 |
 |
 |
| |
(IDSA) Predictors of Response in GRACE (Gender, Race And Clinical Experience)
|
| |
| |
Reported by Jules Levin
47th Annual Meeting of the Infectious Diseases Society of America (IDSA), Philadelphia, Pennsylvania, USA, October 29 to November 1, 2009
Princy Kumar, MD1, Judith Currier, MD, MSc2, Kathleen Squires, MD3, Joseph Mrus, MD, MSc4, Bruce Coate, BS, MPH5, and Robert Ryan, MS5, on behalf of the GRACE Study Group
1Georgetown University School of Medicine, Washington, DC, USA; 2University of California, School of Medicine, Los Angeles, CA, USA; 3Jefferson Medical College of Thomas Jefferson University, Philadelphia, PA, USA; 4Tibotec Therapeutics, Bridgewater, NJ, USA; 5Tibotec, Inc., Yardley, PA, USA
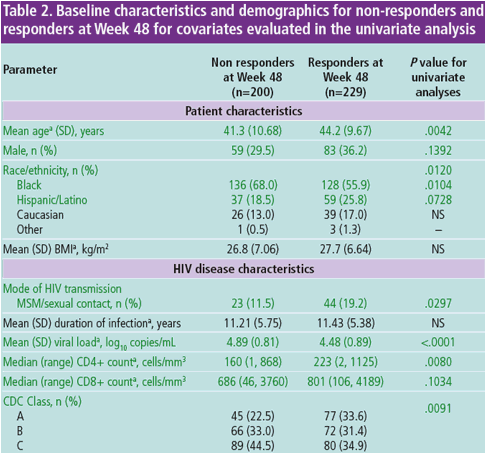
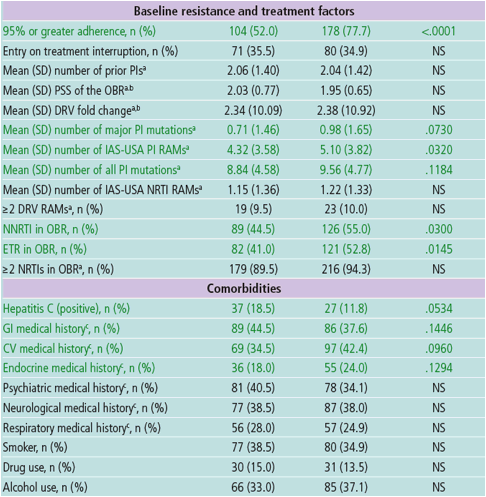
"Black race was correlated with lower response in the final multivariate model; however, in North America, race is likely a surrogate for other factors both measured and not measured in GRACE; Black patients had numerically lower adherence rates (63.3%) than Hispanics (66.7%) or Caucasians (73.9%), which may have contributed to their lower response rate. Black patients had more advanced disease at baseline than the other groups, which, among other potential reasons, could reflect differences in care and may have led to increased treatment complexity Lower response rates in blacks in GRACE may have been due to socioeconomic and other factors unique to North America. participation at a study site with limited experience in conducting a clinical trial and ETR in the OBR were predictors of improved response in GRACE These factors may include lower health literacy, more poverty or incarceration, or higher levels of depression. The HEAT study, another North-American based trial, also observed this correlation between race and virologic response"
AUTHOR CONCLUSIONS
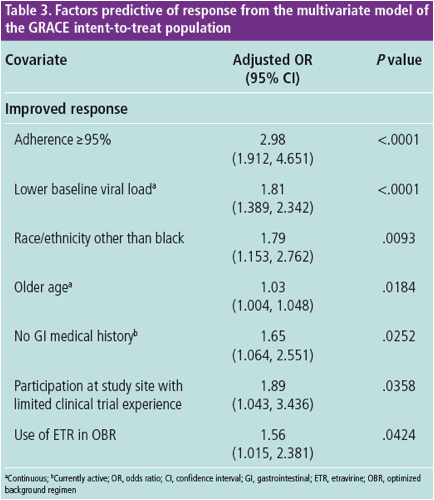
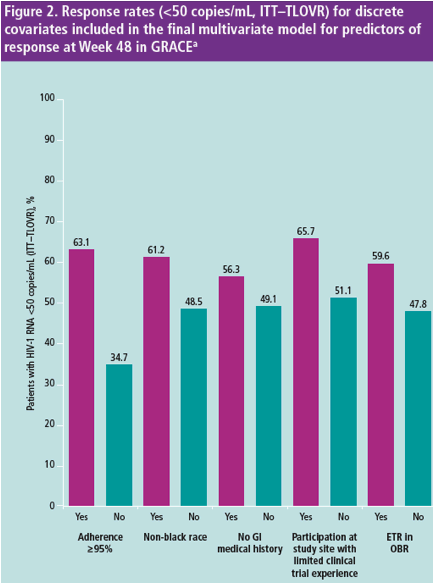
In the final multivariate model, ≥95% adherence, lower baseline VL, race/ ethnicity other than black, older age, no GI medical history, participation at
a study site with limited experience in conducting a clinical trial and ETR in
the OBR were predictors of improved response in GRACE
- Some of these covariates are likely surrogate markers for other variables
both measured and not measured in GRACE that could affect response
- Further investigation into factors that may impact race-based differences
in response rates, such as pharmacokinetics, differences in care,
socioeconomic disparities, health literacy and adherence, is warranted
Sex was not identified in the multivariate analysis as a significant predictor
of response to DRV/r plus an OBR at Week 48 in GRACE
AUTHOR DISCUSSION
The multivariate analysis performed on the overall GRACE population, which
accounted for differences in baseline characteristics, confirmed that sex was
