 |
 |
 |
| |
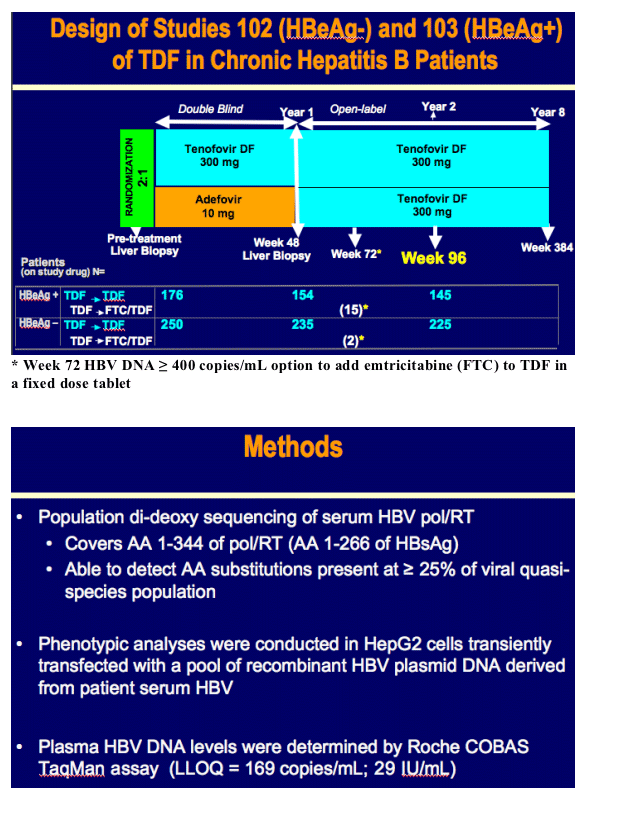
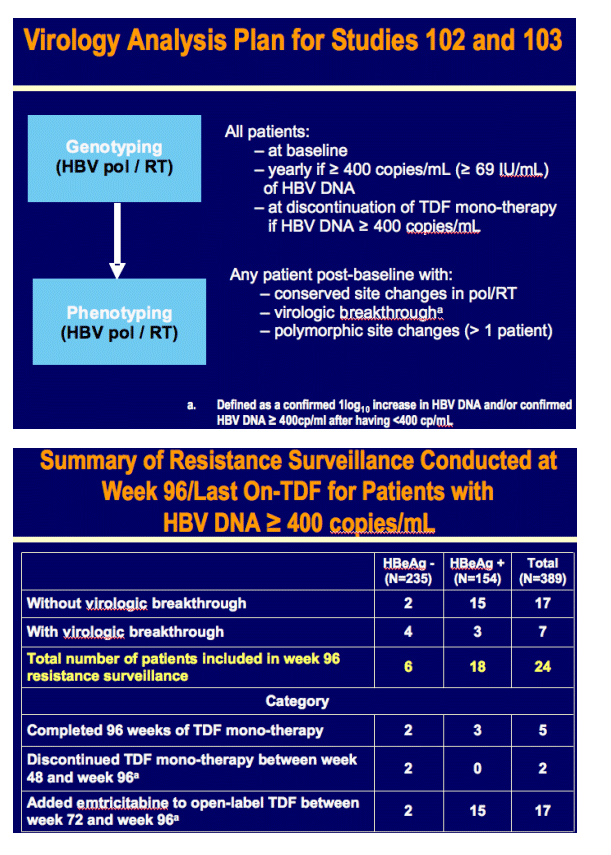
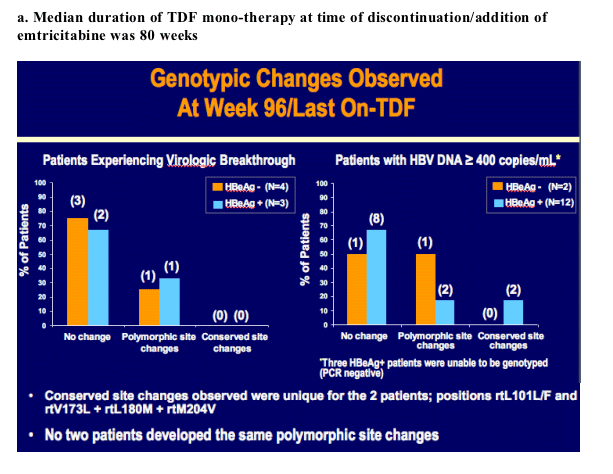
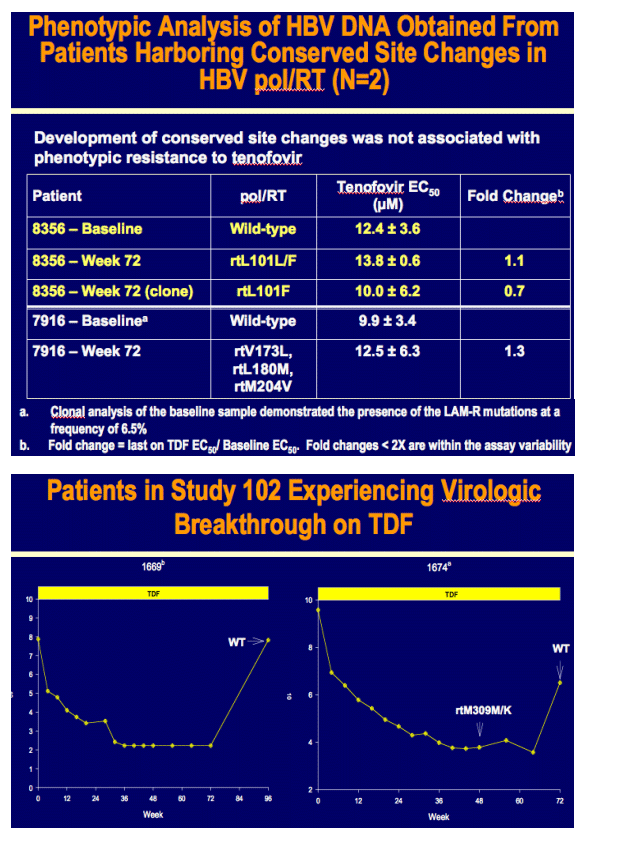
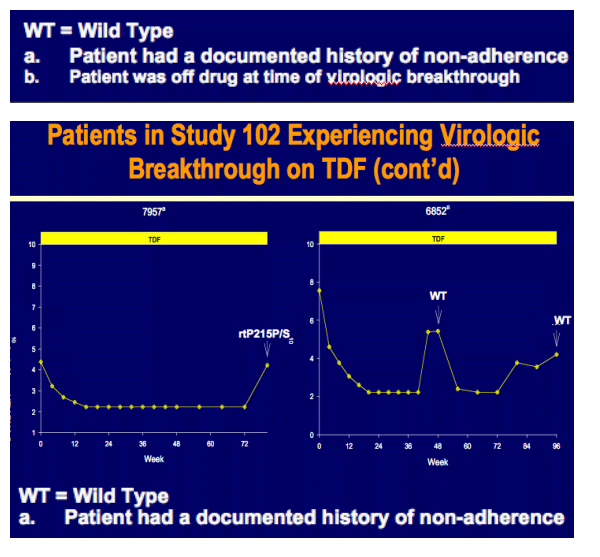
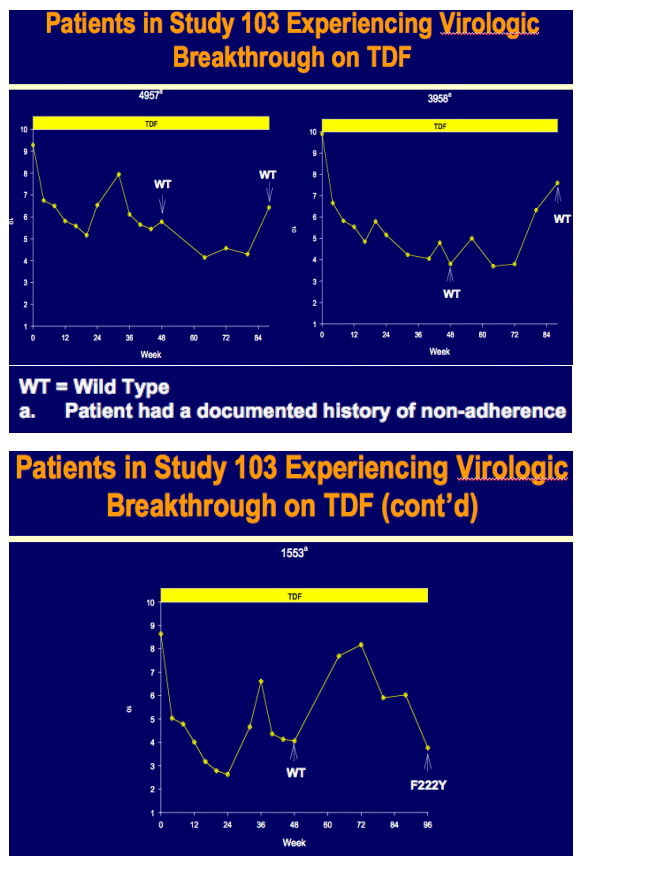
Week 96 Resistance Surveillance for HBeAg Positive and Negative Subjects with Chronic HBV Infection Randomized to Receive Tenofovir DF 300 mg QD
|
| |
| |
Reported by Jules Levin
13th ISVHLD Wash Dc March 20-24 2009
A Snow-Lampart1, B Chappell1, M Curtis1, Y Zhu1, J Heathcote2,
P Marcellin3, K Borroto-Esoda1
1Gilead Sciences Inc., Durham, NC, USA; 2University of Toronto, Toronto ON, Canada; 3University of Paris, Hospital Beaujon, Clichy, France



|
| |
|
 |
 |
|
|