 |
 |
 |
| |
Preclinical Pharmacokinetic and ADME Characterization of VCH-222, a Novel Non-Nucleoside HCV NS5B Polymerase Inhibitor from Vertex/ViroChem
|
| |
| |
44th EASL Apr 23-26 2009 Copenhagen Denmark
Reported by Jules Levin
N. Chauret, C. Chagnon-Labelle, M. Diallo, J. Laquerre, J. Laterreur, S. May and L. Ste-Marie
ViroChem Pharma Incorporated, Laval, QC, Canada
Supported by ViroChem Pharma Incorporated, a wholly-owned subsidiary of Vertex Pharmaceuticals Incorporated
AUTHOR CONCLUSIONS
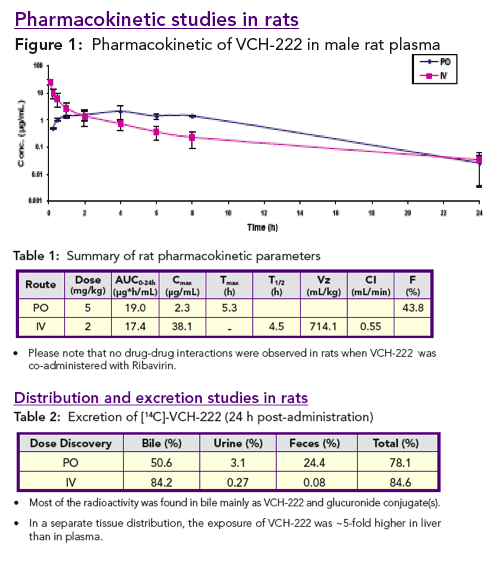
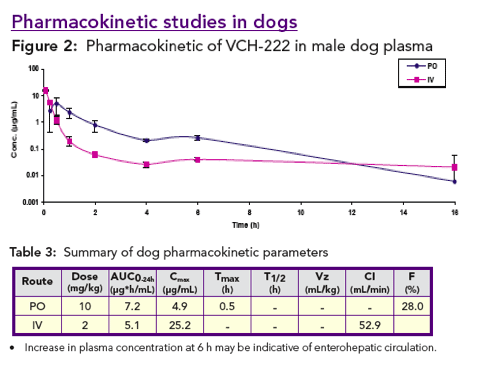
VCH-222 has good oral bioavailability in rats and dogs. In rats, VCH-222 concentrates in the liver and is mainly eliminated through biliary excretion.
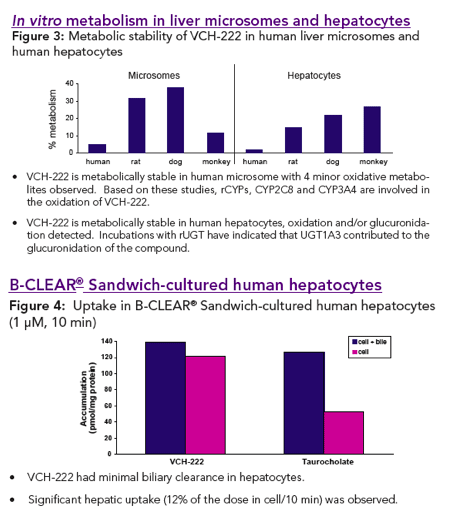
VCH-222 exhibits very good ADME properties in terms of permeability and metabolic pathways.
In vitro studies revealed that several pathways (oxidation and glucuronida-tion) using several enzymes (CYPs and UGT) are involved in the biotransfor-mation of VCH-222. There was no evidence for the potential of reactive intermediates upon oxidative bio-activation (results not shown).
Significant uptake and minimal biliary excretion clearance was observed for VCH-222 in cultured human hepatocytes.
