| |
Benefits and risks of interferon therapy for hepatitis B Meeting Report Review Robert Perillo
|
| |
| |
Hepatology Apr 27 2009 Epub ahead of print
Robert Perrillo *
Hepatology Division, Baylor University Medical Center; Dallas, TX
Potential conflict of interest: Dr. Perrillo is on the Speakers Bureau for Bristol-Myers Squibb and Roche Pharmaceuticals. He currently receives research support from Bristol-Myers Squibb and Pharmasett, Inc.
Abstract
Alpha interferon is the only licensed drug for hepatitis B with immunomodulatory as well as viral inhibitory properties. Potential advantages of interferon compared to nucleoside analogs include a lack of drug resistance, a finite and defined treatment course, and a higher likelihood for hepatitis B surface antigen (HBsAg) clearance. Approximately 30% of hepatitis B e antigen (HBeAg)-positive and 40% of HBeAg-negative cases have a sustained virological response (when defined as HBeAg seroconversion and/or hepatitis B virus (HBV) DNA levels below 20,000 copies/mL, respectively) 6 months after completion of a 48-week course of peginterferon alfa-2a These responses remain durable in 80% and 50% of cases, respectively, when evaluated several years later. Recent studies have shown that changes in HBsAg and HBeAg concentration during treatment predict sustained virological response and serial monitoring of HBsAg is helpful in predicting HBsAg clearance. HBeAg-positive patients with genotype A have higher rates of HBeAg and HBsAg clearance, whereas HBeAg-negative patients with genotype D have the lowest rate of response to interferon therapy. Long-term follow-up of virological responders to either standard alpha interferon or peginterferon has demonstrated a progressive increase in the rate of HBsAg clearance, particularly in patients who were initially HBeAg-positive. Future studies need to address if specific virological benchmarks during therapy can be used to tailor treatment duration.
Conclusion: Peginterferon alfa has a place as first-line therapy of hepatitis B in patients who are carefully selected on the basis of pretreatment serum HBV DNA and aminotransferase levels, safety considerations, and viral genotype.
Introduction
Chronic hepatitis B is caused by a persistent infection with the hepatitis B virus (HBV), but the level of disease activity as well as the frequency of response to antiviral therapy are strongly influenced by host immune responses.[1][2] Alpha interferon is the only licensed antiviral in the United States that has both immunomodulatory and antiviral activity against HBV. Currently, interferon is infrequently used for hepatitis B, having been replaced by the oral nucleoside analogs, which have potent activity against HBV and fewer side effects.[3] This article will focus on the efficacy and safety of peginterferon, with an emphasis on data that have emerged in the last few years (Table 1). These data support the conclusion that interferon has an important therapeutic role in selected patients.

Abbreviations
ALT, alanine aminotransferase; cccDNA, covalently closed circular DNA; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; HBV, hepatitis B virus; PCR, polymerase chain reaction.
Mechanisms of Action
Alpha interferon acts by inducing an antiviral state in cells, through engagement of cell surface receptors and subsequent activation of pathways that lead to increased expression of intracellular genes that cause an increase in breakdown in viral RNAs and protection against viral injury.[4] Alpha interferon also stimulates cell-mediated immune responses which target infected hepatocytes leading to a decrease in cells that harbor the intrahepatic, HBV covalently closed circular DNA (cccDNA) molecules responsible for persistence of HBV infection. Early clinical studies with standard alpha interferon emphasized the immunoregulatory properties of this drug.[5][6] By contrast, clinical trials of the more potent pegylated forms of alpha interferon have tended to emphasize its antiviral activity.[7-9] There are multiple indirect lines of evidence, however, that immune regulation is important to its effectiveness in both hepatitis B e antigen (HBeAg)-positive and HBeAg-negative chronic hepatitis B. For example, loss of HBeAg and seroconversion to antibody (anti-HBe) with interferon treatment typically occurs weeks to months after the end of treatment, a feature which is consistent with prolonged immunological control over HBV replication.[10] In addition, host-derived flares of disease characterized by marked increases in serum alanine aminotransferase (ALT) levels occur during treatment in 25%-40% of HBeAg-positive patients, often in association with clearance of HBeAg. These flares have been associated with higher rates of HBeAg seroconversion, and their magnitude appears to be important in overcoming high level viremia.[9][11][12]
Review of Clinical Trials with Peginterferon Alfa
There has been a resurgence of interest in interferon therapy over the past 5 years, largely based on results of large clinical trials demonstrating that peginterferon has more potent antiviral activity than standard alpha interferon and, in contrast to nucleoside analogs, does not result in antiviral resistance and can be given for a finite period rather than indefinitely. Thus, when compared to standard interferon alfa-2a in a dose of 4.5 million units three times weekly, peginterferon in a dose of 180 ug once weekly for 12 months resulted in a greater decline in HBV DNA levels and a higher rate of HBeAg seroconversion (33% versus 25%).[7]
Three large multicenter trials of peginterferon therapy have been published, two in HBeAg-positive and one in HBeAg-negative chronic hepatitis B.[8][9][13] Each study included treatment arms in which peginterferon was used alone or in combination with lamivudine. Two studies used peginterferon alfa-2a and one used peginterferon alfa-2b.
