 |
 |
 |
| |
The MERIT Study of Maraviroc in Antiretroviral-Naive Patients With R5 HIV-1: 96-Week Results
|
| |
| |
Reported by Jules Lein
5th IAS Capetown July 19-22 2009
Heera J, Ive P, Botes M, DeJesus E, Mayer H, Goodrich J, Clumeck N, Cooper D, Walmsley S, Craig C, Reeves J, van der Ryst E and Saag M

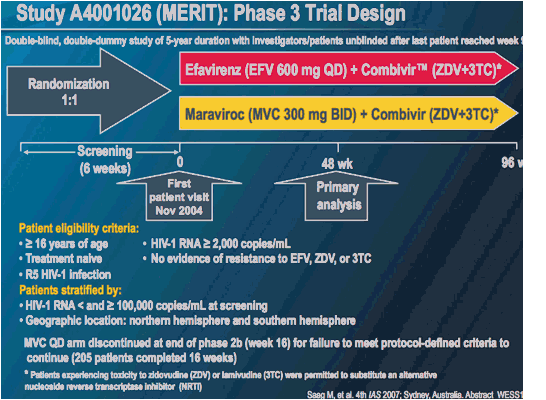
MERIT ES
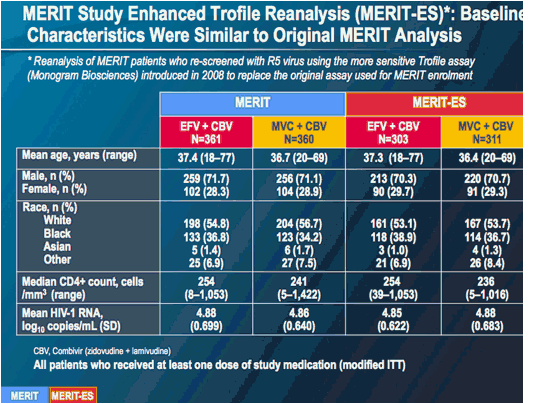
MERIT ES
* Adjusted for randomization strata
Includes all patients with R5 virus at screening by enhanced Trofile assay who received at least one dose of study medication; missing values classified as failures/non-responders; MERIT-ES per-protocol analysis (MVC+CBV N=285; EFV+CBV N=268): < 50 copies/mL difference = -0.029 (-10.7)
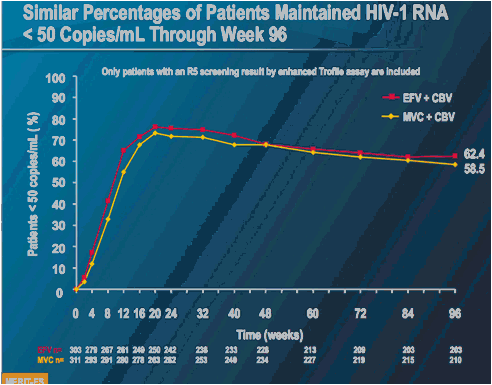
MERIT ES
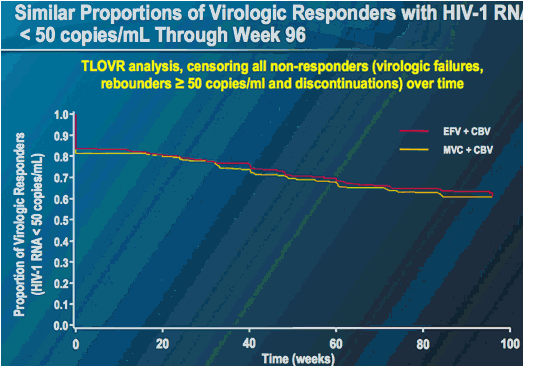
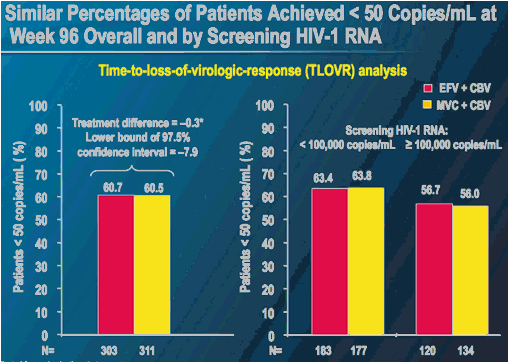
MERIT ES
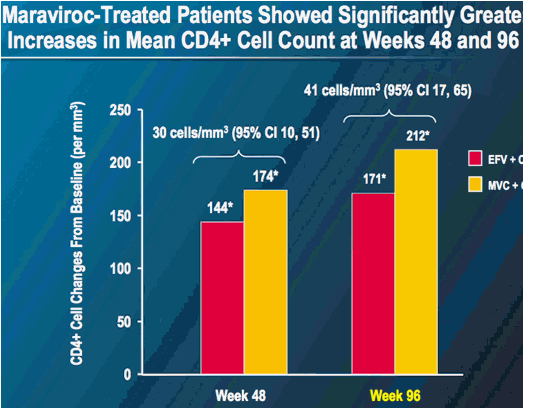
* Week 48 or 96 mean value adjusted for randomization strata
