| |
Complete cure of HBV-HDV co-infection after 24weeks of combination therapy with pegylated interferon and ribavirin in a patient co-infected with HBV/HCV/HDV/HIV
|
| |
| |
Joel Gozlan14, Karine Lacombe256, Elyanne Gault3, Gilles Raguin2, Pierre-Marie Girard256
Jnl of Hepatology Feb 2009
Multiple hepatitis co-infections are frequent in HIV-infected patients, often resulting in severe liver diseases that are difficult to treat. We report here the complete resolution of a chronic hepatitis B and D co-infection in a patient who was also infected with HCV and HIV. This cure was observed after 24weeks of combination therapy associating pegylated-IFN and ribavirin, which was initially given to treat HCV. An unexpected and extensive HDV replication was observed in this patient after HBs Ag had cleared from the serum, which was followed by a stable halt in HDV replication. Implications of this unusual observation will be discussed.
Associate Editor: F. Zoulim
1. Background
Chronic HBV-HDV co-infection usually results in severe and rapidly progressive liver disease, which is difficult to treat. interferon-α (IFN) is the only approved treatment against such co-infections and must be administered at high doses for a prolonged period of time if a long-term response is to be achieved [1]. Clinical trials evaluating pegylated interferon-α (peg-IFN) in HBV-HDV co-infected patients have recently demonstrated promising, but conflicting results [2], [3], [4]. We describe here the complete resolution of HBV-HDV co-infection in a patient also infected with HCV and HIV, after a 24-week course of peg-IFN and ribavirin (RBV). Surprisingly, a transient but extensive HDV replication occurred in this patient after HBsAg seroconversion, followed by a stable halt in HDV replication.
2. Case report
A 44-year-old man presented with positive HIV-1 serology at first observation in March 1996. He had quadruple infection with positive HBsAg, anti-HCV, and total anti-HDV antibodies. HCV RNA (genotype 1) was detected in serum, but neither IgM anti-HBc nor IgM anti-HDV were found. Initial biochemical liver tests were normal. Because of a high HIV viral load (VL) and a low CD4 cell count, combined antiretroviral therapy (cART) with stavudine, lamivudine and indinavir was immediately initiated. This therapy was successful in rapidly reducing the HIV viral load and increasing the CD4 cell count. HIV-1 infection remained controlled under ART, which consistently included lamivudine. Hepatic tests remained within the normal range until February 2001 at which time liver enzymes significantly increased (Fig. 1). A liver biopsy performed in April 2001 indicated a Metavir score of A2 F1. HBsAg was still detectable at this time (October 2001), but the HBV viral load was below 125,000IU/ml (sensitivity threshold of the branched DNA assay used) and HBeAg was undetectable. HDV RNA quantification was not available at that time. Liver enzymes fluctuated after that, reaching the highest levels between September 2001 and February 2002. In consideration of the significant aminotransferase flare, a combination therapy with PEG-IFN α2b (120μg per week) and RBV (1000mg per day) was started in April 2002, during which time the ART regimen comprised lamivudine, stavudine and efavirenz. Baseline HCV RNA was over 3x106IU/ml and was still detectable after 24weeks of treatment (45,700IU/ml) when both PEG-IFN and RBV were discontinued due to poor tolerability. However, the interrupted anti-HCV therapy had an unexpected effect on HBV infection resulting in HBsAg clearance in October 2002. A sensitive PCR assay (HBV Cobas Monitor Roche Diagnostic, sensitivity: 60IU per ml) showed negative serum HBV DNA. Moreover, the patient seroconverted to anti-HBs (15mIU/ml), suggesting cure of HBV infection. HBsAg clearance was surprisingly associated with a high level of HDV replication, with HDV RNA levels of 9.3x106copies/ml by real time PCR [5]. This peak of HDV replication was transient as HDV infection resolved after HBV, with clearance of serum HDV RNA 6 months later (tested with the same PCR assay, sensitivity: 100copies/ml). This profile of dual cure of HBV and HDV persisted over time, and was accompanied by an increase in anti-HBs titre (>150mIU/ml on October 2003); HBsAg, HBV DNA, and HDV RNA had also been negative in serum every year since 2003. No further treatment for HCV infection was attempted. At present the patient is doing well and his HIV infection is currently under control with abacavir, lamivudine and efavirenz. Biochemical markers of liver functions have remained persistently normal, whereas transient elastometry indicated a liver stiffness of 7.8kPa, suggesting the presence of moderate fibrosis.