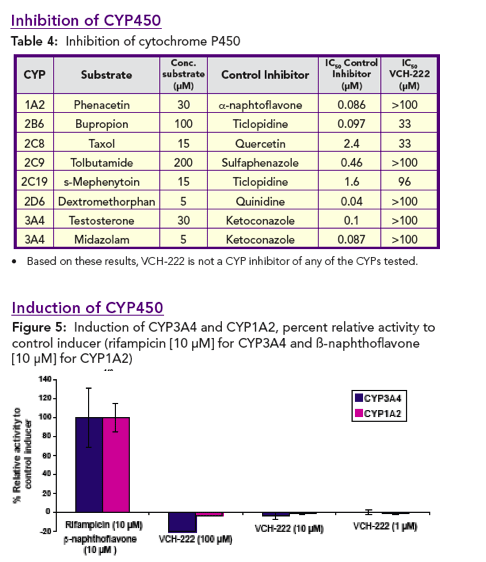
Based on these in vitro studies, VCH-222 is not a CYP inhibitor, CYP inducer or MDR1 inhibitor. No drug interactions were observed in rats when VCH-222 was co-administered with Ribavirin.
BACKGROUND
HCV is a debilitating human pathogen affecting an estimated 3% of the world's popu-lation. Current therapies are associated with limited efficacy and severe adverse side effects.1,2 Therefore, the development of new safe and effective HCV-specific antiviral agents is needed to alleviate this serious health problem.
The RNA-dependent RNA polymerase (NS5B) from HCV is a key enzyme in HCV replica-tion and is a major target for the development of antiviral compounds against HCV.3
VCH-222, a thiophene 2-carboxylic acid derivative, is a novel non-nucleoside HCV RNA poly-merase inhibitor, currently in clinical evaluation in HCV-infected patients.
OBJECTIVES
To evaluate the pharmacokinetic profiles of VCH-222 in rats and dogs.
To determine the excretion and distribution of [14C]-VCH-222 in rats
To evaluate the in vitro metabolic characteristics of VCH-222 in liver microsomes and hepa-tocytes from preclinical species and from humans.
To evaluate, via in vitro studies, the potential of VCH-222 to cause drug-drug interac-tions through CYP and MDR1 enzymes.
METHODS
Pharmacokinetic profiles: VCH-222 was administered to rats and dogs orally in 30% PEG 400 at 5 and 10 mg/kg respectively, and intravenously in 60% PEG 400 for rats and in 30% PEG 400 for dogs at 2 mg/kg. Ribavirin was co-administered with VCH-222 to rats at 40 mg/kg in 0.5% CMC.
In vivo excretion Studies: [14C]-VCH-222 was administered to rats at 5 and 0.2 mg/kg (PO and IV, respectively) and selected fluids were collected over a 24 h period.
In vitro excretion Studies: Biliary excretion studies were performed in B-CLEAR® Sandwich-Cultured human hepatocytes using 1 uM of [14C]-VCH-222.
In vitro metabolism studies: VCH-222 (5 µM) was incubated with liver microsomes (1.0 mg/mL) or hepatocytes (106 cells/mL) obtained from rat, dog, monkey and human for 1.5 h in presence of NADPH (1.5 mM) or 2 h, respectively. Incubations in microsomes expressing individual CYP (20 pmol) or UGT (0.5 mg/mL) using NADPH (1.5 mM) or UDPGA (1.5 mM) as co-factor, respectively, were also conducted. Incubates were ana-lysed by HPLC/UV and MS for parent disappearance and for metabolite formation.
In vitro CYP inhibition: The effect of VCH-222 on CYP450 activity was studied in hu-man liver microsomes supplemented with NADPH (1 mM) using selective substrates.
In vitro CYP induction:The effect of VCH-222 on the CYP3A4 and 1A2 activity and mRNA levels was evaluated in cultured human hepatocytes (1.4 x 105 cells, 48 h).
Permeability and in vitro inhibition of MDR1: The permeability of VCH-222 and its effect on the efflux of digoxin was studied in CaCo-2 cell monolayers grown to conflu-ence on polycarbonate membranes.
RESULTS

REFERENCES
1. Washley A; Alter MJ. Epidemiology of hepatitis C: geographic differ-ences and temporal trends. Semin. Liver Dis. 2000;20:1-16.
2. Fried MW; Shiffman ML; Reddy KR; et al. Peginterferon alfa-2a plus ribo-virin for chronic hepatitis C virus infection. NEJM 2002;347:975-82.
3. De Francesco R; Tomei L; Altamura S; et al. Approaching a new era for hepatitis C virus therapy: inhibitors of the NS3-4A serine protease and the NS5B RNA-dependent RNA polymerase. Antiviral Res 2003;58:1-16.
|
| |
|
 |
 |
|
|