In a multinational European study, peginterferon alfa-2b was given in a dose of 100 ug weekly for 32 weeks followed by 50 ug weekly until completion of 52 weeks of treatment with or without lamivudine (100 mg daily) in 266 patients who were HBeAg-positive.[8] Loss of HBeAg by 6 months after treatment occurred in similar proportions of patients receiving monotherapy as combination therapy (36% versus 35%: P = 0.91) as did loss of HBsAg (7% versus 7%). Suppression of HBV DNA levels and loss of HBeAg were greater on combination therapy than monotherapy, but relapse rates were higher in the group that received lamivudine so that sustained responses 6 months after stopping treatment were equivalent. A comparison group receiving lamivudine alone was not included.
In a second larger, multicenter trial, a total of 814 patients with HBeAg-positive chronic hepatitis B were given either peginterferon alfa-2a alone (180 g once weekly), lamivudine alone (100 mg daily), or the combination for 48 weeks.[9] Again, HBV DNA suppression was greater in patients receiving combination therapy than in those receiving either peginterferon or lamivudine monotherapy. However, rates of HBeAg seroconversion 6 months after stopping therapy was greater with peginterferon than lamivudine monotherapy (32% versus 19%) and was no higher with combination therapy (27%). Loss of HBsAg occurred in 16 of 542 patients (3%) who received peginterferon (alone or with lamivudine) but in none of 272 patients receiving lamivudine alone (P = 0.004).
Finally, in the third large, multicenter trial, patients with HBeAg-negative hepatitis B were treated with peginterferon alfa-2a alone (180 ug once weekly), lamivudine alone (10 mg daily), or the combination for 48 weeks.[13] Six months after stopping therapy, the percentage of patients with normal ALT values or HBV DNA levels below 20,000 copies/mL was significantly higher with peginterferon monotherapy (59% and 43%, respectively) than with 48 weeks of lamivudine monotherapy (44% and 29%, respectively). Again, the addition of lamivudine to peginterferon therapy did not appear to increase the response rates even though there was greater HBV DNA suppression on combination therapy. Furthermore, 3% (12/356) of patients who received peginterferon but 0% of patients (0/181) who received lamivudine alone became HBsAg-negative.
These three studies showed that a 1-year course of peginterferon induced HBeAg seroconversion in about one-third of HBeAg-positive patients and induced a lasting biochemical and virological response in about 40% of HBeAg-negative patients. Furthermore, therapy with peginterferon led to loss of HBsAg in a small proportion of patients, an outcome not seen with a 1-year course of lamivudine. Adding lamivudine to peginterferon did not raise the rate of sustained responses. These results suggested that a trial of a 1-year course of peginterferon might be appropriate in selected patients with chronic hepatitis B, before embarking on long-term suppressive therapy with a nucleoside analog.
