| |
Peginterferon Alfa-2a and Ribavirin in Latino and
Non-Latino Whites with Hepatitis C: LATINO Study NEJM
|
| |
| |
NEJM Jan 15 2009__
Maribel Rodriguez-Torres, M.D., Lennox J. Jeffers, M.D., Muhammad Y. Sheikh, M.D., Lorenzo Rossaro, M.D., Victor Ankoma-Sey, M.D., Fayez M. Hamzeh, M.D., Ph.D., Paul Martin, M.D., for the Latino Study Group
From Fundacion de Investigacion de Diego, San Juan, and the Ponce School of Medicine, Ponce - both in Puerto Rico (M.R.-T.); the Miller School of Medicine, University of Miami, Miami (L.J.J., P.M.); the University of California, San Francisco, Fresno Medical Education Program, Community Regional Medical Center, Fresno (M.Y.S.); the University of California- Davis Medical Center, Sacramento (L.R.); the University of Texas Medical School and St. Luke's Episcopal Hospital Center for Liver Disease, Houston (V.A.-S.); and Roche, Nutley, NJ (F.M.H., P.M.).
ABSTRACT
Background- Race has been shown to be a factor in the response to therapy for hepatitis C virus (HCV) infection, and limited data suggest that ethnic group may be as well; however, Latinos and other ethnic subpopulations have been underrepresented in clinical trials. We evaluated the effect of Latino ethnic background on the response to treatment with peginterferon alfa-2a and ribavirin in patients infected with HCV genotype 1 who had not been treated previously.
Methods- In a multicenter, open-label, nonrandomized, prospective study, 269 Latino and 300 non-Latino whites with HCV infection received peginterferon alfa-2a, at a dose of 180 μg per week, and ribavirin, at a dose of 1000 or 1200 mg per day, for 48 weeks, and were followed through 72 weeks. The primary end point was a sustained virologic response. We enrolled Latinos whose parents and grandparents spoke Spanish as their primary language; nonwhite Latinos were excluded.
Results- Baseline characteristics were similar in the Latino and non-Latino groups, although higher proportions of Latino patients were 40 years of age or younger, had a body-mass index (BMI, the weight in kilograms divided by the square of the height in meters) of more than 27 or more than 30, and had cirrhosis. The rate of sustained virologic response was higher among non-Latino whites than among Latinos (49% vs. 34%, P<0.001). The absence of HCV RNA in serum was more frequent in non-Latino whites at week 4 (P=0.045) and throughout the treatment period (P<0.001 for all other comparisons). Latino or non-Latino background was an independent predictor of the rate of sustained virologic response in an analysis adjusted for baseline differences in BMI, cirrhosis, and other characteristics. Adherence to treatment did not differ significantly between the two groups. The numbers of patients with adverse events and dose modifications were similar in the two groups, but fewer Latino patients discontinued therapy because of adverse events.
Conclusions- Treatment with peginterferon alfa-2a and ribavirin for 48 weeks resulted in rates of sustained virologic response among patients infected with HCV genotype 1 that were lower among Latino whites than among non-Latino whites. Strategies to improve the sustained virologic response in Latinos are needed.
Latinos are the fastest-growing minority group in the United States and will represent 15% of the total U.S. population by 2010.1 The prevalence of infection with the hepatitis C virus (HCV) has been reported to be 2.1% among Mexican Americans, as compared with 1.5% among non-Hispanic whites.2 However, the overall prevalence among Latinos may be higher than that in the Mexican-American subgroup. Another analysis has shown that the HCV-related mortality rate among Latinos is nearly twice the rate among non-Latino whites.3 HCV infection in Latinos is characterized by more aggressive inflammatory activity and fibrosis,3,4,5,6 greater disease progression,7,8 and greater risk of cirrhosis6,9 than in non-Latino whites and blacks.
