 |
 |
 |
| |
(IDSA) Darunavir, Ritonavir, and Etravirine Steady-state Pharmacokinetics in the Cervicovaginal Fluid and Blood Plasma of HIV-infected Women Enrolled in the GRACE Study
|
| |
| |
Reported by Jules Levin
47th annual meeting of the Infectious Diseases Society of America (IDSA), Philadelphia, PA, USA, October 29 to November 1, 2009
Kristine Patterson1, Naser Rezk1, Steven Jennings1, Ron Falcon2, Joseph Mrus2, Angela Kashuba1
1University of North Carolina Chapel Hill, NC, USA; 2Tibotec Therapeutics, Bridgewater, NJ

DRV and ETR achieve CVF exposures 150% and 130% higher, respectively, than BP in pre- and postmenopausal women
Further research is warranted to defi ne the effect of DRV and ETR on HIV-1 RNA in genital secretions

To date, DRV and ETR achieved among the highest penetration into CVF of other protease inhibitors and non-nucleoside reverse transcriptase inhibitors, respectively3-5 (Figure 5)
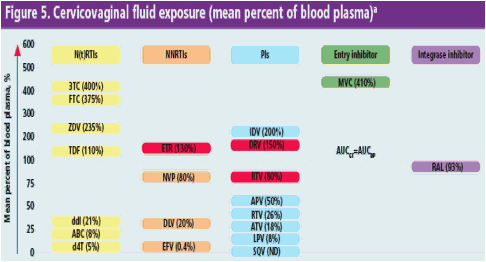
aAdapted from Dumond et al., 20073. N(t)RTI, nucleoside/nucleotide reverse transcriptase inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; PI, protease inhibitor; AUCGT, genital tract area under the curve; AUCBP, blood plasma area under the curve; ND, not detected; 3TC, lamivudine; MVC, maraviroc; FTC, emtricitibine; ZDV, zidovudine; IDV, indinavir; TDF, tenofovir disoproxil fumarate; ETR, etravirine; DRV, darunavir; RAL, raltegravir; NVP, nevirapine; APV, amprenavir; RTV, ritonavir; ddl, didanosine; DLV, delavirdine; ATV, atazanavir; ABC, abacavir; LPV, lopinavir; d4t, stavudine; EFV, efavirenz; SQV, saquinavir
DRV exposure in the female genital tract is 1.5-fold higher than in BP
- This CVF exposure is 3- to 20-fold higher than other protease inhibitors with the exception of indinavir
Ritonavir exposure in the female genital tract is higher in this study than in previous evaluations
ETR exposure in the female genital tract is 1.3-fold higher than in BP, unlike efavirenz, which shows minimal penetration into CVF3
For DRV, there was no difference in CVF and BP exposure between pre- and postmenopausal women; however, only two out of eight women were postmenopausal
The role of DRV and ETR in primary prevention remains uncertain as this study did not evaluate first dose PK.
Nonetheless, the concentrations measured are similar to other agents being investigated for primary prevention3-5
INTRODUCTION
Antiretroviral (ARV) therapy can be used in HIV prevention strategies
- Primary prevention through pre-exposure prophylaxis or post-exposure prophylaxis
- Secondary prevention through the decrease of HIV replication in the genital secretions of HIV-infected individuals
Both prevention strategies depend on adequate penetration of ARVs into the genital tract
GRACE (Gender, Race And Clinical Experience) is the largest ARV trial to focus on women with HIV-1 in North America, and was designed to assess sex and race differences in effi cacy, safety and tolerability of darunavir/ ritonavir (DRV/r) plus an optimized background regimen over 48 weeks in treatment-experienced patients1
The aim of this prospective substudy was to assess the pharmacokinetics (PK) of DRV, ritonavir and etravirine (ETR) in female cervicovaginal fluid (CVF) and blood plasma (BP) in HIV-infected women from the GRACE study.
We also investigated the effect of menopause on both CVF and BP ARV PK
METHODS
The GRACE study design
GRACE was a multicenter, open-label, single-arm, Phase IIIb study conducted at 65 sites across the United States, Puerto Rico and Canada, which enrolled treatment-experienced patients (viral load ≥1000 HIV-RNA copies/mL) aged ≥18 years with documented HIV-1 infection (Figure 1)


aPatients were allowed to enter the study on treatment interruption of ≥4 weeks; bInvestigator-selected nucleoside reverse transcriptase inhibitors and NNRTIs included; ENF, TPV or agents from novel classes were not allowed; PI, protease inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; HAART, highly-active antiretroviral therapy; DRV/r, darunavir/ritonavir; ETR, etravirine; ENF, enfuvirtide; TPV, tipranavir; bid, twice daily; OBR, optimized background regimen

