 |
 |
 |
| |
Both once-daily saquinavir / ritonavir and atazanavir / ritonavir, when combined with tenofovir / emtricitabine conserve adipose tissue, only modestly affect lipids and exhibit similar mild reductions in glomerular filtration rate over 48 weeks: the BASIC trial
|
| |
| |
Reported by Jules Levin
11th International Workshop on Adverse Drug Reactions and Co-morbidities in HIV, 26-28 October 2009, Philadelphia, USA
SME Vrouenraets1,2, E Fernandez Garcia2, A Jackson3, F Raffi4, DT Jayaweera5, C Katlama6, M Fisher7, L Slama8, D Hardy9, S Mauss10, E DeJesus11, A van Eeden12, D Prelutsky13, FWNM Wit1,2, G Moyle3 and P Reiss1,2 for the BASIC Study Group
1 Academic Medical Center, Amsterdam, the Netherlands; 2 IATEC, Amsterdam, the Netherlands; 3 Chelsea and Westminster Hospital, London, UK; 4 CHU Hotel Dieu, Nantes, France; 5 University of Miami School of Medicine, Miami, USA; 6 Hopital Pitie-Salpetriere, Paris, France; 7 Royal Sussex County Hospital, Brighton, England; 8 Hopital Tenon, Paris, France; 9 Cedars-Sinai Medical Centre, Los Angeles, USA;
10 Gemeinschaftspraxis Mauss, Dusseldorf, Germany; 11 Orlando Immunology Center, Orlando, USA; 12 St. MC Jan van Goyen, Amsterdam, the Netherlands and 13 Central West Clinical Research, St Louis, USA
This investigator-initiated study was supported by an unrestricted scientific grant from Hoffmann la Roche and by supply of fixed dose TDF/FTC from Gilead Sciences
AUTHOR CONCLUSIONS
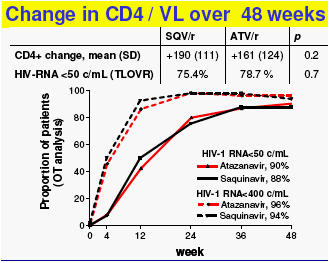
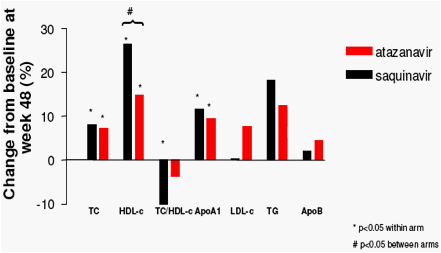
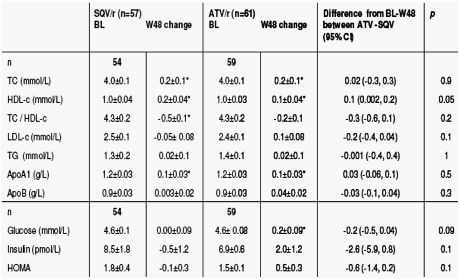
Once-daily SQV/r 2000/100mg and ATV/r 300/100mg, combined with TDF/FTC as initial therapy for HIV-1 infection were virologically effective and had comparable modest and potentially favourable effects on lipids and no effect on insulin sensitivity.
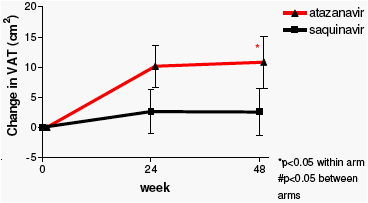
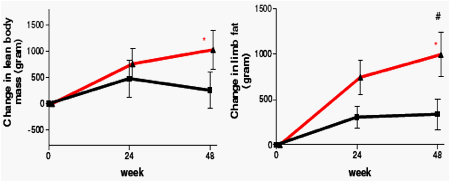
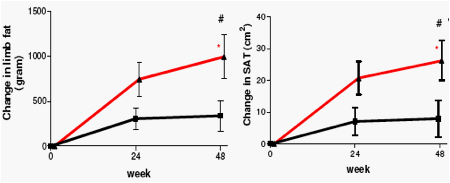
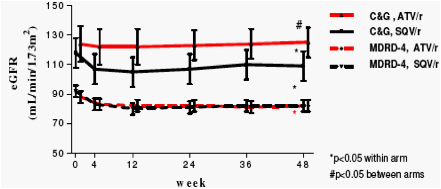
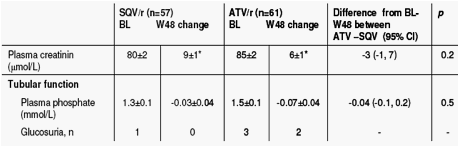
Neither treatment regimen was associated with loss of adipose tissue. In contrast, in patients on ATV/r fat mass increased significantly in all compartments, with the increase in both limb fat and abdominal subcutaneous fat being significantly greater when compared to in SQV/r-treated patients. eGFR decreased modestly in both treatment arms in the first months but remained stable thereafter. The difference between treatment arms in total and lean body mass changes may explain the discrepancy between GFR estimations by C-G and MDRD-4, respectively.
Both treatment combinations may represent a valid choice for initial HIV treatment particularly in patients at high risk for cardiovascular disease.
INTRODUCTION
Potential metabolic and renal adverse effects are important factors to be taken into account when choosing an optimal antiretroviral treatment regimen for an individual patient. The effects of once-daily (OD) ritonavir boosted saquinavir (SQV/r) and atazanavir (ATV/r) in combination with tenofovir/emtrictabine (TDF/FTC) on lipids, glucose metabolism, body composition and renal function have not yet been directly compared.
METHODS
BASIC (Boosted Atazanavir or Saquinavir Induced lipid Changes) is a 48-week, investigatorinitiated, randomized multinational trial comparing:
- SQV/r 2000/100 mg once daily with TDF/FTC
- ATV/r 300/100 mg once daily with TDF/FTC
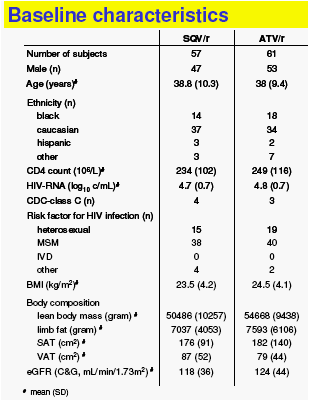
118 of the 123 randomized treatment-naïve HIV-1 infected patients were included in the ITT-analysis, because they had received at least one dose of study drug; this includes 32 participants of the 24 week SSAR 2004/0002 study with the same design conducted at the Chelsea & Westminster site
(UK). The SSAR 2004/0002 study was extended to 48 weeks to co-enrol these participants in the
BASIC study. Study labs were not drawn at the week 12 timepoint in these patients.
Fasting lipids and glucose metabolism were measured in a central laboratory.
Body composition was assessed by centrally-read whole-body DXA- and single-slice abdominal CTscan (at level L4). No scans were available from the SSAR 2004/0002 study patients.
eGFR was estimated by Cockcroft-Gault (C&G, corrected for body surface area) and MDRD-4.
Missing lipid/glucose/insulin values of the SSAR study patients at week 12 were imputed systematically. All other missing values were imputed using a last observation carried forward approach. The percentage change was modelled by repeated measurements.
In tables and graphs estimated means and SEM are depicted unless stated otherwise.


Change in lipid profile and glucose metabolism over 48 weeks
Change in body composition from baseline to 48 weeks
Change in renal function from baseline to 48 weeks
|
| |
|
 |
 |
|
|