 |
 |
 |
| |
Efficacy and Safety of raltegravir plus etravirine and darunavir/ritonavir in treatment-experienced patients with multidrug-resistant virus:
48-week results from the ANRS 139 TRIO trial
|
| |
| |
Reported by Jules Levin
5th IAS Capetown July 19-22 2009
Fagard C.1, Descamps D.2, Dubar V.3, Colin C.1,Taburet AM.4, Roquebert B.2, Katlama C.5, Jacomet C. 6, Piketty C.7, Molina JMM.8, Chene G.1, Yazdanpanah Y 3and the ANRS 139 TRIO Trial Group
1INSERM U897, Bordeaux, France; 2Hosp Bichat-Claude Bernard, Paris, France; 3Hosp Tourcoing, France;4Hosp Bicetre, Le Kremlin Bicetre, France; 5Hosp Pitie-Salpetriere, Paris, France;6Hosp Clermont-Ferrand, France; 7Hosp Georges Pompidou, Paris, France; 8Hosp Saint Louis, Paris, France


METHODS
Study design: Phase II non-comparative multi-center trial.
Eligibility criteria:
· age ≥ 18, written informed consent, HIV-1 infected patients
· viral load (VL) >1000 copies/ml
· patients naïve to the investigational drugs with a history of virologic failure while on NNRTI
· genotypic test at screening showing: ≥ 3 primary PI mutations among D30N, V32I, L33F, M46I/L, I47A/V, G48V, I50L/V, I54M, L76V, V82A/F/L/T/S, I84V, N88S and L90M (IAS 2006)
≥ 3 NRTI mutations (IAS)
≤3 darunavir resistance mutations among V11I, V32I, L33F, I47V, I50V, I54L/M, G73S, L76V, I84V and L89V (ANRS resistance algorithm, version16)
≤ 3 NNRTI mutations among A98G, L100I, K101Q/P/E, K103H/N/S/T, V106A/M, V108I, E138G/K/Q, V179D/E/F/G/I, Y181C/I/V/C/H/L, Y188C/H/L, G190A/C/E/Q/S, P225H, F227C/L, M230I/L, P236L, K238N/T and Y318F
Study treatment: raltegravir (one 400 mg tablet twice daily), etravirine (two 100 mg tablets twice daily) and darunavir/ritonavir (two 300 mg darunavir tablets plus 100 mg ritonavir twice daily). Backbone regimens included NRTI and/or enfuvirtide
Primary outcome measure: proportion of patients reaching plasma HIV-1 RNA <50 copies/ml at wk 24
Secondary outcome measures: proportion of patients with: VL >50copies/ml at wk24 or among those with VL<50 copies/ml at wk 24, VL ≥50copies/ml on two consecutive measures between wk 24 and wk 48; VL<50 copies/ml at wk 48; change in plasma HIV-1 RNA levels and CD4 counts. Drug safety and tolerability.
RESULTS
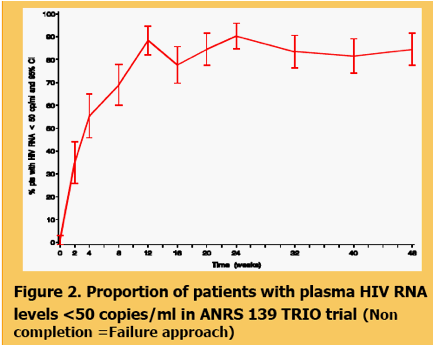

green line is CD4 change & red line is viral load decline
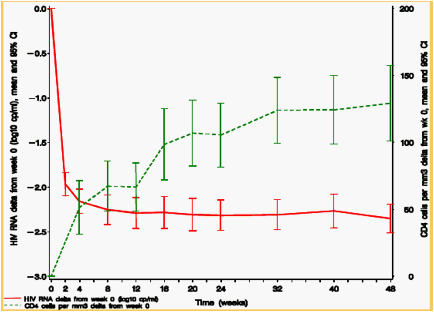
Figure 3. Mean change in viral load and CD4 cell count from baseline in ANRS TRIO trial

|
| |
|
 |
 |
|
|