| |
Antiviral therapy for hepatitis B-related
liver cancer prevention is more cost-effective than cancer screening
|
| |
| |
Articles in Press
"HCC prevention (selected treatment) appears to be a cost-effective public health strategy in at-risk populations in Australia and is preferable to HCC surveillance as a cancer control strategy."
"While CHB treatment is expensive, targeting pharmacological management to patients at highest risk (i.e. people with high HBV DNA and elevated ALT levels) provides more substantial gains in health outcomes and offers better value for money, compared to HCC surveillance in individuals with CHB infection. A focused program of screening and treatment of CHB among people born in Asian countries would address the public health challenge of offering a cost-effective treatment to populations most likely to benefit from them, where CHB infection rates are highest. Tailoring such a program to particular high-risk groups would raise medical practitioner awareness for the need to recommend HBV screening, ensure the provision of appropriate health care for those infected and allow improved measurements of the disease burden in these populations. Whether the same benefits are applicable to Caucasian-born populations remains unclear, as differences in the natural history of hepatitis B infection in Caucasian patients may lead to differences in liver-related morbidity and mortality [48].
The burden of CHB and HCC in developed nations is likely to increase significantly in the future, due to the large population at risk, changing patterns of immigration and the long latency for progression from CHB to HCC. Therefore, devising cost-effective programs to reduce the burden of disease should be a key component of health planning and in this context, our study provides valuable information to help guide the clinical implementation of such a program."
Journal of Hepatology
Monica C. Robotin12, Melanie Kansil3, Kirsten Howard2, Jacob George45, Steven Tipper1, Gregory J. Dore6, Miriam Levy7, Andrew G. Penman8
Received 25 August 2008; received in revised form 19 December 2008; accepted 29 December 2008. published online 17 February 2009.
Corrected Proof
Background/Aims
In Australia, Asian-born populations are 6-12 times more likely to develop hepatocellular cancer (HCC) than Australian-born individuals. We therefore, modelled the consequences of different management strategies for chronic hepatitis B (CHB) in Asian-born adults aged >35 years.
Methods
A Markov model compared (1) enhanced surveillance for HCC alone (HCC surveillance), or (2) enhanced HCC surveillance coupled with CHB treatment (HCC prevention) to the current practice, of low CHB treatment uptake. Patients were stratified and managed according to risk categories, based upon hepatitis B virus (HBV) viral load and alanine aminotransferase (ALT) levels. We measured costs, health outcomes [cases of HCC and deaths averted, quality-adjusted life-years (QALYs) gained] and incremental cost-effectiveness ratios (ICERs).
Results
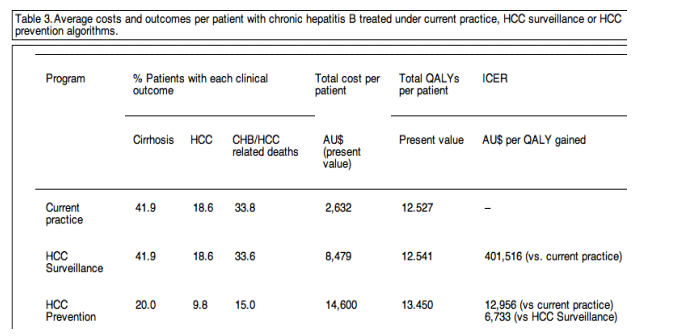
HCC surveillance would cost on average AU$8479 per person, compared to AU$2632 with current clinical practice and result in a gain of 0.014 QALYs (AU$401,516/QALY gained). A HCC prevention strategy would cost on average AU$14,600 per person, result in 0.923 QALYs gained (AU$12,956/QALY gained), reduce cases of cirrhosis by 52%, HCC diagnoses by 47% and CHB-related deaths by 56%, compared to current practice.
Conclusions
HCC prevention appears to be a cost-effective public health strategy in at-risk populations in Australia and is preferable to HCC surveillance as a cancer control strategy.
Associate Editor: J.M. Llovet
Background
Although relatively uncommon in Australia (where it ranks 15th in males and 20th in females), hepatocellular cancer (HCC) incidence has been progressively rising, with age-standardised rates projected to increase by 27% in females and 43% in males and during the current decade [1]. In the state of New South Wales (NSW), HCC incidence rates have been rising faster than those of any other internal cancer [2], with people born in areas of high chronic hepatitis B (CHB) prevalence (in particular Vietnam, Korea, Indonesia and China), being 6-12 times more likely to have a liver cancer diagnosis than the non-indigenous Australian-born populations [3]. As hepatitis B is the most common cause of HCC in the Asian region [4], [5], effective suppression of viral replication may be a promising avenue for cancer control in these populations [6], [7], [8], [9], if questions around treatment effectiveness, cost and duration can be satisfactorily addressed.