Latinos have typically been underrepresented in large clinical studies5,6; however, small studies of the use of peginterferon alfa-2b plus ribavirin in Latino patients infected with HCV genotype 1 have suggested a low rate of sustained virologic response (23 to 24%).10,11 The Latino Study was a multicenter, prospective study designed to examine the effect of Latino ethnic background on the response to treatment with peginterferon alfa-2a plus ribavirin in patients who were infected with HCV genotype 1 and who had not previously been treated.
Discussion
This large, prospective study shows that Latino white patients infected with chronic HCV genotype 1 have a lower rate of response to standard therapy with peginterferon alfa-2a and ribavirin than do non-Latino whites. The primary efficacy analysis showed that the sustained virologic response rate was lower in the Latino group than in the non-Latino group.
Multiple logistic-regression analysis with adjustment for baseline characteristics revealed that non-Latino ethnic background was a strong independent predictor of a sustained virologic response. The baseline viral load was predictive of the rate of sustained virologic response in the total study population as well as in both study groups, a finding that is consistent with the results of other studies.12,13,14,15 In this study, a low alanine aminotransferase quotient and the absence of cirrhosis predicted a sustained virologic response among Latinos, whereas a high alanine aminotransferase quotient and male sex were predictive among non-Latinos.
The difference in the rate of sustained virologic response between Latinos and non-Latinos could not be explained completely by the baseline characteristics of the two groups. Characteristics that have been reported to be associated with the response to therapy, including the HCV RNA level, sex, and weight, appeared to be similar in the two groups.16,17 Contrary to previously published data,18 the response rate among patients with higher BMI values was at least as high as the rate among patients with lower BMI values. This finding may be due to a recognition that higher ribavirin doses were required to optimize therapy in obese patients, as well as to the use of 200 mg instead of 600 mg as the step-down dose reduction to manage anemia.19 The finding is consistent with the observation that the BMI, which was higher in Latinos, did not explain the difference in response between the two groups after adjustment for multiple factors, including the presence or absence of cirrhosis, alanine aminotransferase quotient, age, and HCV RNA viral load.
Other factors related to treatment, such as the total amount of drug received, need for dose reduction, discontinuation of a study drug, and compliance with therapy, did not explain the lower sustained virologic response rate among Latinos than among non-Latinos. More Latinos than non-Latinos discontinued the study drugs owing to an insufficient therapeutic response, whereas there was a trend toward a higher rate of discontinuation because of safety concerns among non-Latino patients. In a noncontrolled study of treatment with interferon alfa-2b and ribavirin, Cheung et al.20 found that premature discontinuation of treatment was a reason for a lower rate of sustained virologic response among Latino patients than among non-Latino whites, but the authors did not differentiate between discontinuation due to side effects and discontinuation for other reasons.
Excessive weight, steatosis, and insulin resistance have all been shown to reduce the probability of a sustained virologic response to treatment.18,21,22 Latinos have a higher prevalence of insulin resistance and diabetes mellitus than do non-Latino whites and blacks.23 In our study, greater proportions of Latino patients were obese, had diabetes as a concomitant disease, and had more than 33% fatty cells on liver biopsy, results that are consistent with previous findings.6 Although the metabolic profiles of patients were not assessed in this trial, it would be of interest to explore the interplay between metabolic status and treatment response among Latinos.
Latinos also have an increased prevalence of other risk factors that are associated with a reduced rate of response to standard HCV therapy, including cirrhosis, HIV coinfection, heavy alcohol consumption, and drug abuse.5,6,8 Therefore, we controlled for these factors by excluding patients with HIV coinfection and lifestyle risk factors and balanced the proportions of patients with cirrhosis in the two groups. The effects of race and HCV genotype were also controlled by enrolling only whites with genotype 1 infection. In particular, blacks are known to have a low rate of response to standard therapy.14,24 We further controlled for the effect of race by limiting the Latino population to those whose parents and grandparents were also white Latinos. Because Latinos of Caribbean descent are more likely than other Latinos to have relatives of African origin in their ancestry, in a post hoc analysis we compared the rates of sustained virologic response among Latinos of various countries of origin and found no significant differences.
