 |
 |
 |
| |
EFFICACY AND SAFETY OF PEGINTERFERON ALFA-2A 360 µg/WEEK IN COMBINATION WITH RIBAVIRIN IN HEPATITIS C GENOTYPE 1 PATIENTS - IMPACT Of CIRRHOSIS: ANALYSIS FROM THE CHARIOT STUDY
|
| |
| |
EASL April 23-26 2009 Copenhagen
Reported by Jules Levin
W.S.C. Cheng,1 S. Roberts,2 M. Weltman,3 D.H.G. Crawford,4 W. Sievert,5 G. McCaughan,6 W. Rawlinson,7 P. Desmond,8 M. Yoshihara,9 B. Rizkalla,9 J. DePamphilis,10 P. Marks,11 G.J. Dore11 on behalf of the CHARIOT Study Group
1Royal Perth Hospital, Perth, Australia; 2Alfred Hospital, Melbourne, Australia; 3Nepean Hospital, Sydney, Australia; 4Greenslopes Hospital, Brisbane, Australia; 5Monash Medical Centre, Melbourne, Australia; 6Royal Prince Alfred Hospital, Sydney, Australia; 7SEALS Microbiology, Prince of Wales Hospital, Randwick, Australia;
8St. Vincent's Hospital, Melbourne Australia; 9Roche Products, Sydney, Australia; 10Roche, Nutley, NJ, USA; 11National Centre in HIV Epidemiology and Clinical Research, Sydney, Australia
This study is sponsored by Roche Pty Ltd, Australia
AUTHOR CONCLUSIONS
Patients with no or mild fibrosis respond significantly better to pegylated interferon and ribavirin therapy than patients with advanced fibrosis. The high SVR rates in patients with no or mild fibrosis support early treatment for CHC.
There was no difference in SVR between induction and standard dosing regimens of peginterferon alfa-2a (40KD) in patients with advanced fibrosis or cirrhosis. From Jules: see the table below which shows a slight benefit with induction dosing in patients with 0-2 cirrhosis although the incremental benefit may not be very much nor statistically significant. Perhaps week 4 and 12 evaluation of therapy and adherence to dosing might help to identify patients who could benefit from induction dosing.
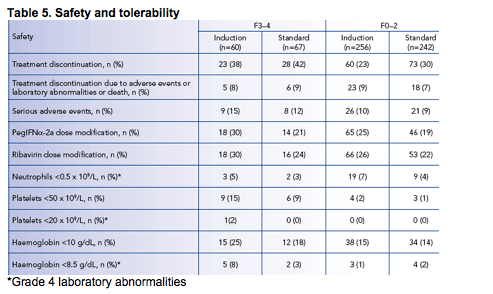
Induction dosing was well tolerated in patients with advanced fibrosis or cirrhosis (METAVIR F3/4).
Lower EVR and higher relapse rates among patients with F3/4 compared with F0-2, despite receiving similar cumulative dosages of peginterferon alfa-2a (40KD) and ribavirin, suggest that differential intrahepatic viral clearance or drug metabolism may partly explain the poorer responses.
BACKGROUND
Chronic hepatitis C (CHC)-related compensated cirrhosis is associated with a high risk of progression to liver decompensation, hepatocellular carcinoma (HCC) and liver-related mortality.[1] However, CHC patients with cirrhosis who achieve a sustained virological response (SVR) to treatment have a significantly lower risk of end-stage liver disease.[2, 3]
CHC patients with compensated cirrhosis have SVR rates of around 30% with combination pegylated interferon and ribavirin therapy, which is significantly lower than the overall approximate 50% SVR in genotype 1 patients.[4, 5] Optimal therapy for patients with compensated cirrhosis associated with hepatitis C virus (HCV) has not been well established. Induction therapy with pegylated interferon for the first 12 weeks has the potential advantage of increasing rapid virological response (RVR) and early
virological response (EVR), both of which have been shown to be good predictors of SVR.
The CHARIOT study is an international, multicentre, randomised, open-label active-controlled study that evaluates the efficacy and safety of a pegylated interferon induction dosing regimen in treatment-naïve patients with HCV genotype 1 infection.
OBJECTIVE
To compare the efficacy and safety of induction dosing with 360 µg weekly peginterferon alfa-2a (40KD) (PEGASYS to standard-dose 180 µg weekly peginterferon alfa-2a (40KD) (both in combination with standard-dose ribavirin) in patients with advanced fibrosis or cirrhosis (F3/F4 by METAVIR) enrolled in the CHARIOT study.
