 |
 |
 |
| |
144Week Efficacy & Safety of Raltegravir in TreatmentExperienced Patients, TripleClass Resistant
|
| |
| |
Reported by Jules Levin
Infectious Disease Societyof America (IDSA)
47th Annual Meeting
Philadelphia, PA
October 29 November 1, 2009
J.M. Gatell, MD1, B.Y. Nguyen, MD2, B. Grinsztejn, MD3, C. Katlama, MD4, J.J. Eron, MD5, A. Lazzarin, MD6, D. Vittecoq, MD7, C.J. Gonzalez, MD8, C. Lu, PhD2, C. Welebob, MS2, R.D. Isaacs, MD2, for the Protocol 005 Team
1University of Barcelona, Spain; 2Merck Research Laboratories, North Wales, PA; 3Evandro Chagas Clinical Research Institute, Oswaldo Cruz Foundation, Rio de Janerio, Brazil; 4Hopital PitieSalpetriere, Paris, France; 5University of North Carolina, Chapel Hill, NC; 6San Raffaele Science Institute, Milan, Italy; 7Hopital Paul Brousse, Villejuif, France; 8New York University School of Medicine, New York, NY

ABSTRACT
Background: RAL, an HIV integrase inhibitor, demonstrated potent efficacy in combination with optimized background therapy (OBT) during the doubleblind (DB) phase of Protocol 005, a phase II study in patients failing therapy with tripleclass resistant virus. The study extension provides longterm data on RAL.
Methods: After ≥24 weeks of DB therapy (RAL 200, 400, or 600 mg bid or placebo), all patients were offered openlabel (OL) RAL 400 mg b.i.d. Efficacy measurements included % patients with HIV RNA (vRNA) <50 copies/mL and change from baseline in CD4 cells.
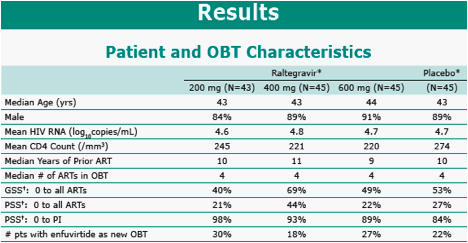
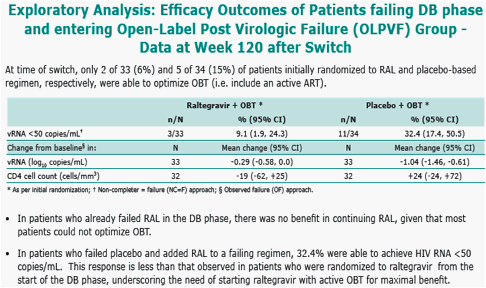
Results: 133 pts received RAL and 45 placebo. Baseline characteristics demonstrated heavily pretreated patients (∼10 years prior ARTs); >60% of patients had OBT with limited activity (Genotypic Sensitivity Score = 0). All RAL dose groups, including 94 patients (71%) who entered OL extension, were combined since all dose levels were efficacious during DB therapy; placebo group data are not reported since only 6 patients (13%) entered OL. Efficacy is summarized below:
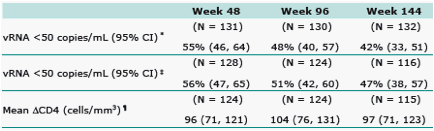
NonCompleter=Failure
Observed Failure
Baseline values carried forward for virologic failures.
RAL was generally well tolerated with few discontinuations (5 patients) due to adverse events.
Conclusions: In patients with limited treatment options, RAL in combination with OBT had potent and durable antiretroviral effect through week 144, and was generally well tolerated.



* All treatment groups also received OBT [investigational ARTs at the time (e.g. darunavir, tipranavir) were not permitted]
GSS/PSS = Genotypic/Phenotypic sensitivity score by Phenosense GT.
Enfuvirtide use in OBT in enfuvirtide naïve patients was counted as one active drug in OBT and added to the GSS/PSS.
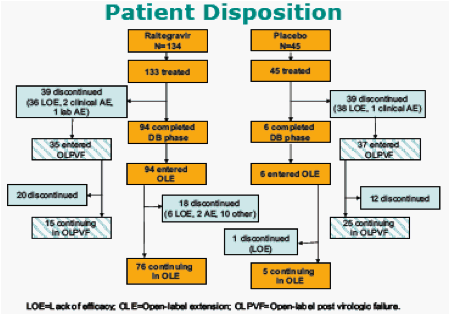
The median duration of DB phase was 40 weeks for the Raltegravir groups and 24 weeks for the placebo group. By Week 48, > 85% of patients had switched to openlabel Raltegravir (OLE).
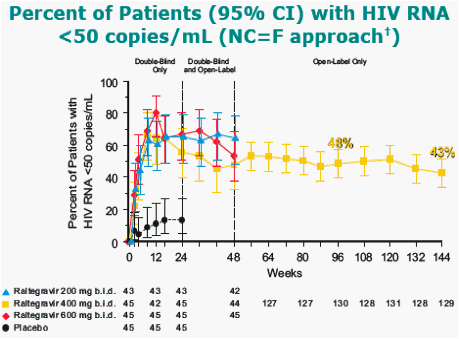
All treatment groups also received OBT [investigational ARTs at the time (e.g. darunavir, tipranavir) were not permitted].
Noncompleter equals failure (NC=F) approach treats all discontinuations as failures.
At week 144, 5 of the 6 patients from the placebo group who switched to RAL 400 mg had HIV1 RNA < 50 copies/mL.