Thus, by controlling for multiple variables in the design of the study, we demonstrated that there are underlying factors related to Latino or non-Latino background, possibly due to genetic and immune differences, in the response to HCV infection - and particularly in the proportion of patients without a response. An analogy can be drawn with black patients in whom a diminished sustained virologic response probably reflects reduced sensitivity to interferon25 or diminished HCV-specific immunity.26 Toll-like receptors, which normally contribute to innate immunity27,28 and are associated with protection from the development of fibrosis,29 may contribute to reduced response rates. Studies have suggested that a poor response to therapy with peginterferon and ribavirin may be due to genetic variation, but little is known about whether specific haplotypes are more common in certain racial or ethnic groups, and we did not conduct any genetic analyses.30,31 The multiple exclusions that were needed in our study to ensure that the two groups were comparable is also a limitation of the study. The estimated sustained virologic response of Latino patients may be generalizable only to the Latino population with characteristics similar to those in this study; however, the study design allowed us to explore underlying differences in two relatively homogeneous populations.
The results of this large, prospective trial show that Latino white patients have a lower rate of response to standard HCV therapy than do non-Latino whites, substantiating findings from other small or retrospective studies.20,32,33,34,35 These findings point to an important public health issue, since Latinos, the fastest-growing minority population in the United States, have a higher prevalence of HCV2 and more rapid fibrosis progression6 than do non-Latino whites. The predicted expansion of the number of Latinos in the United States suggests that the prevalence of HCV infection will increase. These factors are likely to contribute to the already increasing rates of illness and death due to liver disease among Latinos.
The results of this study add to a growing body of evidence of differences in treatment responses among ethnic groups and underscore the need to optimize treatment strategies in order to improve the rate of sustained virologic response among Latino patients infected with HCV genotype 1. Latinos should be considered for clinical trials involving specifically targeted antiviral therapies for hepatitis C to determine whether the addition of investigational agents to standard therapy improves the rate of response in this difficult-to-treat population.
Supported by Roche.
Dr. Rodriguez-Torres reports receiving consulting fees from Roche, Abbott Laboratories, Anadys, Pharmasset, Vertex, GlaxoSmithKline, Bristol-Myers Squibb, and Merck, lecture fees from Roche, and grant support from Vertex, Anadys, Roche, Valeant, GlaxoSmithKline, Novartis, Wyeth, Bristol-Myers Squibb, Virochem Pharma, Idera, Intarcia, Pharmasset, Sanofi-Aventis, Human Genome Sciences, Idenix, Pfizer, Gilead, Tibotec, and Merck; Dr. Jeffers, consulting and lecture fees from Roche and Bristol-Myers Squibb and grant support from Roche, Gilead Sciences, Bristol-Myers Squibb, and the Health and Environmental Science Institute; Dr. Sheikh, lecture fees and grant support from Roche; Dr. Rossaro, lecture fees from Roche, Schering, and Three Rivers, consulting fees from Roche and Schering, and grant support from Roche, Schering-Plough, Salix, Novartis, and Bristol-Myers Squibb; and Dr. Ankoma-Sey, consulting fees from Roche, Schering, Three Rivers, and Gilead, lecture fees from Axcan Pharma, Bristol-Myers Squibb, Roche, Gilead Sciences, Schering-Plough, Three Rivers, and Valeant, and grant support from Roche; Dr. Hamzeh, being an employee of Roche and owning equity in the company; and Dr. Martin, receiving consulting fees from Roche, Gilead, Bristol-Myers Squibb, and Vertex, lecture fees from Gilead, Bristol-Myers Squibb, Novartis, and Roche, and grant support from Gilead, Bristol-Myers Squibb, Vertex, and Roche. No other potential conflict of interest relevant to this article was reported.
