 |
 |
 |
| |
Pharmacokinetics, Safety and Tolerability of the HCV Protease Inhibitor ABT-450 with Ritonavir Following Multiple Ascending Doses in Healthy Adult Volunteers
|
| |
| |
Reported by Jules Levin
HepDART 2009 - 06-10 December 2009 - Kohala Coast, Hawaii, USA
Barry Bernstein, Rajeev M Menon, Cheri E Klein, Adebayo A Lawal, Adel Nada, Isabelle Gaultier, Thomas J Podsadecki, Walid M Awni (Abbott, Abbott Park, IL USA)
Corresponding author: Rajeev Menon, Abbott, 100 Abbott Park Road, Dept. R4PK, AP13A, Abbott Park, IL 60064, USA
AUTHOR CONCLUSIONS
Overall ABT-450/r was well tolerated in healthy subjects.
Adverse events were generally similar to those observed in the single dose studies, most were mild and all resolved by the end of study follow-up.
Transient asymptomatic increases in indirect bilirubin levels were observed. Elevations were not associated with increases in direct bilirubin, jaundice or other liver function tests, and resolved with ongoing ABT-450 dosing.
The increase in indirect bilirubin levels strongly correlate with ABT- 450 trough concentrations, but not the Cmax or AUC. The transient rise and fall of indirect bilirubin levels with continued dosing correlated with the change in ABT-450 trough levels over time.
Mean ABT-450 Cmax and AUC values increased in a greater than dose proportional fashion following multiple dosing.
Mean ABT-450 trough concentrations showed an initial increase followed by a gradual decrease and stabilized by Day 9 to 12.
The initial increase in trough concentrations was higher when coadministered with ritonavir 100 mg BID dose than with ritonavir 100 mg QD.
This can be explained by the initial potent inhibition of CYP3A by ritonavir followed by ritonavir-mediated induction and is consistent with that observed when ritonavir is coadministered with HIV protease inhibitors.
ABT-450 exposures on Day 14 were ~2 to 3 fold higher than that on Day 1 following multiple dosing of ABT-450/r.
For the same total daily dose of ABT-450 (200 mg), QD dosing provides higher mean Cmax and AUC values and comparable mean steady state Ctrough at a lower daily dose of ritonavir (100 mg vs 200 mg), suggesting that QD dosing may be appropriate for ABT-450 when coadministered with ritonavir
ABT-450/r administered for 14 days at doses ranging from 50/100 mg BID to 300/100 mg QD demonstrated good plasma exposures, and the potential for once-daily dosing.
INTRODUCTION
ABT-450 is a potent acylsulfonamide protease inhibitor of the hepatitis C virus (HCV) being developed for the treatment of HCV genotype 1 infection.
ABT-450 has shown inhibitory concentrations in the sub-nanomolar range in genotype 1a and 1b subgenomic replicon systems in the absence of serum.
Preclinical data suggested that ritonavir coadministration can boost the pharmacokinetics of ABT-450. This was verified in the single ascending dose study where a 28-48 fold increase in ABT-450 Cmax and AUC were observed when coadministered with ritonavir (RTV).1
ABT-450 is therefore being developed with low dose ritonavir (ABT-450/r) to
optimize dose and dosing frequency.
ABT-450 with and without ritonavir has been shown to be safe and well tolerated in single ascending dose studies.1
OBJECTIVE
Assess the safety, tolerability, and pharmacokinetics of escalating, multiple oral
doses of ABT-450 administered with ritonavir (ABT-450/r) under non-fasting
conditions for 14 days in healthy adult subjects.
METHODS
Study Design
This was a Phase 1, multiple-dose, randomized, placebo-controlled,
parallel group study conducted according to a sequential design.
The protocol was reviewed and approved by an Independent
Review Board prior to start of the study. All subjects who participated
in the study gave written informed consent prior to starting any study procedures.
Adult male and female subjects in general good health were selected
to participate in the study according to the protocol-defined selection criteria.
In Group 1, eight subjects were randomized in a 3:1 fashion and in Groups 2 to 4, ten subjects were randomized in a 4:1 fashion to receive multiple doses of
ABT-450/r or matching placebo from Days 1 through 14.
Safety, tolerability, and preliminary pharmacokinetic results were assessed in each group prior to the start of the next group.
The ABT-450/r doses in the study were 50/100 mg BID, 100/100 mg BID, 200/100 mg QD, and 300/100 mg QD.
Pharmacokinetic Assessments
Samples for complete pharmacokinetic profiles for ABT-450 and ritonavir were
drawn on Day 1 and Day 14. Samples for trough concentration measurements were drawn prior to morning dose on days 2, 3, 5, 7, 9, 12 and 14
ABT-450 and ritonavir concentrations were determined using a liquid
chromatography tandem mass spectroscopic method (LC-MS/MS) with a lower limit of quantitation of 0.5 ng/mL.
Pharmacokinetic and statistical analysis was conducted using SAS version 9.1 (Cary, NC).
Exposure response analyses for bilirubin blood levels were conducted using NONMEM version 6.1.
Safety and Tolerability
Assessments
Safety and tolerability assessments included physical examination, vital sign
measurements, and clinical laboratory tests.
12-lead Electrocardiogram (ECG) were recorded in triplicate at multiple timepoints before and after dosing.
Adverse events were monitored throughout the study.
Adverse events were coded using the Medical Dictionary for Regulatory Activities (MedDRA 11.1).
RESULTS
Baseline Characteristics
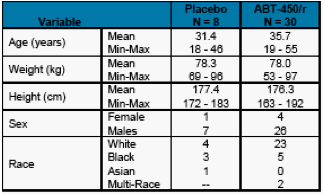
A total of 38 healthy adult subjects were enrolled in the study and randomized to receive ABT-450/r or placebo.
A summary of the subjects' demographic characteristics at baseline is presented in Table 1.
Table 1. Demographic Summary for all Subjects Dosed with ABT-450 and Placebo
|
| |
|
 |
 |
|
|