Currently, patients with HCC often present late, when options for treatment are limited and survival rates low [10]. Population-based screening for HCC in people with CHB is not practiced in Australia, so HCC cases are commonly diagnosed when symptomatic, or are incidental findings in patients investigated for other medical conditions. HCC screening is practiced by some clinical groups, but fewer than 20% of all HCCs are being detected by HCC surveillance [10].
The aim of this analysis was to compare the costs and health outcomes of two possible strategies of reducing HCC disease burden against current standards of practice (of clinical HCC diagnosis and limited HCC surveillance) in at-risk populations resident in Sydney, NSW. The two interventions assessed were (1) a program of HCC surveillance and (2) a strategy of CHB monitoring and treatment, coupled with HCC surveillance (termed HCC prevention) based upon categories of risk, as detailed below. The two different management strategies were chosen as possible options for modeling a public health intervention to reduce the number of new liver cancer cases in a population that is at significantly higher than average risk of HCC and end-stage liver disease.
Discussion
Compared to current practice, a strategy of HCC surveillance in patients with CHB with minimal CHB treatment over the next 50 years would not reduce the number of cases of advanced liver disease, but would marginally reduce the number of deaths from HCC or liver failure. The program would have a high and unfavourable cost-effectiveness ratio of AU$401,516 per QALY gained, compared to current practice. In contrast, a HCC prevention strategy would significantly reduce the proportion progressing to cirrhosis (by 52%), the number of HCC diagnoses (by 47%) and the number of deaths from HCC or liver failure (by 56%). The strategy would save 0.924 QALYs per patient and have a favourable ICER (AU$12,956 per QALY gained), compared to current practice.
The results of the HCC prevention strategy are driven primarily by the reduced likelihood of progression of liver damage in those with CHB. The HCC prevention strategy offers excellent value for money, when compared to a strategy of HCC surveillance, with an ICER of approximately AU$6733 per QALY gained.
The HCC prevention strategy with treatment individualised according to HBV viral load and ALT level, has a cost-effectiveness comparable to that of the existing population-based cancer screening programs in Australia, as breast cancer screening is estimated to have an incremental cost-effectiveness ratio of approximately AU$10,000 per QALY gained; cervical cancer around AU$45,000 per QALY gained and colorectal cancer about AU$20,000 per QALY gained [31], [32].
While screening for HCC has been advocated as a means of improving survival, [18], [33], [34], both its effectiveness in reducing mortality from liver cancer [35], [36] and its cost-effectiveness [37], [38] are a matter of debate. Programs of twice-yearly surveillance of patients with cirrhosis for HCC using AFP and ultrasound [30], [39], [40], [41], [42] found ICERs ranging from $US 26,689 per QALY gained [39] to $US 73,789 USD per QALY gained [40]. However, direct comparisons of cost-effectiveness are not appropriate, due to substantial differences in the patient populations modelled, the comparators used in the analyses, variations in health system structure, health care funding, and clinical practice, including the use of transplantation. The aetiology of cirrhosis in these studies was either hepatitis C, mixed or not specified; only one study specifically examined the cost-effectiveness of screening for HCC in patients with hepatitis B-related cirrhosis, followed by treatment, which included resection or transplantation [30]. Thompson-Coon and colleagues found that biannual surveillance with AFP and ultrasound in patients with cirrhosis due to HBV had an ICER of 26,800 per QALY gained compared to no surveillance [30], [42]. The ICER of 6 monthly HCC surveillance with AFP and ultrasound was higher for cirrhosis of mixed aetiology (i.e. HBV, HCV and alcoholic liver disease) and lowest in HBV-related cirrhosis [30]. The combination of biannual ultrasound and AFP measurement had an ICER of 60,100 per QALY gained [30], with the cost of liver transplants and care post-transplant the variables with the greatest influence on ICER [30], [42]. A modelled surveillance programs for HCC in Japan found an ICER of $US 29,900 per QALY gained, when assuming no clinical diagnosis of disease (i.e. earlier diagnosis), compared to $US 35,400 per QALY gained, assuming that 20% of HCC were detected clinically [41]. Our modelled cost-effectiveness ratio is substantially higher than these reported results, primarily because previous analyses modelled costs and effects of HCC surveillance in patients with existing cirrhosis. In contrast, our analysis considered a population with CHB, of which fewer than 0.5% had cirrhosis at the start of the program. Therefore, the absolute benefit, in terms of detecting or preventing HCC and subsequent mortality in our modelled population would be substantially lower than in patients with existing cirrhosis. This lower benefit, coupled with similar surveillance costs means that our ICER is substantially higher than those reported for surveillance in patients with cirrhosis. Additionally there are differences in the comparisons made and in clinical practice between our analysis and previous studies, also likely to have influenced the ICER upwards. For example, previous analyses have compared surveillance strategies to no surveillance, whereas our analysis compared enhanced surveillance of CHB patients to 'usual care', in which approximately half the patients who received treatment also had some form of disease surveillance. Our analysis also did not consider transplantation, as there are so few performed in Australia each year.