HBsAg Loss and Seroconversion
HBsAg loss with or without seroconversion to antibody to HBsAg (anti-HBs) is a clinically important event that is associated with improved survival and a lower risk for hepatocellular carcinoma in patients with cirrhosis.[14] HBsAg clearance represents the closest point to a clinical cure and clearly happens earlier in patients treated with interferon compared to those treated with nucleoside analogs. Loss of HBsAg after antiviral therapy was first reported more than 20 years ago in clinical trials of standard alpha interferon for HBeAg-positive hepatitis B,[6][15] and it was reported to occur in 3%-7% of patients in the three large clinical trials of peginterferon.[8][9][13] These multicenter trials of peginterferon alfa-2a convincingly demonstrated that HBsAg loss and seroconversion was confined to patients receiving peginterferon, either alone or in combination.[9][13]
During long-term follow-up, most studies of standard alpha interferon or pegylated interferon have described a substantial increase in the proportion of virological responders with HBsAg clearance, particularly in those treated for HBeAg-positive disease[16-23] (Table 2). A broad range of HBsAg clearance rates has been reported in follow-up studies of HBeAg-positive patients who were successfully treated with interferon, varying from 30% to 71% of initial virological responders. This may be due to varying lengths of follow-up, different treatment regimens, and different distributions of viral genotypes.
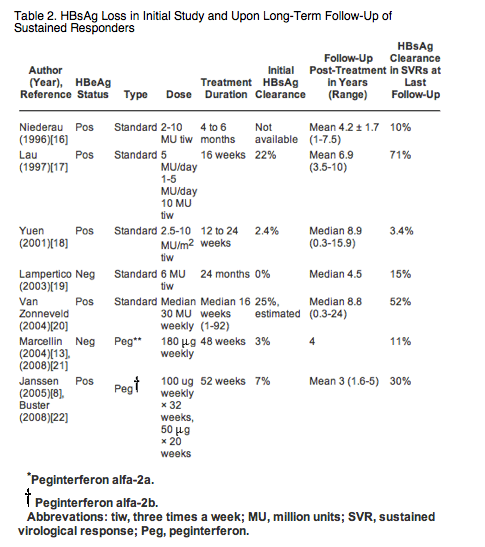
HBsAg clearance has been reported to occur less commonly in HBeAg-negative hepatitis, reflecting the generally lower rates of sustained virological response with this disorder and perhaps differences in immune features and duration of infection among patients who are HBeAg-negative compared to those who are HBeAg-positive[19][21][24] (Table 2). Prolongation of treatment beyond 48 weeks may be more effective in achieving sustained virological response and HBsAg clearance, but this has not been systematically studied. In one long-term follow-up study of HBeAg-negative patients treated with standard alpha interferon for 24 months, HBsAg clearance occurred in 15% of patients.[19]
Effects of Antiviral Therapy on Intrahepatic HBV cccDNA
Few studies have addressed quantification of HBV cccDNA in liver tissue of interferon-treated patients because a sensitive and reliable means of quantification was not available until recently. Using real-time polymerase chain reaction (PCR), however, several studies have demonstrated that HBV cccDNA persists in the liver of the majority of patients during treatment.[25] The serum concentration of HBsAg has been shown to correlate to some extent with levels of intrahepatic HBV cccDNA and total HBV DNA.[26] The decline of intrahepatic HBV cccDNA has been shown to parallel that of serum HBsAg concentration in patients treated with peginterferon in combination with lamivudine.[26] In one study, a -0.80 log copies/mL decline in intrahepatic HBV cccDNA was shown to predict sustained HBeAg seroconversion.[27] These studies involve limited numbers of patients and samples. Thus, further studies that evaluate changes in serum HBsAg level and the relationship of this finding to changes in intrahepatic HBV cccDNA are necessary to clarify to what extent a decline in serum HBsAg concentration reflects the loss of intrahepatic cccDNA.
