| |
DISCOVERY OF POTENT, BIOAVAILABLE HCV NS3/4A PROTEASE INHIBITORS THAT DISPLAY UNIMPAIRED ACTIVITY AGAINST AN NS3 SEQUENCE VARIANT ASSOCIATED WITH RESISTANCE TO LINEAR TETRAPEPTIDES AND MACROCYCLIC INHIBITORS
|
| |
| |
Reported by Jules Levin
EASL 2009
B. Buckman, K. Kossen, D. Ruhrmund, R. Rajagopalan, S. Stevens, L. Hooi, N. Snarskaya, T. Wang, J. Hong, C. Moy, S. R. Lim, L. Pan, L. Huang, A. Stoycheva, L. Beigelman, and S. Seiwert
InterMune, Inc., Brisbane, CA, USA
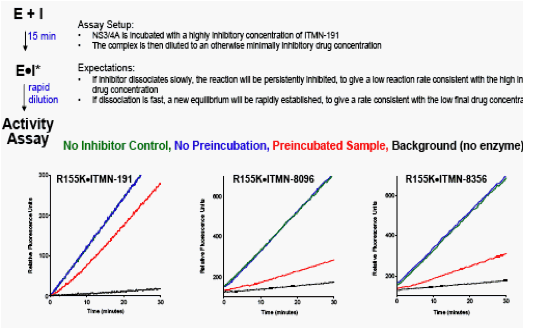
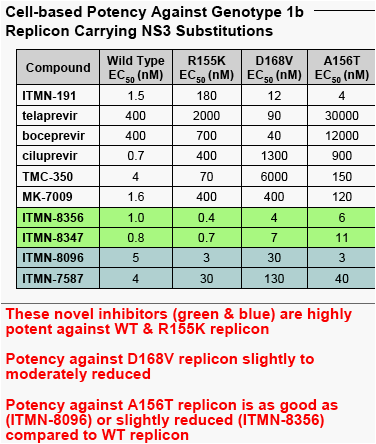
"Two novel, structurally distinct classes of NS3/4A PIs show very potent activity against WT & R155K NS3
-- These novel inhibitors (green & blue) are highly potent against WT & R155K replicon
-- Potency against D168V replicon slightly to moderately reduced
-- Potency against A156T replicon is as good as (ITMN-8096) or slightly reduced (ITMN-8356) compared to WT replicon"

ABSTRACT
Background: Agents that inhibit serine protease activity of HCV NS3/4A have emerged as potentially significant components of regimens targeting HCV virus. ITMN-191 is a highly potent and selective macrocyclic inhibitor of the HCV NS3/4A serine protease. In short duration human clinical studies, administration of ITMN-191 has demonstrated multi-log10 reductions in circulating HCV RNA and a very favorable safety profile. As with the linear tetrapeptide telaprevir, a fraction of patients receiving ITMN-191 monotherapy experience virologic rebound driven by the emergence of drug resistant mutations. The NS3 escape mutation R155K is the only NS3 substitution that is common to both classes of structurally diverse inhibitors in monotherapy. While the clinical significance of escape mutations is yet to be defined, it would be valuable to identify chemical matter with unimpaired activity against NS3 with R155K.
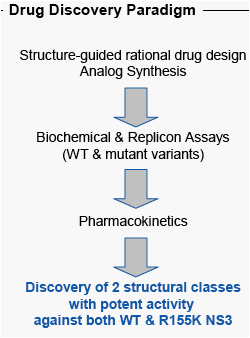
Methods: Structure-guided rational drug design employed both molecular modeling and protein-inhibitor complex x-ray structures of both WT and R155K NS3. Biochemical and replicon assays were used to determine activity against NS3 variants. Pharmacokinetic performance was examined by standard procedures that were IACUC approved.