We thank Ellen Lentz, Ph.D., of Roche, and Ambrose Kwok, Ph.D., of Everest Clinical Services, for statistical support and Susan Sutch, Pharm.D., of Envision Pharma for editorial assistance, which was funded by Roche.
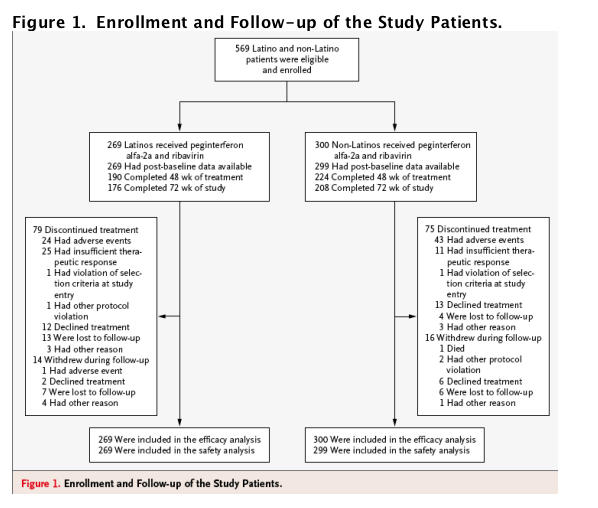
Results
Patients
Of the 569 patients enrolled between August 2004 and September 2007 who received one or more doses of the study drugs, 269 were Latino whites and 300 were non-Latino whites. Similar proportions of Latino and non-Latino patients withdrew from the study (Figure 1). More than twice as many Latino patients as non-Latino patients were withdrawn from the study because of an insufficient therapeutic response or loss to follow-up.
Most of the Latino patients had origins in Mexico and Puerto Rico. As compared with the non-Latino group, the Latino group had higher percentages of patients with an age of 40 years or younger, a body-mass index (BMI, the weight in kilograms divided by the square of the height in meters) of more than 27 or more than 30, an alanine aminotransferase quotient (the alanine aminotransferase level divided by the upper limit of the normal range [ULN]) of more than 3, and cirrhosis (Table 1). The enrollment of Latinos with cirrhosis neared the planned proportion of 10% during the study, at which point any patients undergoing screening who met the enrollment criteria were allowed to participate; by the end of the study, there was a slightly higher proportion of patients with cirrhosis in the Latino group (13%, vs. 10% in the non-Latino group). With regard to preexisting conditions, more Latino patients had diabetes mellitus (9%, vs. 3% of non-Latino patients) and more non-Latino patients had hypertension (24%, vs. 18% of Latinos). In both groups, 5% of patients had dyslipidemia.
Virologic Response Rates and Histologic Findings
The primary analysis revealed that the rate of sustained virologic response was significantly lower among Latinos than among non-Latinos (34% vs. 49%; absolute difference, -16 percentage points; 95% confidence interval [CI], -24 to -8; P<0.001). The rate of virologic response was lower for the Latino group than the non-Latino group at every time point at which data were available (Figure 2A). A post hoc analysis showed that rates of sustained virologic response were similar regardless of the country of origin of Latinos (see Table 1 in the Supplementary Appendix).
Figure 2. Clinical Outcomes among the Study Patients.