To our knowledge, this paper is one of the first to examine a strategy of screening and treating CHB with a view to pre-empt malignant transformation, as opposed to diagnosing and treating hepatitis B-related complications, or monitoring patients with existing cirrhosis. Our scenario included HBV viral load in the surveillance algorithm, not previously included in modelled analyses [22], [23], [43], [44]. Hutton et al. examined the cost-effectiveness of combinations of HBV screening, treating and vaccination among Asian and Pacific Islander groups in the US; the strategy closest to the HCC prevention program in our analyses ('screen and treat') demonstrated an incremental cost-effectiveness of $US 36,088 [45]. Although not directly comparable, these results confirm that both a CHB screen and treat strategy, and a screen, treat and ring vaccinate strategy (which had an ICER of $US 39,903 per QALY gained) are associated with favourable ICERs [45].
Study limitations
Some of our assumptions were based on imperfect data, as no consensus exists on the levels for ALT that would mandate treatment. This situation is made even more complex by the fact that ALT levels in patients with CHB may be due to co-existing overweight or obesity, present in >60% of Australian adults [46]. Additionally, the level of surveillance for CHB and HCC in at-risk communities is not well ascertained. However, a study such as this one allows clinicians and policy makers to engage with this topic and permits more detailed scenarios to be modelled in the future.
We excluded transplantation from the model, as of the 152 adult liver transplants carried out in Australia and New Zealand in 2005, only 5% of were performed for hepatitis-B related causes [47], so they would have very little impact on the cost-effectiveness of the modelled program. An analysis of the SEER database in the US found that of nearly 3000 Medicare patients diagnosed with HCC, only 13% received potentially curative treatments, with 0.9% (27 patients) receiving a liver transplant. Attempting to quantify the extent of use of liver transplantation to treat HCC at a population level, another US study found that only one fifth of individuals diagnosed with small tumours (i.e. <5cm) received a transplant [41].
Conclusions
While CHB treatment is expensive, targeting pharmacological management to patients at highest risk (i.e. people with high HBV DNA and elevated ALT levels) provides more substantial gains in health outcomes and offers better value for money, compared to HCC surveillance in individuals with CHB infection. A focused program of screening and treatment of CHB among people born in Asian countries would address the public health challenge of offering a cost-effective treatment to populations most likely to benefit from them, where CHB infection rates are highest. Tailoring such a program to particular high-risk groups would raise medical practitioner awareness for the need to recommend HBV screening, ensure the provision of appropriate health care for those infected and allow improved measurements of the disease burden in these populations. Whether the same benefits are applicable to Caucasian-born populations remains unclear, as differences in the natural history of hepatitis B infection in Caucasian patients may lead to differences in liver-related morbidity and mortality [48].
The burden of CHB and HCC in developed nations is likely to increase significantly in the future, due to the large population at risk, changing patterns of immigration and the long latency for progression from CHB to HCC. Therefore, devising cost-effective programs to reduce the burden of disease should be a key component of health planning and in this context, our study provides valuable information to help guide the clinical implementation of such a program.
Results
Over the 50 years modelled, we estimated that the HCC surveillance strategy would cost on average AU$8479 per patient and result in 12.541 QALYs per patient, compared to AU$2632 and 12.527 QALYs, if managed according to current clinical practice. This represents a very small improvement in health outcomes (0.014 QALYs saved) and a less than 1% reduction in deaths from HCC or liver failure (from 0.338 to 0.336) over current practice; its ICER is of AU$401,516 per QALY gained, compared to current practice.