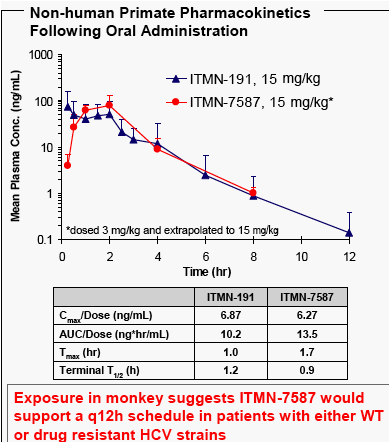
Results: We have discovered inhibitors with no reduction in potency against R155K-bearing NS3. The best compounds display an equivalent, sub-nanomolar potency against WT and R155K replicons. These compounds also display a slow-binding mechanism that is associated with slow dissociation and persistent inhibition of protease activity. Pharmacokinetics and plasma AUC following PO dosing in monkeys are equivalent or better than ITMN-191, a compound with robust virologic effect in q12h dosing regimens.
Conclusion: The overlap in NS3 escape mutations observed with linear tetrapeptides and macrocyclic compounds indicates that multiple drug resistant strains of HCV may develop. Using structure-based drug design, we have discovered potent, bioavailable inhibitors that show equal activity against WT and R155K-bearing NS3; the NS3 variant observed in monotherapy with both telaprevir and a macrocyclic compound. The favorable pharmacokinetic profile of these compounds in monkeys suggests that their exposure would support a q12h schedule for both WT and drug resistant strains. Further investigation of these compounds is warranted.
BACKGROUND
In short duration human clinical studies, administration of NS3/4A protease inhibitors (PIs) can lead to multi-log10 reductions in HCV RNA
PI monotherapy results in viral rebound due to the emergence of drug resistant viral variants
PI in combination with SOC (pegylated interferon + RBV) suppresses the emergence of drug resistant viral variants except in:
- Treatment naïve patients with suboptimal interferon responses
- Many patients who failed prior SOC therapy
The spectrum of NS3 substitutions conferring PI resistance is compound specific, but certain amino acid substitutions are observed across multiple PIs
R155K confers a resistant phenotype to both linear tetrapeptides & macrocycles
NS3/4A PIs that are active against R155K could have value either as a frontline therapeutic option or for treatment of HCV resistant to the current generation of PIs
RESULTS
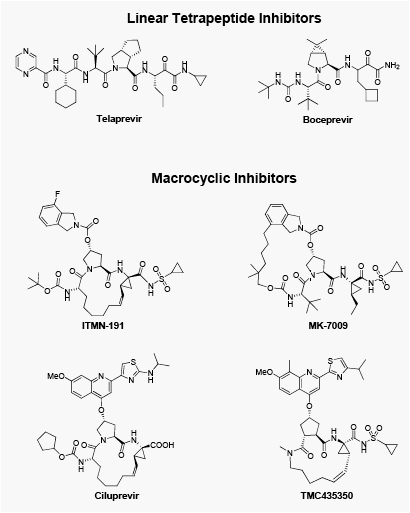
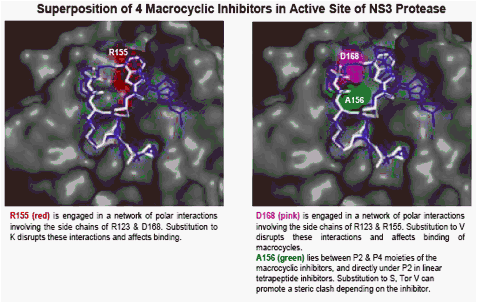
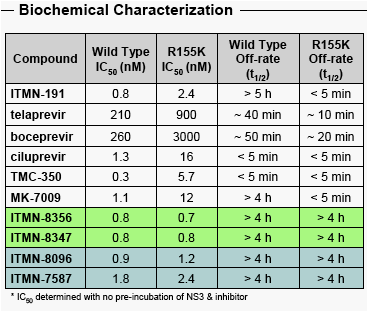
Two novel, structurally distinct classes of NS3/4A PIs show very potent activity against WT & R155K NS3
Slow dissociation/persistent inhibition of R155K is observed for these compounds, but not current clinical candidates
|
|
| |
| |
|
|
|