 |
 |
 |
| |
Efficacy and Safety of Atazanavir/RTV vs. Lopinavir/RTV in Treatment-Naïve Patients
with Advanced Disease: CASTLE Study 96-Week Results
|
| |
| |
Reported by Jules Levin
5th International AIDS Society, July 19-22, 2009, Cape Town, South Africa
J. Uy,1 R. Yang,2 V. Wirtz,2 L. Sheppard,3 and J. Absalon2 for the BMS AI424138 Study Group
1Bristol-Myers Squibb, Research and Development, Plainsboro, NJ, US; 2Bristol-Myers Squibb, Research and Development, Wallingford, CT, US;
3Bristol-Myers Squibb, Research and Development, Braine-l'Alleud, Belgium



RESULTS

Figure 1. CVR (HIV RNA < 50 copies/mL) by Baseline CD4 Cell Counts and HIV RNA Strata at Week 96 in (A) the ITT Population CVR (NC = F) and (B) the As-Treated Population CVR (NC = M)
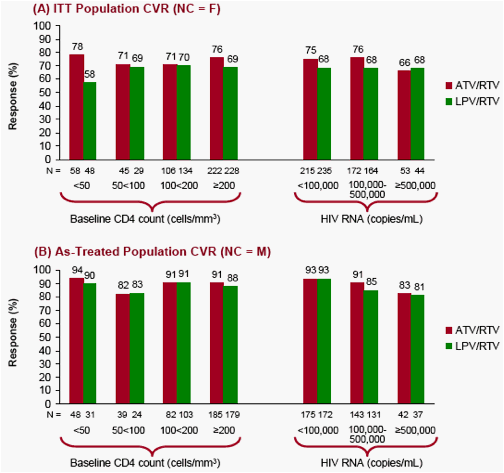
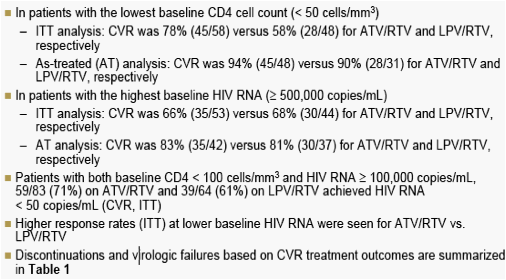
Table 1. Discontinuations and Virologic Failures Based on CVR Treatment Outcomes Through Week 96
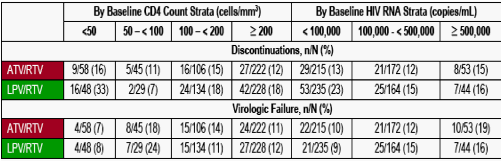
Virologic failure is defined as patients who never suppressed on the study through Week 96, discontinued due to insufficient viral load response through Week 96, or rebounded without resuppression

Analysis of mean change in CD4 cell count according to baseline CD4 count strata and HIV RNA strata are summarized in Table 2

Tolerability and Safety
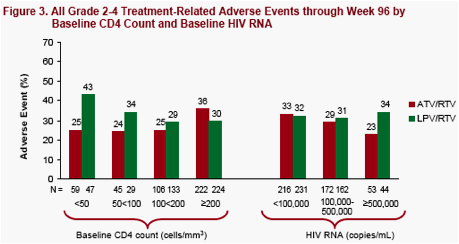
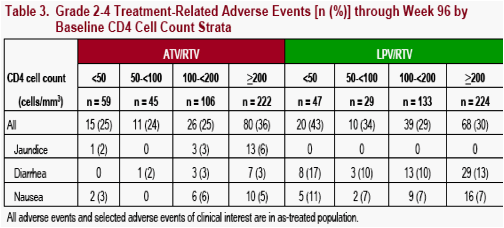
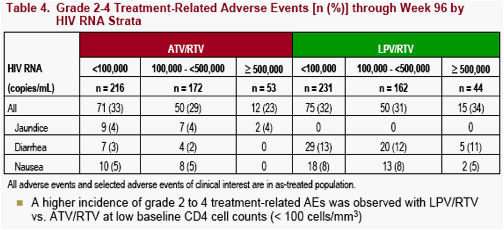
The incidence of all treatment-related grade 2 to 4 adverse events and selected grade 2 to 4 treatment-related adverse events (AEs) of clinical interest through Week 96 are summarized by baseline CD4 cell count strata (Figure 3 and Table 3) and baseline HIV RNA strata (Table 4)
REFERENCES
1. Johnson M, et al. 96-week comparison of once-daily atazanavir/ritonavir and twice-daily lopinavir/ritonavir in patients with multiple virologic failures. AIDS 2006;20(5):711-8.
2. Malan DR, et al. Efficacy and safety of atazanavir, with or without ritonavir, as part of once-daily highly active antiretroviral therapy regimens in antiretroviral-naive patients. J Aqcuir Immun Defic Syndr 2008;47(2):161-7.
3. Molina JM, et al. Atazanavir/ritonivir vs lopinavir/ritonavir in antiretroviral-naïve HIV-1-infected patients: CASTLE 96 week efficacy and safety. Poster presented at: The 48th Interscience Conference on Antimicrobial Agents and Chemotherapy/46th Infectious Disease Society of America Conference; 2008; Washington DC.
4. Uy J, etal. Efficacy and safety by baseline HIV RNA and CD4 count in treatment-naive patients treated with atazanavir/RTV and lopinavir/RTV in the CASTLE study. Poster presented at: 9th International Congress on Drug Therapy in HIV Infection; 2008; Glasgow, UK.
|
| |
|
 |
 |
|
|