A HCC prevention strategy would cost, on average, AU$14,600 and result in 13.450 QALYs (representing an additional 0.923 QALYs compared to current practice). This would reduce the proportion of people progressing to cirrhosis by 52% (from 0.419 to 0.200), the number of HCC diagnoses by 47% (from 0.186 to 0.098) and the number of deaths from HCC or liver failure by 56% (from 0.338 to 0.150). The HCC prevention strategy has an ICER of AU$12,956 per QALY gained, compared to current practice and AU$6733 per QALY gained, if compared to the HCC surveillance arm (see Table 3). More than half (58%) of the total program costs for HCC prevention are attributable to antiviral medications, costing AU$17,500 for interferon (where treatment is administered for one year) and AU$4600 per year for entecavir.
Table 3. Average costs and outcomes per patient with chronic hepatitis B treated under current practice, HCC surveillance or HCC prevention algorithms.
With current practice, we estimated that over 50 years, approximately 42% of the cohort would develop cirrhosis and 19% HCC.
Sensitivity analysis
The variables that showed the greatest influence on the ICER were: the proportion of the cohort where pharmacological treatment is indicated, the effectiveness of drug treatment in slowing liver damage and the cost of drug treatment, particularly entecavir (see Table 4). The proportion of patients with high viral load and high ALT and the cost of drugs jointly influence program costs. It is noteworthy that despite changes in these key variables, the ICER for the HCC prevention program remained within a favourable range.
Methods
We used a Markov model of CHB progression to assess the clinical and economic consequences of the two different management strategies, as compared to current clinical practice, in a hypothetical cohort of 10,000 Asian-born adults with CHB, aged 35 years at program entry; gender distribution was 60% males and 40% females.
A health care funder perspective was taken and all future costs and health outcomes discounted at 5% per annum [11]. Patients were followed for 50 years and costs and health outcomes of the intervention and the comparator [cases of HCC averted, deaths averted and quality-adjusted life-years (QALYs) gained] calculated over the follow-up period..
Intervention
The HCC surveillance and HCC prevention interventions were modelled as follows:
Patients eligible for the surveillance programs (aged -35 years and HBsAg positive for >6 months) were stratified and managed according to risk categories, based upon HBV DNA and ALT levels (Fig. 1). Patients with a low HBV viral load (defined as <20,000IU/ml for age <50 years and <2000IU/ml for age >50 years) and ALT levels <1.5 X the upper limit of normal (ULN) (reference range <40IU/L) received routine CHB monitoring, consisting of six-monthly primary care follow-up and repeat testing for HBsAg, HBeAg, viral load and ALT (or routine care). Patients with a high HBV viral load (defined as >20,000 IU for age <50 years and >2000IU/ml for age -50 years) and ALT <1.5 times ULN received the same CHB monitoring at primary care level, combined with HCC surveillance, consisting of biannual liver ultrasound and alpha fetoprotein (AFP) level measurements (or enhanced HCC surveillance). Patients with a high HBV viral load (see above) and elevated ALT levels (-1.5 ULN) were referred for specialist management, including routine CHB monitoring, HCC surveillance, biopsy assessment of liver damage and pharmacologic treatment of CHB (or HCC prevention).
Comparator
The comparator was the current clinical practice, of limited treatment of CHB and some HCC surveillance, with most patients receiving neither. [12], [13] Based on government medication prescription data, we estimated that approximately 2% of CHB cases receive pharmacological treatment under current practice [13]. We assumed that people who received neither CHB treatment, nor HCC surveillance would progress through the disease process in a manner similar to the natural history of hepatitis B. The small proportion of patients receiving treatment in this group was modelled by taking into account a CHB treatment benefit.
Markov model
A Markov model with an annual cycle length was used to model the progression of patients through a series of CHB health states: CHB without cirrhosis; CHB with cirrhosis; CHB with liver failure; CHB and HCC; spontaneous HBsAg clearance and death due to CHB related causes (liver failure or HCC), or death from other causes (see Fig. 2a and b). Management decisions in the intervention groups were guided by HBV viral load and liver function.
Treatment cut-offs regarding viral load were informed by clinical guidelines suggesting that threshold levels of HBV DNA should factor in whether patients are HBeAg positive (with treatment recommended for viral loads >105copies/ml, or approximately 20,000IU/ml) or HBeAg negative (with treatment suggested for VL>104copies/ml, or approximately 2000IU/ml) [14], [15], [16]..
The lower age limit (age >35 years) was informed by the first large study examining spontaneous HBeAg sero-conversion, which occurred at a median age of 34.5 years [17]. As a significant proportion of people aged >50 years are at risk of developing cirrhosis [15], they are also at an increased risk of hepatic decompensation and liver cancer. Hence, for this group, a lower viral load (VL>2000IU/ml) cut-off was used.