Panel A shows the percentages of the 269 Latino patients and 300 non-Latino patients with a virologic response, defined as an undetectable serum hepatitis C virus (HCV) RNA level (<28 IU per milliliter), during the study period. Treatment was administered through week 48, and the primary end point of a sustained virologic response (SVR) was measured at week 72 (24 weeks after treatment cessation). The reductions in the virologic response (and the corresponding 95% confidence intervals) among the Latino patients as compared with the non-Latino patients were as follows: -6 percentage points (-12 to 0) for week 4, -;15 percentage points (-23 to -7) for week 12, -13 percentage points (-22 to -6) for week 24, -17 percentage points (-24 to -9) for week 48, and -16 percentage points (-24 to -8) for week 72. Patients with missing HCV RNA measurements at the various time points were included in the analysis as not having a response. Panel B shows the percentages of the 269 Latino patients and 300 non-Latino patients with various virologic responses at week 72 in the intention-to-treat analysis. A total of 90 patients in the Latino group and 148 in the non-Latino group had a sustained virologic response; 50 in each group had a relapse, and 129 and 102, respectively, did not have a response. In Panels A and B, patients with missing HCV RNA measurements at the various time points were included in the analysis as not having a response. Panel C shows the effect of Latino ethnic background (vs. non-Latino background) on the rate of SVR, according to baseline characteristics. Although the Breslow-Day test of homogeneity showed a significant effect of an alanine aminotransferase (ALT) quotient (the ALT level divided by the upper limit of the normal range [ULN]) that was 3 or less, as compared with a value that was more than 3, the point estimates for the two odds ratios according to ALT category were both less than 1.0. This similarity suggests that the relative effect of Latino versus non-Latino background is more pronounced among patients with an ALT quotient of more than 3. Odds ratios for Latino versus non-Latino background were similar among the categories of all other baseline characteristics. The body-mass index (BMI) is the weight in kilograms divided by the square of the height in meters. CMH denotes Cochran-Mantel-Haenszel.
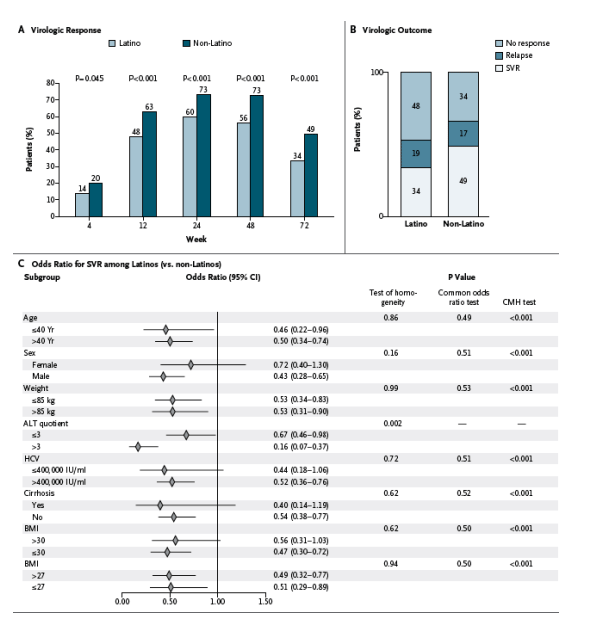
The rate of relapse among patients with a response at week 48 (the end of treatment) was 36% (50 of 138 patients) in the Latino group and 26% (50 of 195 patients) in the non-Latino group. In the intention-to-treat population, however, the proportion of patients who had a relapse was similar in the two groups (50 of 269 Latinos [19%] and 50 of 300 non-Latinos [17%]) (Figure 2B).
The odds ratios for a sustained virologic response among Latinos, as compared with non-Latinos, are shown in Figure 2C according to baseline characteristics. The odds ratios were similar across the baseline characteristics except for the more pronounced effect in the Latino group of an alanine aminotransferase quotient that was more than 3 of the normal range (vs. three times or less). Although the BMI was higher in Latinos than in non-Latinos, the difference in rates of sustained virologic response between the two groups persisted across BMI strata when corrected for the alanine aminotransferase quotient, age, and viral load. We observed a linear effect of a BMI of 30 or less on the rate of sustained virologic response, with higher values having a constant effect, among both Latinos and non-Latinos (data not shown).
