 |
 |
 |
| |
Confirmation of the negative impact of protease mutations I47V, I54M, T74P and I84V and the positive impact of protease mutation V82A on virological response to darunavir/ritonavir
|
| |
| |
Reported by Jules Levin
18th Intl HIV Drug Resistance Workshop
June 9-12 Ft Myers Florida
S De Meyer,1 D Descamps,2 B Van Baelen,1 E Lathouwers,1 A Cheret,3 A-G Marcelin,4 V Calvez,4 G Picchio5
1Tibotec BVBA, Mechelen, Belgium; 2Groupe Hospitalier Bichat-Claude Bernard and Universite Denis Diderot-Paris 7, Paris, France; 3Tibotec, une Division de Janssen-Cilag, Issy les Moulineaux, France;
4Groupe Hospitalier Pitie-Salpêtriere and UPMC Univ 06 UMR_S 943, Paris, France; 5Tibotec Inc., Yardley, PA, USA
AUTHOR CONCLUSIONS
The negative impact of protease mutations I47V, I54M, T74P and I84V and the positive impact of protease mutation V82A on virological response to DRV/r was confirmed in two independent datasets: in the POWER/DUET dataset (this analysis) and in the Descamps et al dataset.6
The impact of protease mutations K14R, K20I, E34Q, E35D and K55R on response to DRV/r was not confirmed in this POWER/DUET dataset.
The presence of the V82A mutation in patients with three DRV RAMs was associated with a virological response comparable to that observed in patients with two DRV RAMs.
INTRODUCTION
The protease inhibitor (PI) darunavir (DRV; TMC114) has shown significant, broad-spectrum in-vitro antiretroviral (ARV) activity against both wild-type and multidrug-resistant HIV-1 strains.1
DRV combined with low-dose ritonavir (DRV/r) has shown significant ARV activity and tolerability in a wide range of adult patients, from treatment-naïve to treatment-experienced patients.2-4
Analyses in 1071 treatment-experienced patients who initiated treatment with DRV/r 600/100mg bid in the POWER 1, 2 and 3 studies (n=467) and in the placebo groups of the DUET-1 and DUET-2 studies (n=604), showed a diminished response* when 33 (genotypic cut-off) of 11 DRV resistance-associated mutations (RAMs) (V11I, V32I, L33F, I47V, I50V, I54L/M, T74P, L76V, I84V and L89V) were present at baseline.5
Using a different dataset (n=153), Descamps et al identified eight mutations (K14R, K20I, E34Q, I47V, I54M, K55R, T74P and I84V) that had a negative impact on virological response and two mutations (E35D and V82A) that had a positive impact on response.6
In this present analysis, the combined POWER (DRV/r 600/100mg bid group) and DUET (placebo group) dataset was used to evaluate the significance of the DRV/r RAMs identified by Descamps et al.
*Diminished response was defined as a response rate that was <75% of the overall response rate.
METHODS
Patients
The analysis was performed on patients who initiated treatment with DRV/r 600/100mg bid in POWER 1, 2 and 3 and in the placebo groups of DUET-1 and DUET-2 (POWER and DUET dataset).
The POWER studies are randomized, Phase IIb trials designed to investigate the efficacy and safety of DRV/r plus an optimized background regimen (OBR) in treatment-experienced patients.4
The DUET-1 and DUET-2 studies are two, randomized, placebocontrolled, double-blind, Phase III trials investigating the efficacy and safety of the NNRTI etravirine (TMC125), as part of a regimen including DRV/r, investigator-selected NRTIs and optional enfuvirtide (ENF) in highly treatment-experienced patients.7,8
All patients included at the time of the database lock for the Week 96 analysis of the POWER trials and for the primary Week 24 analysis of the DUET trials were included in these analyses.7,8
Baseline genotype data were available for 1060 patients. Baseline genotype and DRV phenotype data were available for 1035 patients.
Virological assessments
Genotypes and phenotypes of plasma viruses were determined using population-based sequencing and Antivirogram, respectively (Virco BVBA, Mechelen, Belgium).
Virological response was assessed as change in HIV-1 RNA at Week 24 from baseline (non-completer=failure [NC=F] analysis) and proportion of patients achieving HIV-1 RNA <50 copies/mL at Week 24 (time-to-loss-of-virological-response [TLOVR] analysis), censoring discontinuations for reasons other than virological failure (VF) (non-VF censored) at time points after the last available on-treatment data.
An analysis of variance (ANOVA) model including the covariates: baseline HIV-1 RNA, baseline CD4 cell count, number of susceptible NRTIs in the OBR, ENF use (naïve, not used/re-used), individual DRV RAMs and protease mutations as identified by Descamps et al, was performed to determine whether these mutations were significant predictors of change in HIV-1 RNA (NC=F non-VF censored).
RESULTS
The impact of K14R, K20I, E34Q, E35D, I47V, I54M, K55R,
T74P, V82A and I84V on virological response to DRV/r
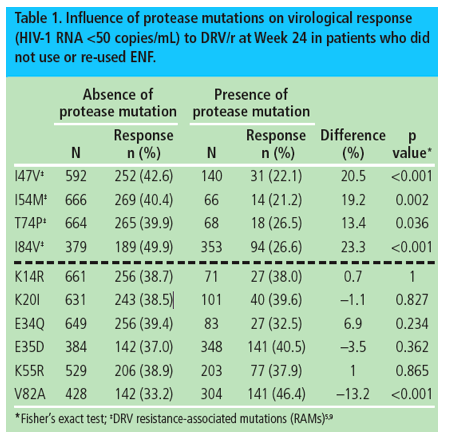
Four of the mutations identified by Descamps et al (I47V, I54M, T74P and I84V) were already described as DRV RAMs using the POWER/DUET dataset.5 These mutations had a significantly negative association with virological response to DRV/r (Table 1).