Baseline Predictors of Response
Early clinical trials of standard alpha interferon in HBeAg-positive patients demonstrated baseline HBV DNA and ALT levels to be predictive of a response to 16 weeks of treatment.[6][28] Multivariate analysis from the registration trial for peginterferon alfa-2a has confirmed that higher baseline ALT levels, lower baseline HBV DNA concentrations (109 copies/mL), and lower concentrations of HBeAg were predictive of HBeAg seroconversion with treatment.[29] In a study using peginterferon alfa-2b in combination with lamivudine, patients with lower baseline intrahepatic HBV cccDNA levels and lower serum HBsAg levels, but not necessarily lower serum HBV DNA levels, were more likely to achieve a sustained HBeAg seroconversion.[26] In addition to this, recent studies in HBeAg-positive disease also have demonstrated a strong correlation between genotype A and higher rates of both HBeAg and HBsAg loss.[8][22][30]
Baseline predictors of sustained virological response are less well-defined for patients with HBeAg-negative chronic hepatitis B. Logistic regression analyses of baseline factors in the large clinical trial of peginterferon alfa-2a in HBeAg-negative chronic hepatitis B found younger age, female sex, higher baseline ALT levels, lower baseline HBV DNA concentrations and HBV genotype to be predictive of a combined virological and biochemical response.[31]
Response to Interferon According to Viral Genotype
Viral genotype is an important factor that is associated with both qualitative as well as quantitative responses to interferon. Genotype may also influence the time to virological response. Early studies with standard alpha interferon found that patients with genotype A responded more frequently than patients with genotypes B, C, and D.[32-34] These studies, however, were limited by small sample size and predominance of genotype A in the populations studied (Northern European). Large multicenter trials of peginterferon alfa with enrollments from Europe, Asia, and North America provided the opportunity to study the relationship of genotype to response in better detail. In patients treated with peginterferon alfa-2b, a statistically relevant association was found between viral genotype and sustained HBeAg loss.[8] When all interferon-treated patients were entered into the analysis (with or without lamivudine), the highest rate of HBeAg clearance at the end of follow-up occurred in patients infected with genotype A (47%), followed by genotype B (44%), C (28%), and D (25%). Further analyses of the same study population demonstrated that HBsAg clearance was also closely linked to viral genotype, being highest in genotype A (14%) compared to B (9%), C (3%), and D (2%).[35] Recently, this cohort was re-evaluated an average of 3.0 years after the end of the initial study. Among patients with genotype A who cleared HBeAg in the initial study, 96% had sustained HBeAg-negativity and 58% had became HBsAg-negative.[22] In contrast, the same endpoints were achieved in 86% and 14%, respectively, of patients with genotype B, 67% and 0% of patients with genotype C, and 76% and 6% of those with genotype D. These data indicate that durable HBeAg loss after interferon therapy occurs most frequently in patients with genotypes A and B, and this is associated with a greater chance for HBsAg clearance upon prolonged follow-up.
The data evaluating the relationship between genotype and virological response in HBeAg-positive patients entering the registration trial for peginterferon alfa-2a were not as robust, because 93% of those enrolled had non-A genotypes.[9] Yet even with the lower numbers, 52% of patients with HBV genotype A given peginterferon monotherapy had an HBeAg seroconversion compared to 32% for the interferon-treated patients as a whole.
The relationship between response to peginterferon and genotype is less well-defined in HBeAg-negative hepatitis B. An analysis of a large patient population treated with peginterferon alfa-2a also indicated that individuals infected with genotypes B or C had a higher chance of response than those with genotype D (P < 0.001).[31] Serial measurement of HBsAg concentration in these same patients revealed the greatest decline in patients with genotype A and the least in patients with genotype D (end-of-follow-up reduction in HBsAg was 1.05, 0.60, and 0.36 log IU/mL for A, B/C, and D, respectively).[36]
A systematic review of 20 interferon trials up to 2007 found HBV genotypes to be informative concerning responses to interferon in all patients with genotype A versus D and in HBeAg-positive patients with genotype B versus C.[37] In a recent pooled analysis of more than 1200 patients treated with either standard alpha interferon or peginterferon, the rates of sustained virological response for HBeAg-positive and HBeAg-negative patients were 36% versus 34%, respectively, for genotype A, 21% versus 32% for genotype B, 19% versus 50% for genotype C, and 15% versus 21% for genotype D.[30] Taken together, the data provide compelling support for the fact that genotype A is the most treatment-responsive genotype in HBeAg-positive hepatitis B. Genotype A is relatively uncommon in patients with HBeAg-negative cases, but non-D genotypes, particularly C, appear to have higher rates of sustained virological response in this form of chronic hepatitis B. The reasons for the different rates of virological response according to genotype remain unclear but may relate to changes in viral sequences during interferon therapy that affect host immune responses.[38][39]
On-Treatment Indicators of Response
A number of recent studies have suggested that achieving specific virological benchmarks during treatment can be helpful in predicting a sustained virological response and HBsAg clearance. One of the more unique and interesting associations has been with serum HBsAg concentration during treatment. The data from several small clinical trials that included serial monitoring of serum HBsAg concentration provide some evidence that a decline in HBsAg concentration can be helpful in predicting response.[40-42] The ability to predict loss of HBsAg would be especially helpful in HBeAg-negative hepatitis, because reliable treatment endpoints that correlate with a durable response have not been established. In a study of 48 patients with HBeAg-negative hepatitis B, a 1-log decline in HBsAg concentration at treatment week 24 of peginterferon alfa-2a had a 97% negative predictive value for a sustained virological response (the latter defined as undetectable HBV DNA by PCR 24 weeks after treatment). Three patients who lost HBsAg had even greater declines in HBsAg concentration by week 24.[42] In 356 HBeAg-negative patients treated with peginterferon alfa-2a, alone or in combination with lamivudine, a major reduction in HBsAg concentration was found to also predict which patients would have HBV DNA levels of fewer than 400 copies/mL 6 months after treatment.[43] End-of-treatment HBsAg levels of less than 10 IU/mL or an on-treatment reduction in HBsAg levels of > 1 log10 IU/mL were significantly associated with durable HBsAg clearance 3 years after treatment.[43] In contrast, HBV DNA levels at the end of treatment were not predictive of a sustained response, and HBsAg levels remained essentially unchanged in the group treated with lamivudine alone.