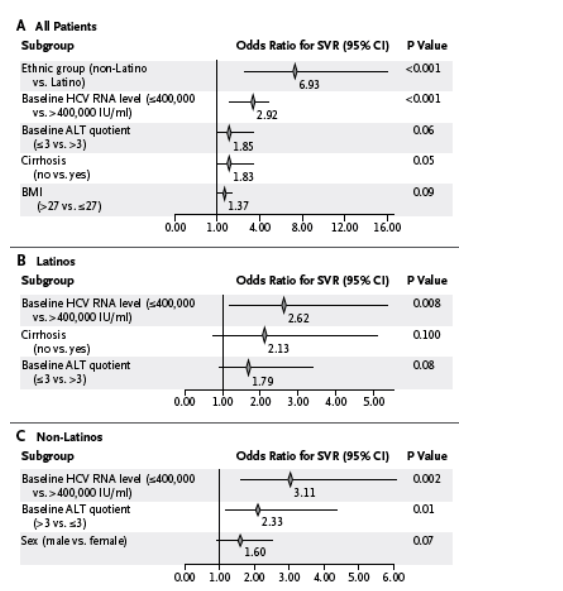
On the basis of multiple logistic-regression analyses, ethnic background was a predictor of the rate of sustained virologic response that remained significant after adjustment for all other significant baseline characteristics - including those with a significant interaction with ethnic group, such as the alanine aminotransferase quotient (Figure 3). Baseline HCV RNA levels were predictive of the rate of sustained virologic response in the entire study population and in each of the two groups. However, other baseline predictors varied between the two groups. A baseline alanine aminotransferase quotient of 3 or less was predictive of a sustained virologic response among Latino patients, whereas an alanine aminotransferase quotient of more than 3 was predictive among non-Latino patients. In addition, whereas an absence of cirrhosis was predictive of a sustained virologic response in the total population as well as in the Latino group, male sex was predictive in the non-Latino group.
Figure 3. Associations of Baseline Prognostic Factors with a Sustained Virologic Response (SVR).
The results of multiple logistic-regression analysis are shown for the total study population (Panel A), the Latino group only (Panel B), and the non-Latino group only (Panel C). A stepwise selection method, with P values of 0.15 or less for entry into the model and P values of 0.20 or more for removal, was used in the multivariable logistic-regression analysis. The odds ratio for the interaction of ethnic group and the alanine aminotransferase (ALT) quotient was 0.22 (95% confidence interval [CI], 0.09 to 0.56; P=0.001). The ALT quotient was calculated as the ALT level divided by the upper limit of the normal range (ULN). The body-mass index (BMI) is the weight in kilograms divided by the square of the height in meters. HCV denotes hepatitis C virus.
In patients for whom histologic data were available from liver biopsies performed before initiation of study treatment (in most patients, within 3 months before initiation or at screening) and at week 72, the histologic response to treatment was greater among non-Latinos than among Latinos. At week 72, 59% of non-Latino patients and 47% of Latino patients had a response (a drop of >/=2 points from the baseline score for the Ishak-modified histologic-activity index, P=0.03). Similarly, with regard to the fibrosis score, more non-Latinos (42%, vs. 25% of Latinos) had an improvement (a decrease of >/=1 point) and more Latinos (22%, vs. 18% of non-Latinos) had a worsening (an increase of 1). The overall response, reflected by fibrosis scores, favored non-Latinos (P=0.003). A greater proportion of Latinos had more than 33% fatty cells on liver biopsy (13%, vs. 8% of non-Latinos).
Drug Doses and Side Effects
The mean and median doses of ribavirin and peginterferon alfa-2a were 13.6 and 13.7 mg per kilogram per day and 173.4 and 180.0 μg per week, respectively (Fig. 1 in the Supplementary Appendix). The peginterferon alfa-2a dose was reduced in 23% of Latinos and 27% of non-Latinos. The ribavirin dose was reduced in 35% of Latinos and 36% of non-Latinos (Table 2 in the Supplementary Appendix). Compliance with treatment was similar in the Latino and non-Latino groups, as reflected by the similar cumulative doses (7039 μg and 7270 μg of peginterferon, respectively; and 301,780 mg and 312,546 mg of ribavirin) and the similar proportions of patients who completed treatment (71% and 75%, respectively).
