 |
 |
 |
| |
Correlation between laboratory parameters and use or discontinuation of nucleoside analogues in the MONET trial
|
| |
| |
Reported by Jules Levin
11th Workshop on Adverse Drug Reactions and Co-morbidities in HIV, Philadelphia, USA, October 2009
Andrew Hill, University of Liverpool, UK, Yvon van Delft, Janssen-Cilag EMEA, Tilburg, The Netherlands, Christiane Moecklinghoff, Janssen-Cila gEMEA, Neuss, Germany
AUTHOR CONCLUSIONS
In the MONET trial, changes in several laboratory parameters were correlated with use or discontinuation of nucleoside analogues.
Use of tenofovir was associated with lower levels of total cholesterol, but higher levels of creatinine, alkaline phosphatase and parathyroid hormone.
Use of zidovudine was associated with lower Red Blood Cell counts, White Blood Cell counts and haemoglobin.
Overall in the trial, elevations in liver enzymes were correlated with chronic or acute infection with Hepatitis A or C.
INTRODUCTION
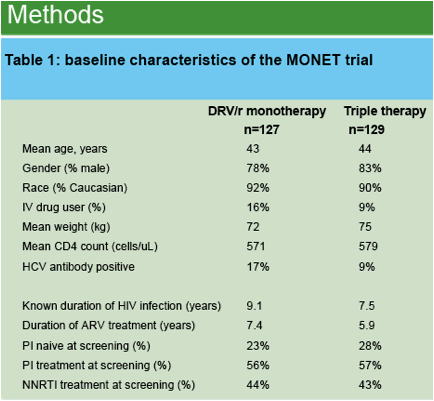
The MONET trial evaluated 48 weeks of treatment with darunavir/ritonavir (DRV/r) 800/100 mg OD, either as monotherapy or with two nucleoside analogues (triple therapy). The trial enrolled 256 patients with HIV RNA <50 copies/mL at screening and no history of virological failure.
In the DRV/r monotherapy arm, patients stopped taking all nucleoside analogues at baseline. In the triple therapy arm, patients were allowed to either continue taking NRTIs taken at screening, or re-optimise their NRTIs at baseline.
After 48 weeks of treatment, the percentage of patients with HIV RNA <50 copies/mL was 86.2% in the monotherapy arm versus 87.8% in the triple therapy arm, proving non-inferior efficacy for DRV/r monotherapy.
Nucleoside analogues are known to have effects on laboratory parameters: for example, zidovudine treatment lowers haemoglobin and white blood cell counts, while starting tenofovir slightly lowers cholesterol levels, but raises creatinine levels, alkaline phosphatase and parathyroid hormone.
Therefore, the effects of starting or stopping NRTIs on laboratory parameters was assessed in the MONET trial.
Full blood counts and clinical chemistry were measured at screening, baseline and at each study visit to Week 48. Parathyroid hormone was measured only at Week 48.
In the DRV/r monotherapy arm (n=127), 42 patients stopped zidovudine at baseline, while 45 patients stopped tenofovir; 40 patients stopped taking abacavir. These NRTIs had been used almost exclusively in combination with 3TC or FTC.
In the triple therapy arm (n=129), 44 patients continued tenofovir from their pre-baseline HAART, 26 patients started tenofovir, 14 patients continued zidovudine, and 45 took abacavir. These NRTIs were almost exclusively used with 3TC or FTC.
Analysis of covariance was used to assess the effects of NRTI use on changes in each laboratory parameter during the trial, adjusting for the baseline values. Analyses were conducted in SAS.

Total Cholesterol
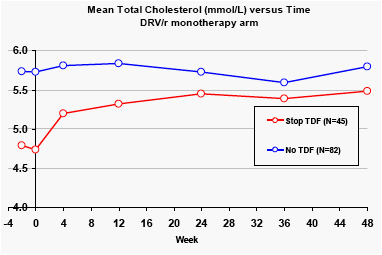
In the DRV/r monotherapy arm, patients who stopped taking tenofovir at baseline showed rises in total cholesterol by Week 4, of approximately 0.5 mmol/L (25 mg/dL). Patients who had not received tenofovir before the trial had higher baseline levels of total cholesterol, which remained stable after stopping these nucleoside analogues.
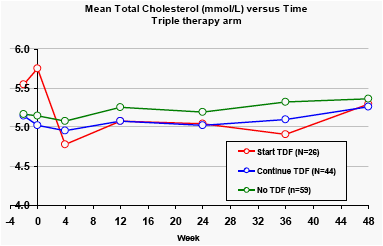
At Week 48, patients who started tenofovir at baseline in the triple therapy arm showed mean falls in total cholesterol of 0.5 mmol/L (25 mg/dL). There were also falls in triglyceride levels for patients who started tenofovir.
