 |
 |
 |
| |
The HIV-1 RT Mutant Q151L Shows Decreased
Replication Capacity, Selective High-Level
Resistance to GS-9148 and Hypersusceptibility
to Tenofovir and Zidovudine
|
| |
| |
XVIII International HIV Drug Resistance Workshop
June 9-13, 2009
Fort Myers, Florida
Rima Kulkarni, Nicolas Margot, Florence Myrick, Jenny Svarovskaia, James
Chen, Eric Lansdon, Adrian Ray, Tomas Cihlar, S Swaminathan, Michael Miller,
and Kirsten White
Gilead Sciences, Inc., Foster City, CA, and Durham, NC, USA
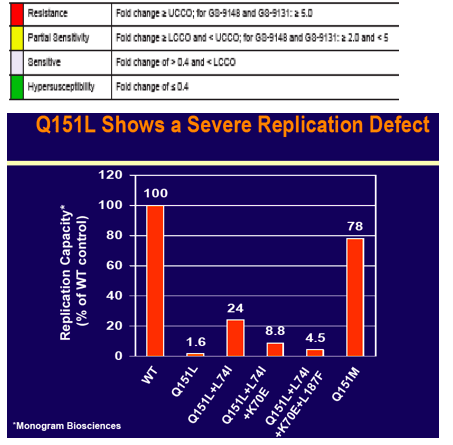
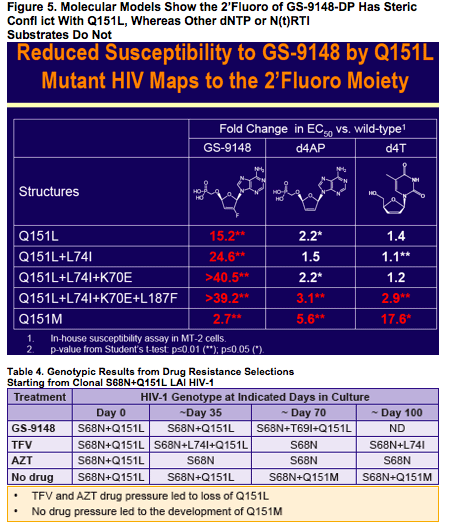
"The investigational nucleotide reverse transcriptase inhibitor (NRTI) GS-9148 (GS-9131 is prodrug) maintains potent activity against a majority of clinically observed resistance mutations". "The hyperssusceptibility of Q151L to tenofovir and AZT suggest potential utility of combination therapy of GS-9131 with these agents". From Jules: it appears the drug is potentially active against tenofovir resistance, is more potent than tenofovir, has less if any renal side effects, and tenofovir might be active against GS-9148 resistance. This drug has potential use for patients. Gilead has placed development of this drug on hold.
AUTHOR CONCLUSIONS

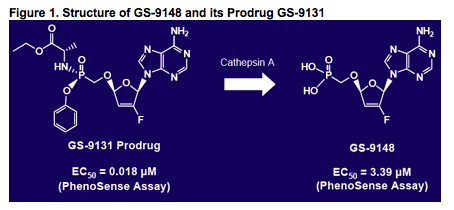
Introduction
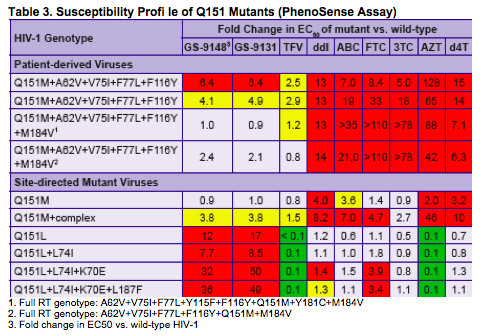
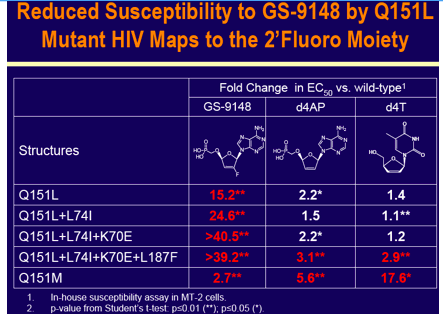
GS-9148 (Fig. 1) is a novel nucleotide HIV RT inhibitor (NtRTI) that
maintains potent activity against a majority of clinically observed
resistance mutations including M184V, K65R, L74V, and ≥ 4 TAMs1