The model structure and variables were informed by the available literature on the epidemiology and natural history of hepatitis-B related HCC [4], [18], [19], [20], [21] and previously published models of CHB and its treatment [22], [23]. In accordance with current guidelines, we took into consideration both viral load, and ALT elevations for the initiation of therapy. Where published estimates were unavailable, expert estimates were used. All modelling was implemented in Microsoft Excel.
General assumptions
In the HCC prevention program, all patients with high HBV viral loads and elevated ALT levels were assumed to receive pharmacological treatment. As general guidelines managing CHB generally avoid recommending particular drugs [16], we chose to model pegylated-interferon and entecavir, based upon local patterns of use and taking into account the National Institute of Health and Clinical Excellence guidelines (NICE 2006), which addressed health economic issues related to treatment [16]. The modelling scenario did not include liver biopsy as part of the decision to treat or not to treat, which was made based upon ALT, viral load levels and age.
We estimated that 30% of people eligible for treatment had interferon weekly for 12 months as first-line treatment and assumed that 30% of this group would achieve sustained viral suppression and have no subsequent treatment. The remainder received lifelong entecavir therapy (0.5mg daily) from year 2 onwards. We estimated that 70% of people eligible for treatment received entecavir, with 20% assumed to seroconvert during the first year of treatment (and receiving no further treatment); the remainder continued entecavir therapy indefinitely. Regular follow-up (every 6 months) was incorporated into the program, to allow for changes in clinical status, ensure optimal treatment monitoring and to detect HCC at an early stage.
We assumed that under current practice, approximately 50% of people with CHB on medical treatment also receive some form of disease surveillance (with HBsAg, HBeAg, ALT and AFP testing and ultrasound examination). Based on published literature, we assumed that 89.5% of the intervention group (in whom HCC was detected by surveillance) were eligible for HCC resection, compared to 11.1% in the comparator group [24]. Patients not eligible for resection received chemoembolisation and /or radiofrequency ablation. In order to simplify modeling and to reflect the nature of the data used, we did not model a distinct survival rate specific for those who had resection, RFA, or other treatment. The modeled survival rates represent the rates of a blended group of patients, in which some receive HCC resection, some RFA, and others receive other (or no) treatment.
Treatment effectiveness was modelled by a reduction in the probability of progression to a more advanced disease state, e.g. from CHB to cirrhosis and from cirrhosis to liver failure or HCC. Probabilities were based upon published data and expert estimates (see Table 1). The age-specific probability of death from causes other than CHB/HCC was based upon Australian Bureau of Statistics (ABS) estimates [25].
Costs
Costs were calculated from the perspective of a central health care funder and reported in 2006 Australian dollars (AU$). Health care utilisation related to CHB and its sequelae included: initial screening for CHB, diagnostic testing, physician and specialist consultations, six-monthly surveillance regimens and treatment of CHB and HCC. The model also included estimates of costs for program administration, recruitment and retention..
Where available, unit costs of resources were based on the Medicare Benefits Schedule (MBS) [26], or Pharmaceutical Benefits Schedule (PBS) [27], or on the National Hospital Cost Data Collection (NHCDC) for teaching hospitals [28]. Costs not available from these sources (HBV DNA testing, chemoembolisation and liver resection) were based upon data provided by one hospital (see Table 2 and Appendix 1). The costs of liver transplantation (where utilisation is capped by supply of donor livers) and palliative care treatment were not included, nor were indirect costs.
Outcomes
The main health outcome of interest was quality adjusted life years (QALYs), a multidimensional health outcome that incorporates both survival and quality of life. Age-specific quality of life weights recommended by Shepherd were used for CHB related health states [29], [30]. We assumed no decrement in quality of life for those who clear CHB. Other clinical outcomes included progression to cirrhosis, liver failure and HCC and CHB-related deaths from HCC or liver failure.
We also calculated incremental cost-effectiveness ratios (ICERs), to quantify the additional costs that would be incurred in order to gain and additional unit of health outcome-in this case, QALYs.
ICERs were calculated as follows:

Sensitivity analysis
We performed a series of one-way sensitivity analyses on a range of variables including: the proportion of patients with high viral load and high ALT, drug prices and main treatment effectiveness parameters, including the relative risk of progression from CHB to cirrhosis, from cirrhosis to liver failure and from liver failure to death. Additionally, two-way sensitivity analyses were conducted by varying simultaneously the relative risk of progression from CHB to cirrhosis and the relative risk of progression from cirrhosis to liver failure. Results of variables influencing the model are reported in Table 4.
|
|
| |
| |
|
|
|