Retrospective analysis of clinical trial data has also permitted examination of whether a decline in markers of viral replication at key treatment intervals can be used to predict virological response. In a study of 57 HBeAg-positive patients treated with a combination of peginterferon alfa-2b and lamivudine, the likelihood ratio for sustained virological response was highest (10.35) for patients with an HBV DNA level < 10,000 copies/mL at week 8.[44] Baseline HBV DNA levels did not significantly differ between responders and nonresponders. Serial measurement of HBeAg concentration also has been used to predict sustained HBeAg seroconversion in 271 patients treated for 48 weeks with peginterferon alfa-2a.[45] In that study, levels of HBeAg (measured in Paul Ehrlich Institute units [PEIU]/mL) consistently decreased during treatment in patients who subsequently underwent HBeAg seroconversion; only 4% of patients with HBeAg levels 100 PEIU/mL at week 24 of treatment achieved HBeAg seroconversion. The negative predictive value was 96% at 24 weeks, exceeding that of an arbitrary cutoff of 9 log10 HBV DNA level (86%) at the same interval. Recently, early loss of HBeAg before 32 weeks of treatment with peginterferon alfa-2b was found to be highly predictive of HBsAg clearance upon long-term follow-up.[46] Use of these on-treatment predictive factors has not been evaluated prospectively.
Increases in ALT levels during therapy have also been shown to be predictive of virological response. In the peginterferon alfa-2a registration trial, HBeAg seroconversion occurred more frequently in patients with on-treatment flares in ALT values (defined as ALT rises above 5 times baseline values).[9][47] Although flares of this magnitude occurred in only 5% of patients treated with peginterferon monotherapy, HBeAg seroconversion occurred more frequently in this subgroup (43%) than in patients without flares. On-treatment rises in ALT values to greater than 10 times the upper limit of the normal range (ULN) occurred in 18% of patients treated with peginterferon monotherapy, and the rate of HBeAg seroconversion (42%) was significantly greater than that in patients in whom ALT never exceeded five times the ULN (26%).[47] In a large study using peginterferon alfa-2b, host immunity-mediated flares of ALT occurred in 36% of patients, and this was associated with HBeAg loss in 58% of patients (P = 0.008).[11]
Durability of Response
Long-term follow-up studies using standard alpha interferon or peginterferon have convincingly shown that HBeAg loss and seroconversion is sustained in at least 80% of patients followed for a mean of 3-6 years after the end of treatment.[17][20][22] Several studies using standard alpha interferon have demonstrated improved histology, better survival, or reduced risk of developing hepatocellular carcinoma in sustained virological responders.[16][17][20] Less favorable long-term responses have been reported in Asian patients who have undergone interferon-induced HBeAg seroconversion.[18]
A higher rate of relapse has been associated with HBeAg-negative hepatitis after standard alpha interferon or peginterferon treatment.[19][21][24] A large cohort of patients treated with peginterferon alfa-2a (230/356) has been followed for an average of 4 years after treatment.[21] In the initial study, sustained virologic response (defined as < 20,000 copies/mL of serum HBV DNA at the 6-month post-treatment interval) occurred in 43% of patients. However, this response was durable in only 25% up to slightly more than half of the initial responders at the 4-year interval. A total of 19% and 17% of patients, respectively, were negative for HBV DNA as measured by PCR at these same intervals, suggesting that greater viral suppression is associated with a higher likelihood of a durable response.