GS-9148 has low-level cross-resistance to Q151M + complex
GS-9131 (Fig. 1), an orally bioavailable phosphonoamidate prodrug
of GS-9148, was designed to effi ciently enter PBMCs in vivo and then
release GS-9148 intracellularly2
GS-9131 has completed initial phase I evaluation in HIV-1 infected
patients
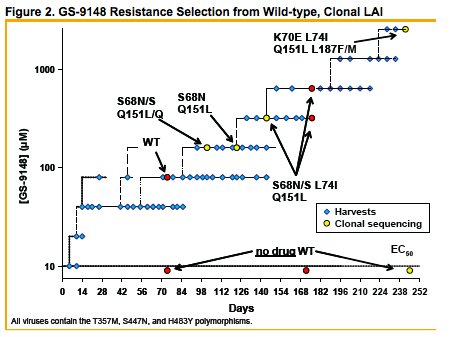
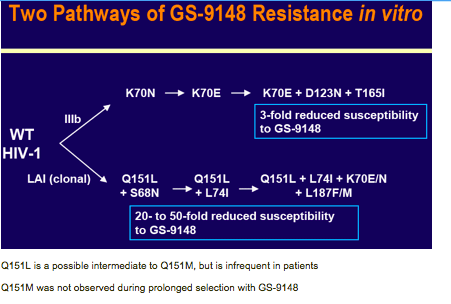
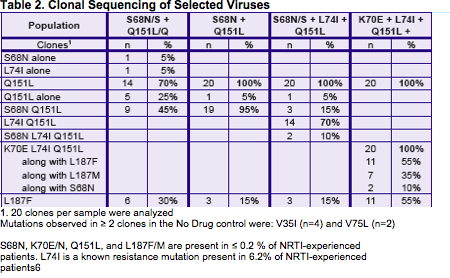
A GS-9148 resistance selection with HIV-1 IIIB previously resulted in
K70E+D123N+T165I RT mutations and low-level reduced susceptibility
to GS-91487
OBJECTIVES
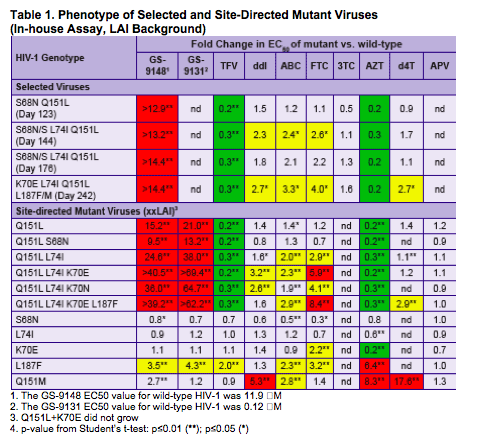
To further characterize the resistance that develops to GS-9148 in vitro
To evaluate the cross-resistance of the selected mutations to other
N(t)RTIs
To characterize the developing resistance mutations for their impact
on viral replication capacity

References
1. Cihlar, et al., Antimicrobial Agents and Chemotherapy (2008) 52(2):655-665.
2. Ray, et al., Antimicrobial Agents and Chemotherapy (2008) 52(2):648-654.
3. Petropoulos, et al., Antimicrobial Agents and Chemotherapy (2000) 44(4):920-928.
4. Lansdon, et al., CROI (2009) Abstract # 66LB.
5. Huang, et al., Science (1998) 282:1669-1675.
6. Stanford HIV database: http://hivdb.stanford.edu/index.html. Database accessed on May 1, 2009.
7. Lafl amme, et al., ICAAC (2007) Poster # H-1037.
8. Kosalaraksa, et al., Journal of Virology (1999) 73(7): 5356-5663.
We would like to thank Eric Lam and Monogram Biosciences for their contributions. We would also like to thank
Bob Shafer and the Stanford HIV Database.
|
| |
|
 |
 |
|
|