groups, respectively. The rates of hematologic abnormalities were numerically lower in albIFN900 versus PEG-IFNα2a.
Conclusions: Albinterferon alfa-2b 900µg administered q2wk demonstrated comparable efficacy to PEG-IFNα2a in patients with chronic hepatitis C genotype 1. The overall incidence of adverse events, serious or severe adverse events was similar between these two treatments.





|
| |
|
 |
 |
|
|