Primary endpoint
SVR, defined as undetectable HCV RNA by Roche Ampliprep/Cobas™ TaqMan™ HCV Test, (limit of detection 15 IU/mL) 24 weeks after completion of 48 weeks of therapy.
Secondary endpoints
RVR, defined as undetectable HCV RNA at the end of 4 weeks of therapy.
Complete EVR (cEVR), defined as undetectable HCV RNA at the end of 12 weeks of treatment.
End of treatment virological response (ETR), defined as undetectable HCV RNA at the end of 48 weeks of treatment.
Relapse, defined as detection of HCV RNA at the end of follow-up in a patient with an ETR (only patients with an ETR are included in the calculation of relapse).
Safety and tolerability.
METHODS
Inclusion criteria
Chronic HCV genotype 1 infection.
Age 18-75 years.
HCV RNA > 600 IU/mL.
Compensated liver disease (Child-Pugh score <7).
Exclusion criteria
Other causes of liver disease.
Infection with non-genotype 1 HCV, hepatitis B virus or HIV.
Decompensated liver disease.
Complications of liver disease such as variceal bleeding and HCC.
Immunomodulatory or antineoplastic drugs within previous 6 months.
Alcohol consumption >20 g/day.
Treatment
Overall, 896 patients were stratified by baseline HCV RNA level and country, and randomised 1:1 to peginterferon alfa-2a (40KD) 180 µg/week for 48 weeks (standard dosing) or peginterferon alfa-2a (40KD) 360 µg/week for 12 weeks followed by 180 µg/week for 36 weeks (induction dosing).
All patients received ribavirin 1000-1200 mg/day for 48 weeks.
In total, 871 patients received the study regimen and were evaluable for intention-to-treat efficacy and safety analysis.
Here we show results from exploratory analyses to generate hypotheses for further research.
RESULTS
Baseline liver histology was obtained in 625 of 871 patients (71.8%).
In total, 127 patients (14.6%) were METAVIR F3/F4 (F3= 97, F4= 30), and 498 patients (57.2%) with F0-2 were identified.
Patients with F3/4 were older with higher mean body weight and baseline alanine aminotransferase levels compared with patients with F0-2 (Table 1).
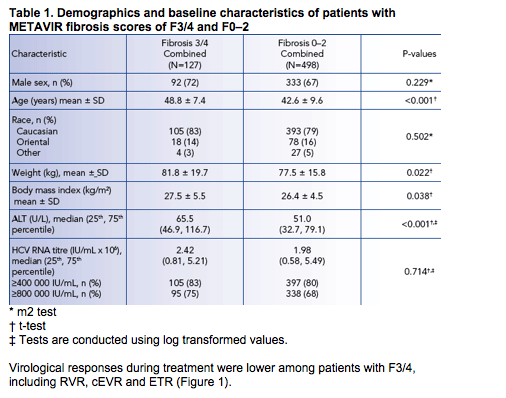
Virological responses during treatment were lower among patients with F3/4, including RVR, cEVR and ETR (Figure 1).
Among patients with F3/4, on-treatment responses were 5-10% higher in the induction than the standard dosing groups (Figure 1), but SVR rates were similar (28% and 24%) and considerably lower than among the corresponding patient groups with F0-2 (58% with induction dosing and 55% with standard dosing)
(Figure 2).
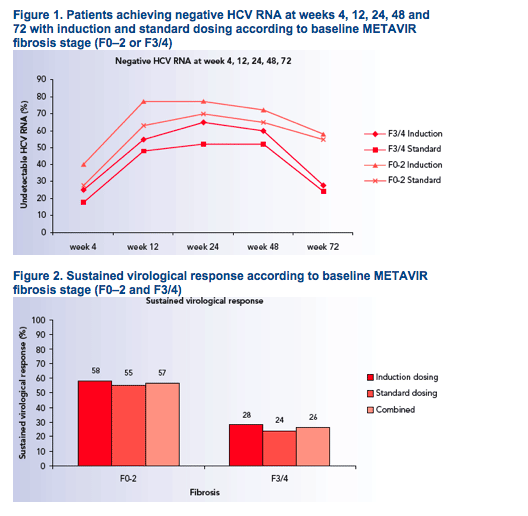
Considerably higher relapse rates contributed to the lower SVR rates among patients with F3/4 (Figures 3 and 4)
Relapse rates in patients who received induction dosing and standard dosing, respectively, were:
- 19% and 10% for F0
- 15% and 15% for F1
- 19% and 15% for F2
- 41% and 34% for F3
-- 56% and 60% for F4 (Figure 4).