not a significant predictor of response at Week 48
Adherence of ≥95%, and lower baseline VL were the covariates most
significantly correlated with response in the final multivariate model (P<.0001
for both), as has been previously observed in other HIV clinical trials3-6
- Poor adherence can lead to inadequate drug exposure and, in turn, VF and
the potential development of antiretroviral resistance7
- Paterson et al showed that >95% adherence to PI therapy resulted in
increased efficacy8
Older age was a significant predictor of response at Week 48 in GRACE,
mirroring reports from previous trials showing that younger age can be a
predictor of VF5,9
- Older age has been associated with improved adherence compared with
younger age in other studies10; however, age and adherence were not
strongly related in GRACE (data not shown) and, in the multivariate
model, the association between age and response was present even after
accounting for differences in adherence
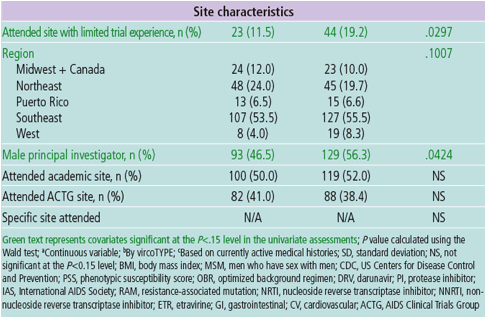
'Participation at a study site with limited experience in conducting clinical
trials' and 'no gastrointestinal (GI) medical history' may relate to more
complex personal characteristics, interactions or variables not fully captured
in GRACE
Association between use of ETR and improved response suggests that this
combination with DRV/r-based therapy is efficacious in treatment-experienced
patients with HIV-1; further interpretation is limited due to the lack of
randomization of ETR use in GRACE
Black race was correlated with lower response in the final multivariate model;
however, in North America, race is likely a surrogate for other factors both
measured and not measured in GRACE
- Black patients had numerically lower adherence rates (63.3%) than
Hispanics (66.7%) or Caucasians (73.9%), which may have contributed
to their lower response rate; however, in the multivariate model, the
association between black race/ethnicity and response was present even
after accounting for differences in adherence
- Black patients had more advanced disease at baseline than the other
groups, which, among other potential reasons, could reflect differences in
care and may have led to increased treatment complexity11, 12
- Lower response rates in blacks in GRACE may have been due to
socioeconomic and other factors unique to North America. These factors
may include lower health literacy, more poverty or incarceration, or higher
levels of depression11
- The HEAT study, another North-American based trial, also observed this
correlation between race and virologic response13
Background
·; GRACE, a multicenter, open-label, Phase IIIb study (Figure 1) was designed to
assess the efficacy and safety of darunavir/ritonavir (DRV/r) plus an investigator-selected optimized background regimen (OBR) in treatment-experienced women
infected with HIV-1 in comparison to treatment-experienced HIV-positive men1