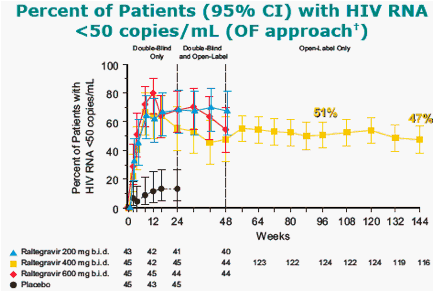
All treatment groups also received OBT [investigational ARTs at the time (e.g. darunavir, tipranavir) were not permitted].
In the Observed Failure (OF) approach, only discontinuations due to lack of efficacy are counted as failures afterwards.
At week 144, 5 of the 6 patients from the placebo group who switched to RAL 400 mg had HIV1 RNA <50 copies/mL.
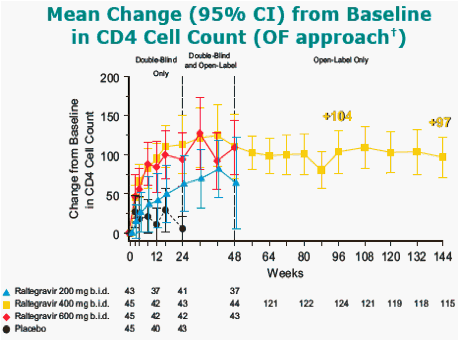
All treatment groups also received OBT [ investigational ARTs at the time (e.g. darunavir, tipranavir) were not permitted].
In the Observed Failure (OF) approach, only discontinuations due to lack of efficacy are counted as failures afterwards and baseline value was carried forward.
At week 144, the 6 patients from the placebo group who switched to RAL 400 mg had mean change from baseline in CD4 cell count about 135 cells/mm3
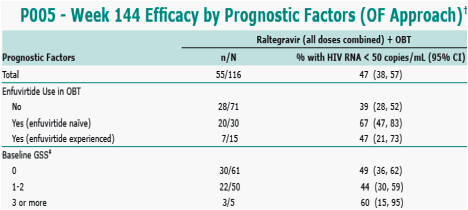
In the Observed Failure (OF) approach, only discontinuations due to lack of efficacy are counted as failures afterwards; GSS was defined as total number of drugs in OBT to which a patient's viral isolate showed genotypic sensitivity. Enfuvirtide use in enfuvirtidenaïve patients was counted as one active drug and added to the GSS.
n/N = (number of responders) / (number of patients) in each category group.
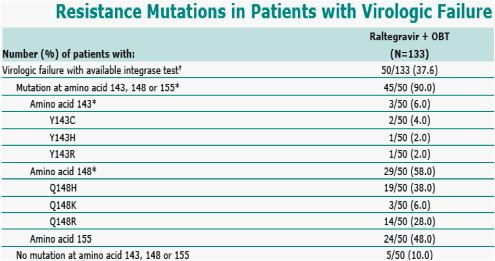
Virologic failure was defined as (1) nonresponse (did not achieve >1.0 log viral reduction or <400 copies/mL) by week 16, or (2) viral rebound starting at week 16 or later, defined as: (a) HIV RNA >400 copies/mL (on 2 consecutive measurements at least 1 week apart) after initial response with HIV RNA <400 copies/mL or (b) >1.0 log increase in HIV RNA above nadir level (on 2 consecutive measurements at least 1 week apart).
* Patients may be counted in multiple subcategories due to the appearance of multiple mutations.
Six (6) additional VF patients without available integrase test are not included in the table.
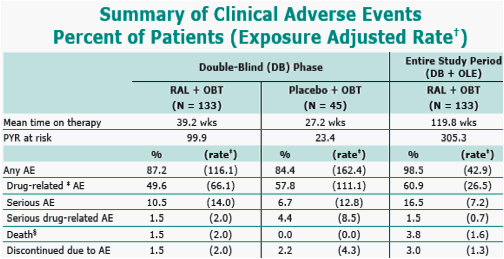
per 100 personyears (PYR).
determined by investigator to be possibly, probably, or definitely related to RAL or placebo (alone or in combination with OBT).
Causes of death include: laceration, suicide; bradycardia, cardiorespiratory arrest, sepsis, shock; abdominal adenomegaly, splenic abscess; and myocardial infarction (2); none of the 5 deaths were drugrelated.
Six patients initially randomized to placebo received RAL in OLE; no deaths or serious drugrelated AEs occurred in these patients; 1 patient had 2 serious AEs (Bell's palsy and headache) that were not drugrelated.
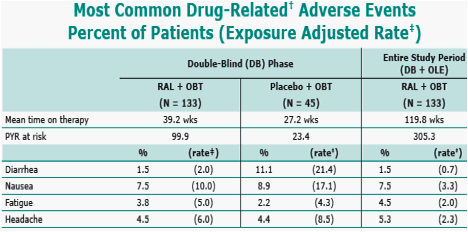
determined by investigator to be possibly, probably, or definitely related to RAL or placebo (alone or in combination with OBT); incidence ≥5% in at least one group; per 100 personyears (PYR).
Six patients initially randomized to placebo received RAL in OLE; remaining placebo patients discontinued during DB phase.
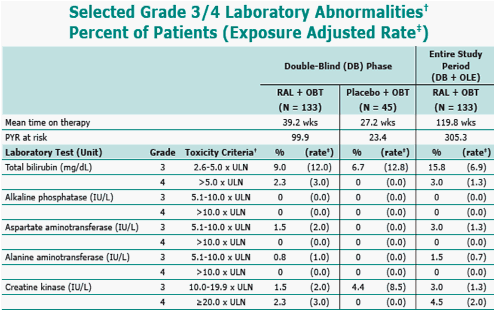
Division of AIDS grading scale, December 2004. per 100 PYR. ULN = upper limit of normal.
Six patients initially randomized to placebo received RAL in OLE; remaining placebo patients discontinued during DB phase.
|
| |
|
 |
 |
|
|