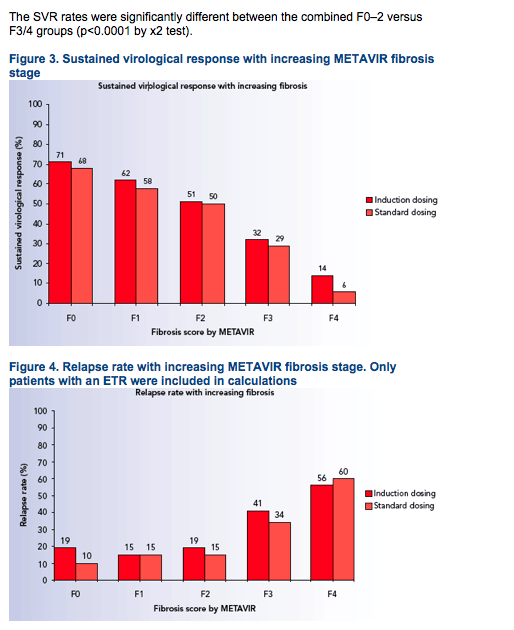
The best negative predictior for SVR was the absence or a >2 log10 drop in HCV RNA at week 12 (Table 2).
The positive predictive value of RVR for SVR was lower in patients with F3/4 (60% with induction dosing and 67% with standard dosing) than F0-2 (79% with induction dosing and 82% with induction dosing).
The negative predictive value of the absence of an RVR for not achieving an SVR was considerably higher in patients with F3/4 (83% with induction dosing and 84% with standard dosing) compared with F0-2 groups (57% with induction dosing and 55% with standard dosing).

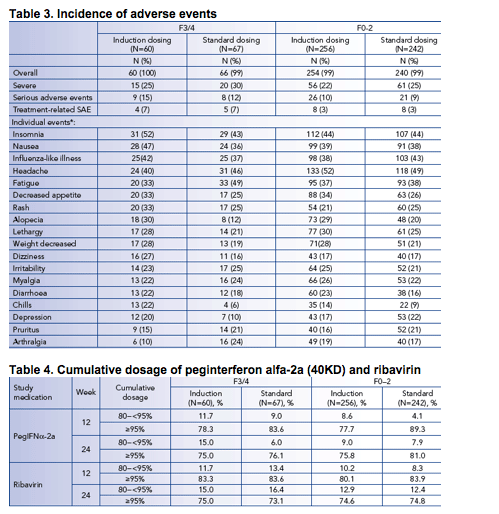
The mean cumulative dose and the proportion of patients receiving ≥80% of the planned dose of peginterferon alfa-2a (40KD) and ribavirin through week 12 was similar for patients with baseline METAVIR fibrosis stages of F3/4 and F0-2 (Table 4).
Treatment discontinuation rates were similar in the induction and standard dosing groups (<10%).
Dosage modification of peginterferon alfa-2a (40KD) and ribavirin was highest in patients with F3/4 in the induction dosing group (Table 5).
References
1. Salomon JA, Weinstein MC, Hammitt JK, Goldie SJ. Empirically calibrated model of hepatitis C virus infection in the United States. Am J Epidemiol 2002; 156(8):761-773.
2. Shiratori Y, Ito Y, Yokosuka O et al. Antiviral therapy for cirrhotic hepatitis C: association with reduced hepatocellular carcinoma development and improved survival. Ann Intern Med 2005; 142(2):105-114.
3. Bruno S, Stroffolini T, Colombo M et al. Sustained virological response to interferon-alpha is associated with improved outcome in HCV-related cirrhosis: a retrospective study 3. Hepatology 2007; 45(3):579-587.
4. Bruno S, Hadziyannis S, Shiffman M et al. Baseline and on-treatment factors associated with high rates of sustained virological response in patients with and without cirrhosis following treatment with peginterferon alfa-2a and ribavirin [abstract 774]. J Hepatol 2008; 48(Suppl 2):S289.
5. Roffi L, Colloredo G, Pioltelli P et al. Pegylated interferon-alpha 2b plus ribavirin: an efficacious and welltolerated treatment regimen for patients with hepatitis C virus related histologically proven cirrhosis. Antivir Ther 2008; 13(5):663-673.
|
| |
|
 |
 |
|
|