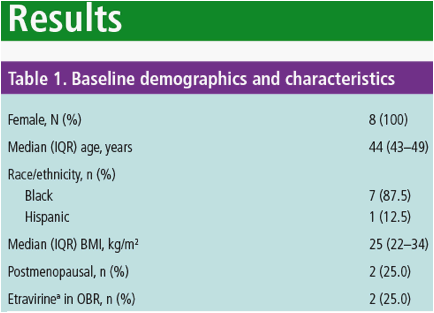
aNo postmenopausal women received etravirine; IQR, interquartile range; BMI, body mass index; OBR, optimized background regimen

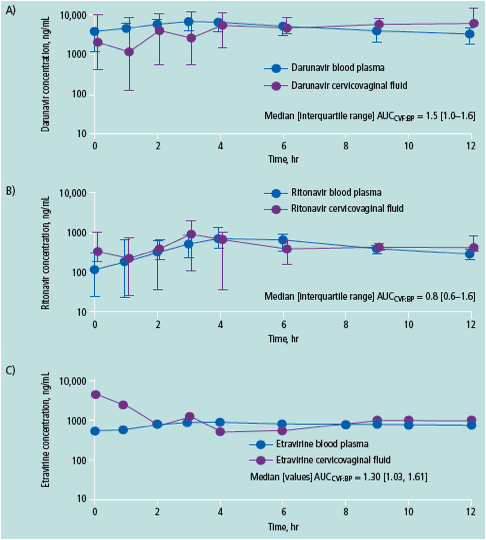

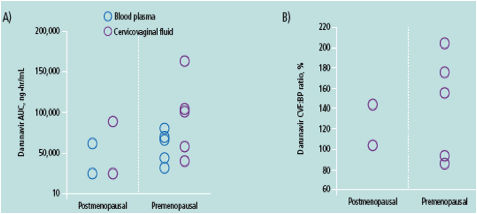
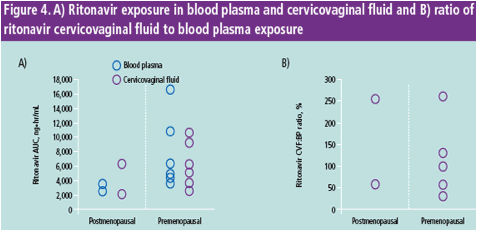
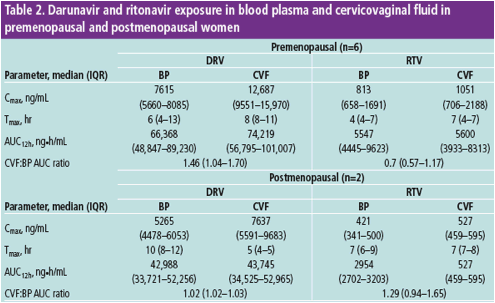
DRV, darunavir; RTV, ritonavir; IQR, interquartile range; BP, blood plasma; CVF, cervicovaginal fl uid; Cmax, maximum concentration in plasma; Tmax, time to maximum concentration; AUC, area under the curve
References
1. Squires K, et al. Poster Presented at the 5th International AIDS Society Conference on HIV Pathogenesis, Treatment and Prevention. July 19-22, 2009, Cape Town, South Africa. Poster MOPEB042
2. Rezk NL, et al. Talanta 2009; 79(5): 1372-8
3. Dumond JB, et al. AIDS 2007; 21: 1899-907
4. Dumond JB, et al. J Acquir Immune Defi c Syndr 2009; 51: 546-53
5. Jones A, et al. Presented at the 10th International Workshop on Clinical Pharmacology of HIV Therapy. April 15-17, 2009, Amsterdam, The Netherlands. Platform presentation
|
| |
|
 |
 |
|
|