Out of the other mutations identified by Descamps et al, V82A had a significantly positive association with virological response to DRV/r, while K14R, K20I, E34Q, E35D and K55R were not associated with virological response to DRV/r (Table 1).
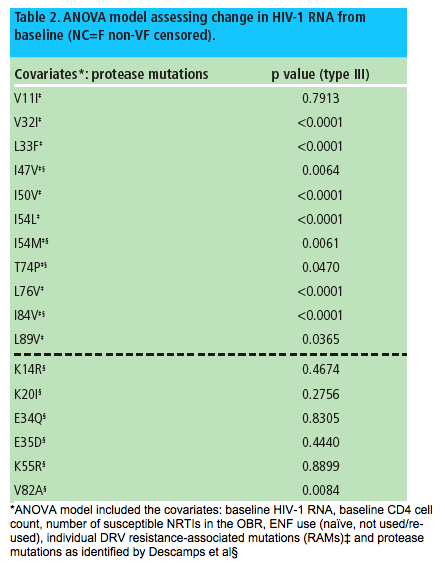
Analysis of the mutations identified by Descamps et al in an ANOVA model assessing change in HIV-1 RNA, that included the covariates described above, showed that I47V, I54M, T74P and I84V (previously identified as DRV RAMs)5 and V82A were significantly associated with change in HIV-1 RNA, while this was not the case with K14R, K20I, E34Q, E35D and K55R (Table 2).
V82A mutation
The impact of V82A on virological response to DRV/r
Overall, in patients who did not use or re-used ENF, the virological response (<50 copies/mL) at Week 24 was 46.4% (141/304) in patients with the V82A mutation at baseline vs 33.2% (142/428) in patients without V82A at baseline (p<0.001; Table1).
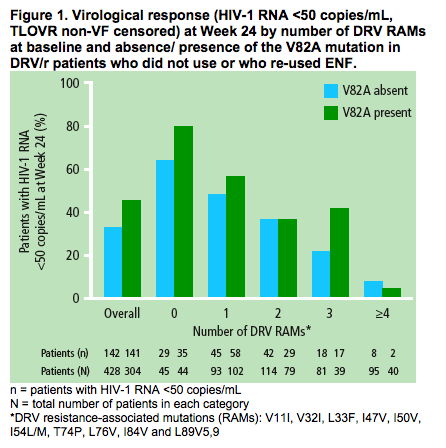
The impact of the presence of the V82A mutation at baseline on Week 24 virological response to DRV/r by number of baseline DRV RAMs is shown (Figure 1)
- the virological response rate decreased as the number of baseline DRV RAMs increased
-- patients with three DRV RAMs at baseline had a higher virological response to DRV/r when the V82A mutation was present at baseline versus when it was absent. The response rate of patients with three DRV RAMs and a V82A was comparable to that observed in patients with two DRV RAMs.
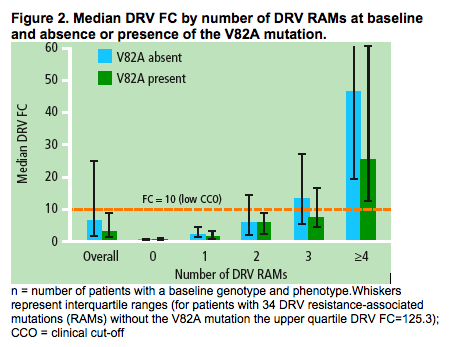
The impact of V82A on DRV fold-change in EC50 value (FC)
Overall, the median DRV FC from baseline samples with genotype and phenotype data (n=1035) was 3.5 vs 6.8 for patients with versus without the V82A mutation at baseline, respectively.
The median DRV FC by number of DRV RAMs and by absence or presence of V82A is shown (Figure 2)
- the median DRV FC increased as the number of baseline DRV RAMs increased
-- in patients with three baseline DRV RAMs and the V82A mutation, the median DRV FC was 7.6, which was below the clinical cut-off of 10, however, the FC value was 13.1 in this subgroup of patients when the V82A mutation was absent.
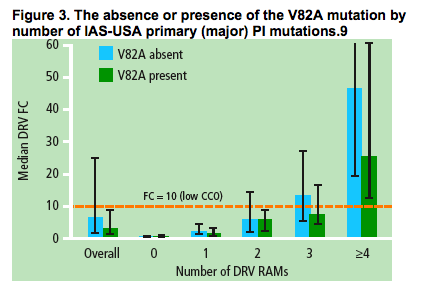
The presence of V82A by number of IAS-USA primary (major) PI
mutations
The V82A mutation was most prevalent together with other IAS-USA primary (major) PI mutations9 (Figure 3).
REFERENCES
1. De Meyer S, et al. Antimicrob Agents Chemother 2005;49:2314-21.
2. Ortiz R, et al. AIDS 2008;22:1389-97.
3. Madruga JV, et al. Lancet 2007;370:49-58.
4. Clotet B, et al. Lancet 2007;369:1169-78.
5. De Meyer S, et al. 6th European HIV Drug Resistance Workshop, Budapest, Hungary, 26-28 March 2008. Abstract 54.
6. Descamps D, et al. J Antimicrob Chemother 2009;63:585-92.
7. Madruga JV, et al. Lancet 2007;370:29-38.
8. Lazzarin A, et al. Lancet 2007;370:39-48.
9. Johnson VA, et al. Top HIV Med 2008;16:138-45.
|
| |
|
 |
 |
|
|