Last observation carried forward; blinded therapy values only. Includes all patients with R5 virus at screening by enhanced Trofile assay who received at least one dose of study medication
MERIT ES
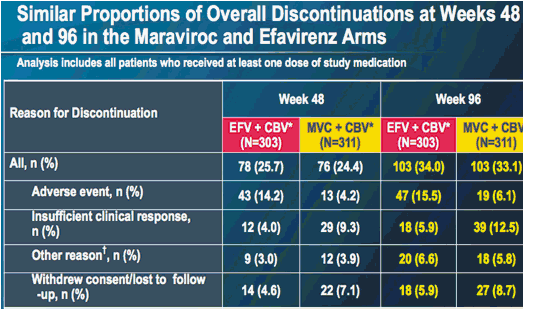
Only patients with an R5 screening result by enhanced Trofile assay are included
* 12% of patients on MVC and 9% on EFV switched to other NRTI backbones over 96 weeks
Includes one death in the MVC arm at week 48 and two deaths each in the MVC and EFV arms at week 96
At week 96, 45 patients in each treatment group (15% and 14% in the EFV and MVC groups, respectively) had experienced a serious adverse event (compared to 34 [11%] and 32 [10%] patients at week 48)
MERIT
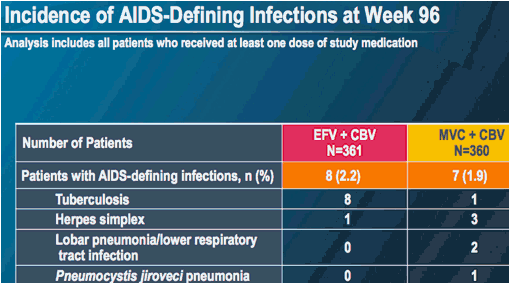
MERIT
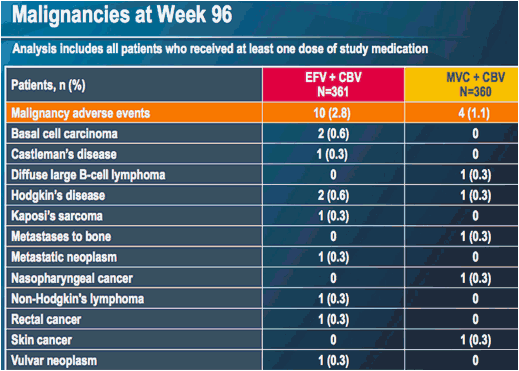
MERIT
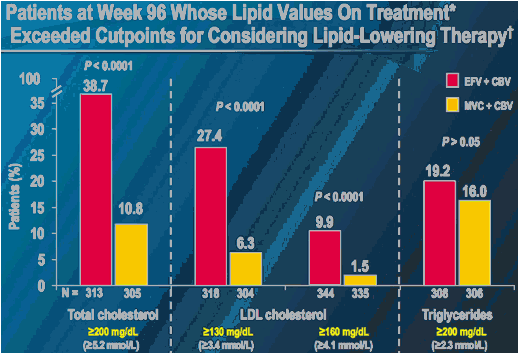
* At one or more on-study assessments (fasting lipoprotein profiles were obtained for each patient at week 24 and week 48, or at early termination)
As defined by NCEP guidelines
P-values are for comparisons of proportions (Pearson's chi-square test)
|
| |
|
 |
 |
|
|