 |
 |
 |
| |
ARTEMIS: Week 96 safety and efficacy of darunavir/r by gender, age and race
|
| |
| |
Reported by Jules Levin
5th IAS Capetown July 19-22 2009
Jan Fourie,1 Jason Flamm,2 Amalia Rodriguez-French,3 Don Kilby,4 Ben Van Baelen,5 Vanitha Sekar,6 Ludo Lavreys5
1Chronic Diseases of Lifestyle Unit, Tygerberg, South Africa; 2Kaiser Permanente Medical Group, Sacramento, CA, USA; 3Hospital Santo Tomas, Panama City, Panama;
4University of Ottawa Health Services, Ottawa, Canada; 5Tibotec BVBA, Mechelen, Belgium; 6Tibotec Inc., Yardley, PA, USA

INTRODUCTION


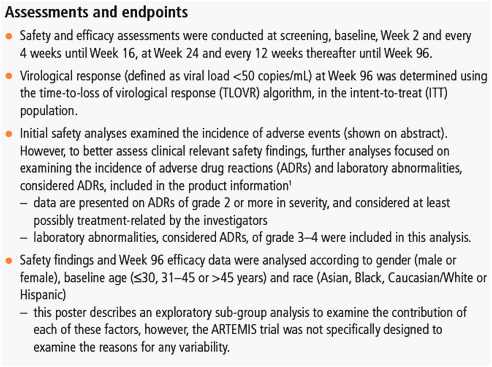

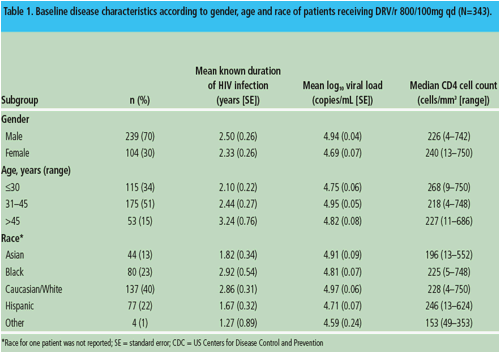
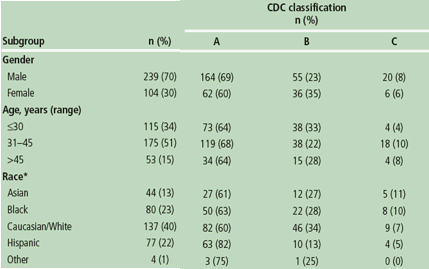

Figure 1. Virological response (percentage of patients with viral load <50 copies/mL) at Week 96 (ITT-TLOVR) according to gender, age and race of patients receiving DRV/r 800/100mg qd.
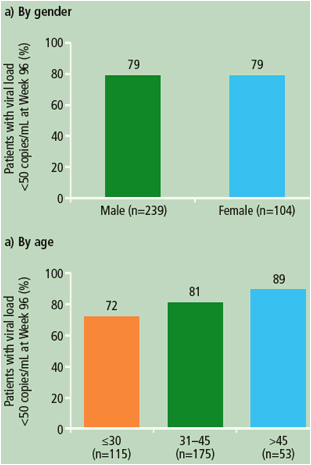
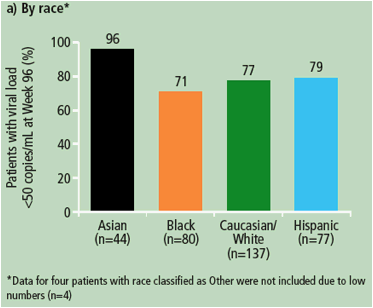
Figure 2. Overview of the most frequently reported ADRs at least grade 2 and possibly related to DRV/r treatment by the investigators (32% incidence in any subgroup) according to gender, age and race in patients receiving DRV/r 800/100mg qd.
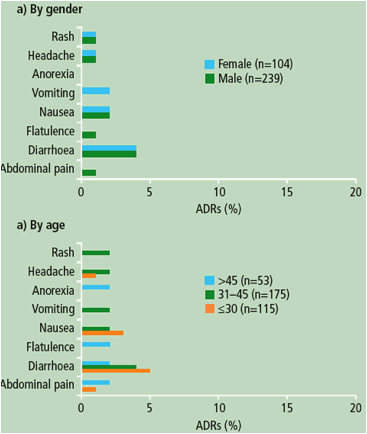
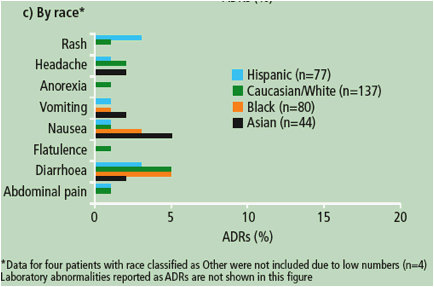

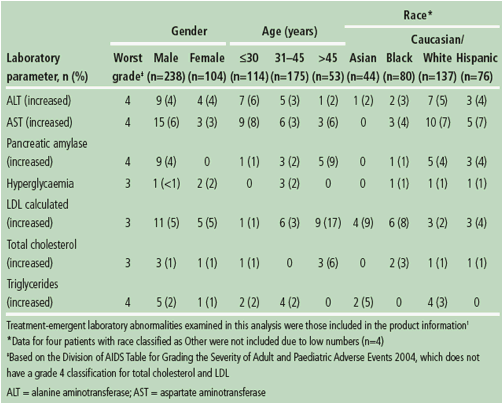
Table 2. Incidence of grade 3-4 treatment-emergent laboratory abnormalities, considered ADRs, observed in >2 patients in any subgroup through Week 96 according to gender, age and race in patients receiving DRV/r 800/100mg qd.
REFERENCES
1. PREZISTA (darunavir). Full Prescribing Information. Tibotec Inc. Revised February 2009 [accessed 17 June 2009]. Available from:
http://www.prezista.com/prezista/documents/us_package_insert.pdf.
2. PREZISTA (darunavir). EPARs for authorised medicinal products for human use. 12 February 2009 [accessed 06 May 2009]. Available from:
http://www.emea.europa.eu/humandocs/Humans/EPAR/prezista/prezista.htm.
3. DHHS. Guidelines for the use of antiretroviral agents in HIV-1 infected adults and adolescents. 3 November 2008.
4. Hammer SM, et al. JAMA 2008;300:555-70.
5. Prise en charge médicale des personnes infectées par le VIH Rapport 2008 (recommandations du groupe d'experts de France). Addendum du 13 avril 2009 au chapitre 5 : Traitement antirétroviral. Version 2 du 20 avril 2009.
6. Ortiz R, et al. AIDS 2008;22:1389-97.
7. Mills A, et al. AIDS 2009. epub.
8. Andrade-Villanueva J, et al. XVIIth International AIDS Conference, Mexico City, Mexico, 3-8 August 2008. Abstract TUPE0064.
|
| |
|
 |
 |
|
|