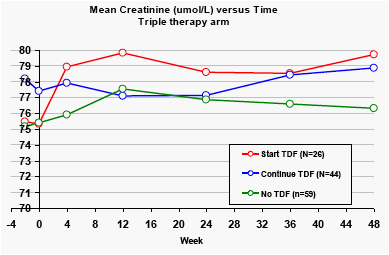
Creatinine
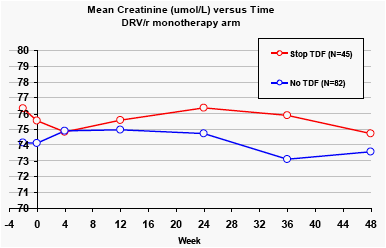
In the DRV/r monotherapy arm, there was a small reduction in creatinine and increases in creatinine clearance for patients who stopped tenofovir.
In the triple therapy arm, patients who started taking tenofovir at baseline showed significant rises in creatinine levels. The mean creatinine levels at Week 48 were highest for patients taking tenofovir versus other nucleoside analogues (p<0.01).
Parathyroid hormone
Parathyroid hormone levels fell from a peak in April (mean 7.21pg/mL) to a minimum in August (mean 3.55pg/mL), and were significantly higher for patients receiving tenofovir (mean 6.09pg/mL) versus no tenofovir (mean 4.56pg/mL).
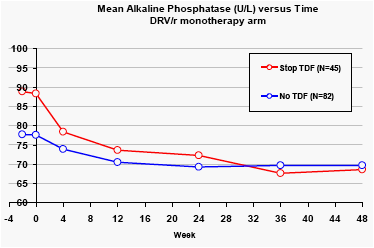
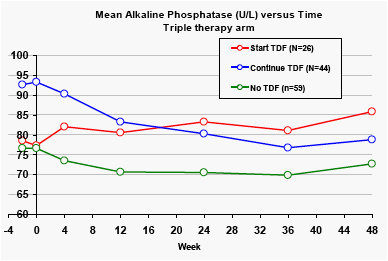
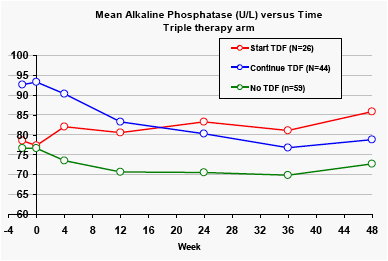
Alkaline phosphatase
Across the two treatment arms, the mean baseline levels of alkaline phosphatase were significantly higher for patients taking tenofovir at baseline (p<0.01). Patients in the DRV/r monotherapy arm who stopped taking tenofovir had significant reductions in alkaline phosphatase levels.
In the triple therapy arm, the alkaline phosphatase levels were highest in the groups receiving tenofovir, but there was substantial variation in these levels over time.
Red Blood Cells
In the DRV/r monotherapy arm, patients who stopped taking zidovudine at baseline showed significant rises in Red Blood Cell count (p<0.001).
Patients in the triple therapy arm who switched from zidovudine to other nucleoside analogues at baseline also showed rapid rises in Red Blood Cell count (p<0.01). Patients who continued to take zidovudine showed persistently low mean Red Blood Cell counts, compared with patients taking other nucleoside analogues. In the triple therapy arm, 42.6% had at least one Red Blood Cell (RBC) result below the lower limit of normal (<4.12 x 1012/L), versus 22.8% in the monotherapy arm (p<0.01).
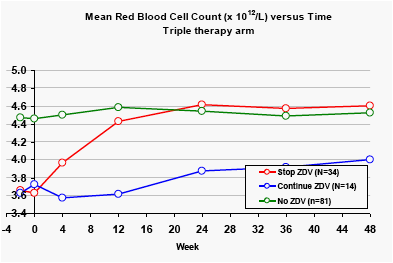
Similar trends were seen for haemoglobin and White Blood Cell Count (data not shown)
Liver enzymes
Six patients in the monotherapy arm and 2 in the triple therapy arm had Grade 3 elevations in ALT and/or AST: these patients all had either acute infection with Hepatitis C (2 cases), presence of Hepatitis C antibodies (5 cases) or acute Hepatitis A infection (1 case).
|
| |
|
 |
 |
|
|