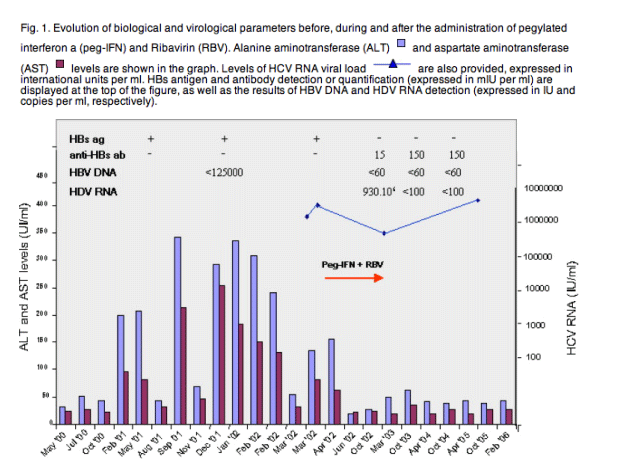
3. Discussion
Two main findings are remarkable in this case report. The first is that a sustained resolution of HBV-HDV co-infection was observed after only 6 months of PEG-IFN and RBV. Although pilot studies have suggested that PEG-IFN may increase the rate of anti-HDV response [2], [3], [4], complete responses are still anecdotal and patients require prolonged exposure to therapy. No cure of HBV-HDV infection under PEG-IFN has ever been described in HIV-HBV-HDV-HCV co-infected patients, with the exception of a single case [6] in which treatment with peg-IFN-α2b for 18 months resulted in HBV DNA and HDV RNA clearance. However, neither HBsAg clearance nor seroconversion to anti-HBs antibodies had occurred. We believe that treatment success may be due to the immune response developed by the patient a few months before starting PEG-IFN, resulting in an aminotransferase flare. We feel that initiation of interferon treatment was timely resulting in enhancement of the immune response, thus positively affecting HBV resolution. The inhibitory effects of HDV and/or HCV on HBV replication [7], [8] might be another factor that facilitated the combined effect of IFN and the immune response. The contribution of lamivudine and/or RBV to HBV-HDV resolution is unlikely since many studies have shown no benefit upon addition of these drugs to IFN in patients with delta hepatitis [8], [3], [9]. The other remarkable finding in this observation is the high HDV replication observed after HBs Ag clearance from serum. HBsAg is required for packaging, export and spread of HDV particles to other cells. However, continuous replication of HDV with the helper function of HBV has been shown in a cell culture system, indicating that replication of HDV genome itself is independent from HBV [10]. In liver transplants for HBV-HDV have related cirrhosis, HDV re-infection of the graft with subsequent HDV viremia has been observed in the absence of detectable HBV in the blood, raising the hypothesis of an "autonomous" HDV infection occurring in vivo [7]. The use of sensitive PCR-based assays for HBV detection however challenged this hypothesis [11], suggesting that these observations likely rely on a true dual infection of hepatocytes, with very low levels of HBV replication. In our opinion, a transient persistence of HBV replication in co-infected hepatocytes, at the time when HBsAg was no longer detectable in the blood, may explain the isolated detection of HDV RNA. Another hypothesis is the occurrence of a restricted, HBV independent, replication of HDV RNA genome, followed by the release of viral ribo-nucleoprotein caused by the immune-mediated destruction of infected hepatocytes. Whatever the precise mechanism of this unusual finding, this "isolated" HDV detection was transient, in agreement with experiments performed in a simian model [11].
4. Conclusions
Our observation confirms the potential efficacy of PEG-IFN α for HBV-HDV co-infection in a multiple infection setting, especially if therapy is administered at the time when spontaneous immune response has started.
|
|
| |
| |
|
|
|