Safety Considerations
As with standard alpha interferon, treatment with peginterferon is associated with many troublesome and occasionally with serious, or even life-threatening, side effects. Dose discontinuation has been reported in 6%-9% and dose modification in 31%-47% of patients treated in the peginterferon alfa registration trials for hepatitis B.[8][9][13] The most frequent causes of dose adjustments were laboratory abnormalities such as thrombocytopenia or leukopenia. These side effects are particularly common in patients with advanced disease and hypersplenism.
Interferon is contraindicated in patients with decompensated cirrhosis. Severe exacerbations of disease with immediate and marked increases in serum ALT levels are common and can have devastating clinical consequences in these patients, even when decompensation is mild and reduced doses of interferon are given.[48][49] A high rate of serious bacterial infections also has been reported.[48] Accordingly, even marginally normal values for serum albumin, serum bilirubin, or prothrombin time prior to treatment should be taken as an indication of limited hepatic reserve, and interferon therapy should be avoided in such patients. The potential serious adverse consequences of interferon therapy in patients with cirrhosis has led to the recommendation in the practice guidelines of the American Association for the Study of Liver Diseases that nucleoside analog therapy is preferred in patients with cirrhosis.[50] This recommendation has been challenged for well-compensated cirrhosis in a study addressing the side effect profile of peginterferon alfa-2b in 70 patients with advanced fibrosis (Ishak fibrosis score 4-6) as compared to 169 patients without advanced fibrosis.[51] Fatigue, anorexia, and thrombocytopenia occurred more often in the group with advanced fibrosis (P < 0.01), but need for dose reduction or early discontinuation of therapy were comparable between the two groups (33% versus 34% and 11% versus 8%, respectively). There was a trend for more acute flares in ALT levels among patients with advanced fibrosis (20% versus 11%, not significant) but none were associated with clinical decompensation. Similarly, in the large registration trials of peginterferon alfa-2a, ALT flares (> 5 times ULN) occurred, but none of them resulted in hepatic decompensation despite the fact that 18% of HBeAg-positive and 31% of HBeAg-negative patients treated with peginterferon monotherapy had advanced fibrosis or cirrhosis.[9][13][52]
Several studies have suggested that side effects of peginterferon are less frequent and less severe in patients with chronic hepatitis B than with chronic hepatitis C. Although there have been no direct, side-by-side comparisons of side effects in these two populations, pooled data from two phase 3 trials of peginterferon alfa-2a in hepatitis B and fivephase 3 trials in hepatitis C support this difference in frequency and severity of side effects.[52] The overall incidence of serious adverse events was less in patients with hepatitis B (4%-5%) than in those with hepatitis C (7%-16%), and fewer drug withdrawals were reported (6%-8% in hepatitis B versus 17%-33% in hepatitis C) despite similar doses and durations of therapy. Depression was also less frequent in patients with hepatitis B (4%) than those with hepatitis C (22%) (P < 0.001). This difference was independent of race and ethnicity and was in agreement with observations that patients with hepatitis B are less likely to suffer major episodes of depression in their lifetime than patients with hepatitis C.[53] This pooled analysis also demonstrated better physical component scores on the SF-36 questionnaire in the hepatitis B group at baseline and during treatment.
Recommendations for use of peginterferon as first-line therapy have been proposed that factor in age and comorbid illnesses, suggesting that therapy be most applicable to younger patients without other major disease conditions.[54][55] However, the large registration trials of peginterferon for chronic hepatitis B did not report dose modification and early discontinuation by age group, past history of depression, and presence of other medical illnesses. Nevertheless, peginterferon therapy is probably most appropriate in younger patients and patients without other major illnesses (such as coronary or cerebral vascular disease, renal dysfunction, poorly controlled autoimmune diseases), and these factors need to be taken into account before making a final decision to use interferon therapy.