- Enrollment into the study was planned to ensure that the racial/ethnic
distribution of the women in GRACE was representative of the demographics
of this population in the United States (US)1,2
- At each study site, three women were enrolled prior to enrollment of one
male patient1, and the number of Caucasian men enrolled was limited
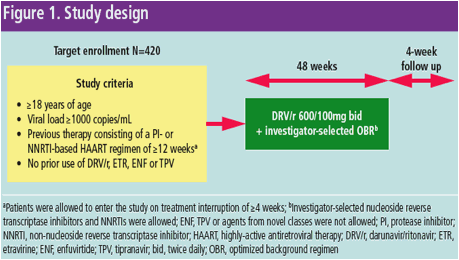
Virologic response (HIV-1 RNA <50 copies/mL) was calculated using a time-to-
loss of virologic response (TLOVR) algorithm in the intent-to-treat (ITT) population, which included patients who took at least one dose of study
medication, and in the non-virologic failure (VF) censored population, which
adjusted for discontinuations that were not due to VF
A total of 429 patients (women, n=287; men, n=142) were enrolled in GRACE and received at least one dose of study drug
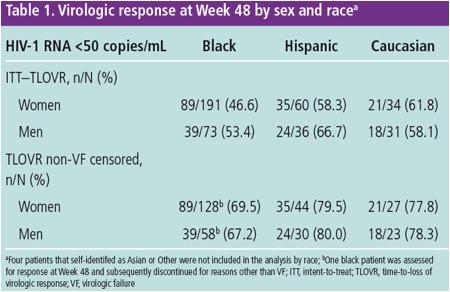
At Week 48, the virologic response rate was 53.4% in the ITT population and
73.2% in the non-VF censored population. The response rates at Week 48 by
sex and race are shown in Table 1
We present data from a post-hoc exploratory analysis, conducted to investigate factors predictive of virologic response in the ITT population at Week 48
- In particular, given the demographics of patients enrolled in GRACE, we
wanted to investigate whether sex or race were predictors of response




REFERENCES
1. Squires K, et al. GRACE (Gender, Race And Clinical Experience): 48-week
results of darunavir/r-based therapy in the largest trial in North America to
focus in treatment-experienced women. Presented at the 5th International
AIDS Society Conference on HIV Pathogenesis, Treatment and Prevention, Cape Town, South Africa, July 19-22, 2009. Poster MOPEB042
2. US Centers for Disease Control and Prevention. HIV/AIDS surveillance report.
Cases of HIV infection and AIDS in the United States and Dependent Areas,
2007, Vol. 19. Available from: http://www.cdc.gov/hiv/topics/surveillance/
resources/reports/2007report/pdf/2007SurveillanceReport.pdf. Accessed
September 14, 2009
3. Campo RE, et al. AIDS Res Hum Retroviruses 2009; 25(3): 269-75
4. Quiros-Roldan E, et al. AIDS Patient Care STDs 2007; 21(2): 92-9
5. Tuboi SH, et al. J Acquir Immune Defic Syndr 2005; 40(3): 324-8
6. Cahn P, et al. Curr Med Res Opin 2004; 20(7): 1115-23
7. Bangsberg DR, et al. AIDS 2006; 20: 223-31
8. Paterson DL, et al. Ann Intern Med 2000; 133(1): 21-30
9. Parienti JJ, et al. Clin Infect Dis 2004; 38(9): 1311-6
10. Maggiolo F, et al. HIV Clin Trials 2002; 3(5): 371-8
11. Cargill A and Stone V. Med Clin N Am 2005; 89: 895-912
12. Turner BJ, et al. Arch Intern Med 2000; 160: 2614-22
13. Smith KY, et al. Poster presented at the 5th International AIDS Society
conference on HIV pathogenesis, treatment and prevention. July 19-22, 2009,
Cape Town, South Africa. Poster MOPEB033
|
| |
|
 |
 |
|
|