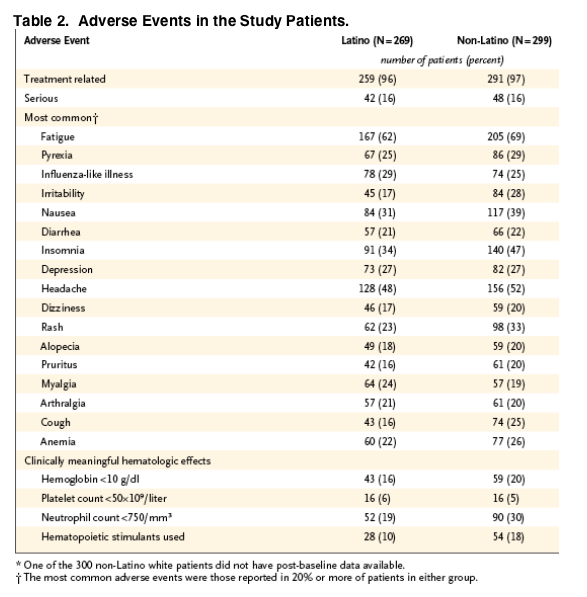
Adverse events were reported in 98% of all study patients, and most of the events were considered to be related to treatment. The incidence was generally higher in the non-Latino group than in the Latino group (Table 2). One non-Latino patient died of liver failure from cirrhosis due to alcohol abuse. Discontinuation of treatment due to adverse events occurred in 9% of Latinos and 14% of non-Latinos (Figure 1), with depression (in three Latinos and four non-Latinos), suicidal ideation (in three Latinos and three non-Latinos), drug abuse (in two Latinos and three non-Latinos), and fatigue (in two Latinos and three non-Latinos) reported as the most common reasons.
Methods
Study Patients
Latino and non-Latino white patients were eligible for the study if they were between the ages of 18 and 65 years, had chronic HCV genotype 1 infection, and had no history of treatment for HCV infection. Eligible patients were identified at 52 centers in the continental United States and Puerto Rico. Patients were also required to have a serum HCV RNA level of 10 IU per milliliter or higher, an elevated serum alanine aminotransferase level within 12 months before enrollment, histologic evidence of chronic hepatitis C on centrally reviewed liver-biopsy specimens obtained within 18 months before the start of the study, and compensated liver disease (Child-Pugh class A cirrhosis with a score of 6). We planned to enroll a sufficient number of patients with cirrhosis to have approximately 10% in each of the two groups.
Latino ethnic background was defined as a geographic, historical, and cultural heritage shared among persons from Spanish-speaking countries in South and Central America, Mexico, and the Caribbean. Both parents and all grandparents of the patient had to be Latino, with Spanish as the primary language. On self-report, Latinos and non-Latinos had to be white; native aboriginal Indians, Asians, and blacks were excluded.
Other exclusion criteria were a hemoglobin level of 12 g per deciliter or lower for women and 13 g per deciliter or lower for men, a white-cell count of 11.5x109 per liter or lower, a platelet count of 100,000 per cubic millimeter or lower, drug abuse within the previous 12 months or alcohol abuse within the previous 6 months, a severe psychiatric disorder or a history of such a disorder, other forms of liver disease, hepatitis A or B infection, human immunodeficiency virus (HIV) infection, clinically significant cardiac or renal disease, a seizure disorder, and retinopathy.
The study was conducted in conformance with the principles of the Declaration of Helsinki and local laws and regulations. The institutional review boards of the participating sites approved the protocol and consent forms. All participants provided written informed consent. Roche sponsored the trial and collected and analyzed the data. The lead author was involved in the design of the trial and analysis of the data and vouches for the accuracy and completeness of the data. One academic and one industry author wrote the first draft of the manuscript; all authors contributed to subsequent drafts.