Conclusions
Peginterferon alfa has been proven to be effective in the treatment of hepatitis B. The major advantages to using interferon are its ability to provide a high quality of response, including serum HBsAg clearance and a relatively short, defined interval of treatment. However, interferon is now used infrequently largely because of its multiple side effects and inconvenience of administration. The data presented in this review lend support to its use as a viable first-line treatment option for both HBeAg-positive and HBeAg-negative hepatitis B. Also, while not discussed, it is currently the only effective therapeutic option for patients with hepatitis delta virus infection.
The importance of appropriate selection of candidates for interferon therapy can not be overemphasized. Before making a final decision, the clinician should always consider pretreatment HBV DNA and ALT levels as well as projected safety. Once these have been determined, the patient should be genotyped to be able to more accurately access the likelihood of response. HBeAg-positive patients infected with genotype A are more likely to have a lasting virologic response and HBsAg clearance. Sustained virological responses and HBsAg clearance can occur in patients with HBeAg-negative hepatitis but are least likely in patients with genotype D.
Needs for Future Research
There are several key clinical areas where more research with peginterferon is needed (Table 3). It is unclear whether the licensed 48-week course of treatment is appropriate for all patients with chronic hepatitis B. Looking at comparable patient populations, 24 weeks of peginterferon alfa-2a resulted in rates of HBeAg seroconversion that were comparable to 48 weeks of therapy (33% and 32%, respectively).[7][9] Thus, an appropriately powered clinical trial comparing 24 weeks to 48 weeks of therapy with stratification according to genotype, baseline ALT, and HBV DNA would provide clinically useful information. HBeAg-positive patients with genotype A, and to a lesser extent those with genotype B, appear to have higher rates of HBeAg loss and HBsAg clearance. Thus, a trial of 24 versus 48 weeks of peginterferon alfa seems particularly relevant for patients with these more favorable genotypes.

By contrast, HBeAg-negative chronic hepatitis B remains a much more difficult disease to treat, even with 48 weeks of peginterferon. Improved rates of sustained virological response have been reported when this disorder is treated with standard alpha interferon for longer than 24 weeks, and several small studies with standard alpha interferon or peginterferon support a role for prolongation of treatment beyond 48 weeks.[19][41] One difficulty, however, is that the durability of responses with longer courses of therapy have not been carefully defined. Accordingly, it appears reasonable to study various durations of peginterferon therapy in HBeAg-negative hepatitis. Such a trial would be particularly relevant in patients with the less favorable genotype D.
Prospective studies are also needed to examine whether achieving certain virological benchmarks during treatment can lead to tailoring the duration of interferon treatment. Data derived from retrospective analysis of large clinical trials suggest that treatment of hepatitis B could ultimately become a situation where the duration of interferon treatment is dictated by the response to therapy in a manner similar to hepatitis C. Future studies should be directed to answering the question of whether serial assessment of HBsAg or HBeAg concentration accurately identifies patients who are unlikely to have a sustained virological response despite continued treatment. Use of an early stopping rule based on virological response would mitigate adverse effects of prolonged interferon and also lead to earlier initiation of alternative treatment. Conversely, serial monitoring of HBsAg concentration could potentially identify patients who would be more likely to derive lasting benefit from extending the duration of treatment. This would be most relevant for HBeAg-negative hepatitis, a condition for which HBsAg clearance is probably the only reliable therapeutic endpoint.
Although combined treatment with peginterferon and lamivudine has not proven useful in enhancing sustained virological response, large multicenter studies have consistently shown greater on-treatment viral suppression with combination therapy when compared to peginterferon monotherapy or nucleoside analog monotherapy.[8][9][13] Because the two types of drugs work through independent mechanisms, further studies using peginterferon in conjunction with a high-genetic-barrier nucleoside analog with continuation of the latter beyond the period of interferon administration appear warranted.
Finally, it is worth emphasizing that in chronic hepatitis B the extent of hepatocyte injury and the likelihood of achieving a virologic response to antiviral therapy are primarily affected by the immune status of the host. Thus, the development and validation of practical ways of assessing the host immune response to HBV before, during, and after treatment would help advance understanding of how to best use interferon and elucidate the relative importance of its immunological and antiviral activities. This understanding might allow assessment of 'immunologic readiness' for a response to interferon therapy, which in turn would lead to better patient selection, and provide a direct way of monitoring the degree of immunological activation during therapy.
|
|
| |
| |
|
|
|