Study Design
This trial was a prospective, multicenter, open-label, nonrandomized study of treatment with peginterferon alfa-2a in Latino and non-Latino whites with chronic HCV genotype 1 infection who had not previously received treatment. Enrolled patients were treated for 48 weeks with peginterferon alfa-2a (Pegasys, Roche), at a dose of 180 μg per week, and ribavirin (Copegus, Roche), at a dose of 1000 mg per day (for patients with a body weight <75 kg) or 1200 mg per day (for those with a body weight 75 kg). Assessments of virologic and biochemical responses were performed at weeks 4, 12, 24, 48, 60, and 72. Clinical adverse events, laboratory values, vital signs, and dose adjustments of the study drugs were monitored throughout the study. Doses of ribavirin and peginterferon alfa-2a could be adjusted to manage side effects (see the Supplementary Appendix, available with the full text of this article at NEJM.org). Compliance was assessed at each clinic visit. Histologic assessments were performed at the time of screening or within 18 months before the study and at week 72.
Clinical Assessments and End Points
The primary efficacy end point was a sustained virologic response, defined as an undetectable HCV RNA level (<28 IU per milliliter) 24 weeks after the end of the treatment period (week 72). Secondary end points were virologic responses during the treatment period: a rapid virologic response (an undetectable HCV RNA level at week 4), a complete early virologic response (an undetectable HCV RNA level at week 12), and virologic responses at week 24 and at the end of treatment (week 48). Relapse was assessed between week 48 and week 72.
Laboratory and Viral Testing
HCV genotyping was performed at screening by ICON Laboratories (Farmingdale, NY) with the use of the VERSANT HCV Genotype 2.0 Assay (LiPA) (Bayer HealthCare). Serum HCV RNA levels were measured by means of the High Pure System-COBAS TaqMan HCV Monitor Test (Roche), which has a limit of detection of 28 IU per milliliter and a range for quantification of 30 to 200 million IU per milliliter. Liver-biopsy specimens blinded with regard to the ethnic group of the patient obtained before and after treatment were evaluated by a central pathologist (at Liver Research Institute, Downey, CA) using the Ishak modification of the histologic-activity index. Laboratory tests were performed at a central laboratory (ICON Laboratories).
Statistical Analysis
The study was designed for the enrollment of equal numbers of Latino and non-Latino whites, with a total of 540 patients, for a statistical power of 94% to determine whether the rate of sustained virologic response in the Latino group was lower than that in the non-Latino group by 15% or more. This calculation was based on a two-group test of equivalence in proportions involving a one-sided 97.5% confidence interval, assuming that both groups of patients had rates of sustained virologic response of 50%. All enrolled patients who took a dose of medication were included in the efficacy evaluations, and patients with missing HCV RNA measurements were considered not to have had a response. All patients who received one or more doses of the study drugs and underwent one or more safety assessments were included in the safety analyses (Figure 1).
For the analysis of the primary efficacy end point, the two-sided 95% confidence interval for the difference in the proportions of patients in the Latino group and the non-Latino group who reached the primary end point was calculated with the use of a normal approximation. The rate of sustained virologic response in the Latino group was considered to be noninferior to that in the non-Latino group if the lower bound of the two-sided 95% confidence interval was greater than -0.15. The Cochran-Mantel-Haenszel test and the Breslow-Day test for homogeneity of the odds ratio were used to assess differences between the two groups with regard to baseline characteristics. The primary efficacy end point was also assessed with the use of a multivariable logistic-regression analysis to evaluate the effects of baseline prognostic factors on the probability of a sustained virologic response. To identify factors that were associated with rates of sustained virologic response, a stepwise variable-selection procedure was used, with a P value of 0.15 or less for entry into the model and a P value of 0.20 or more for removal from the model.
|
|
| |
| |
|
|
|