 |
 |
 |
| |
Efficacy, Safety and Tolerability of Lopinavir/ritonavir (LPV/r) in HIV-Infected Women:
Results of a Meta-Analysis of 7 Prospective,
Randomized Clinical Trials (RCTs) Through 48 Weeks
|
| |
| |
Reported by Jules Levin
1st International Workshop on HIV & Women
from Adolescence through Menopause
Washington DC
January 10-11, 2011
Ashwaq Hermes, Linda Fredrick, Mary Pasley, Roger Trinh, Marisol Martinez, Michael Norton
Abbott, Abbott Park, IL
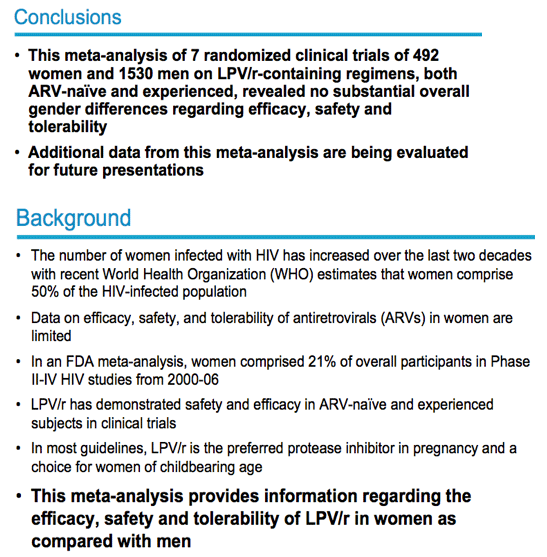
Additional background:
-- Worldwide, women constitute half of all people living with HIV/AIDS.
-- For women in their reproductive years (15–44), HIV/AIDS is the leading cause of death and disease worldwide.
-- In every region of the world, more adult women (15 years or older) than ever before are now living with HIV.

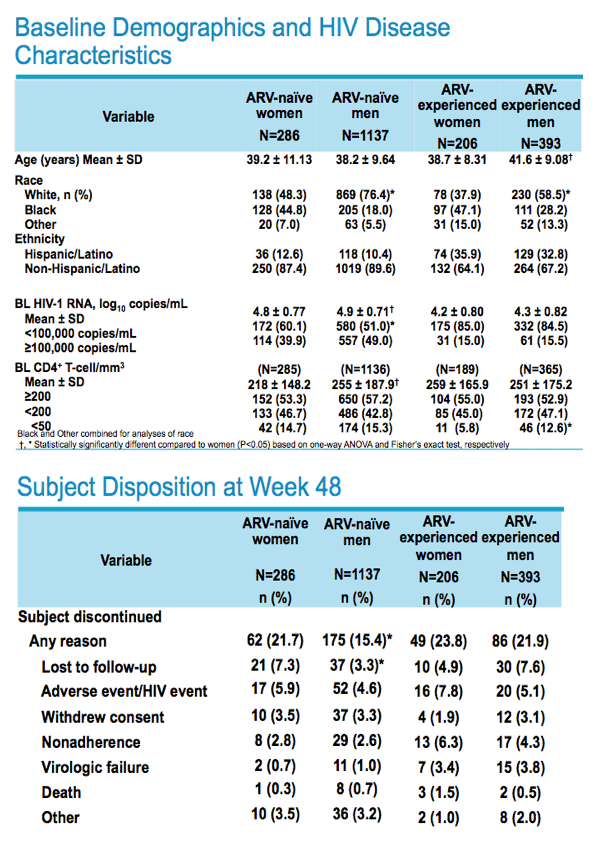
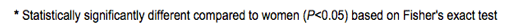
Other reasons for discontinuation, ARV-experienced:
1 PATIENT CANNOT TOLERATE KALETRA (+++ DIARRHOEA)
2 ENROLLED IN ERROR PATIENT RESISTANT TO KALETRA
3 OPPORTUNISTIC INFECTION.
4 WORK OUT SPAIN
5 CONGESTIVE HEART FAILURE, PULMONARY EDEMA, ACUTE RENAL FAILURE
6 ACCORDING TO PRIMARY MD, SUBJECT NEVER TOOK STUDY MEDICATION
7 SUBJECT DID NOT COMPLY WITH STUDY VISITS OR IP RETURNED BY MAIL #102 TABS KALETRA ON 7-30-08
8 PCP DISCONTINUED STUDY DRUG AND STARTED SUBJECT ON NNRTI
9 DRUG INTOLERANCE
10 DOESN'T MEET INCLUSION CRITERIA
Other reasons, ARV-naïve:
1 PATIENT WITHDREW CONSENT TO FOLLOW IN THE STUDY DUE TO AE.
2 LOW DOSAGE OF KALETRA - PATIENT NEEDS TO TAKE BID POSOLOGY.
3 LOW LOPINAVIR LEVELS @ 24 HRS. NEEDED TO TAKE LOPINAVIR BD
4 LOW LOPINAVIR LEVELS @ 24 HRS NEEDS TO TAKE LOPINAVIR BD
5 SUBJECT RELOCATED OUT-OF-STATE
6 RECREATIONAL DRUGS
7 HE'LL LIVE IN PERU
8 SUBJECT WANTED FEWER PILLS
9 WANTED TO TAKE LESS TABLETS
10 THE PATIENT IS GOING TO WORK AT ENGLAND NEXT WEEK
11 SUBJECT HAS TO STOP THERAPY. HE IS UNABLE TO PERFORME LAB SAMPLES
12 RELOCATED TO AFRICA
13 SUBJECT MOVING ABROAD TO WORK
14 SUBJECT ENROLL IN OTHER STUDY
15 MOVING TO SCOTLAND
16 PLASMA STUDY DRUG CONCENTRATION = 0 AT 24 H
17 SUBJECT INCARCERATED
1 DRUG BINGE
2 MOVING TO ENGLAND
3 PATIENT DISCONTINUED PER HIS REQUEST TO WITHDRAW
4 PROBLEMS WITH STUDY MEDICATIONS & GENERAL INTOLERANCE TO STUDY MEDICATIONS
5 PATIENT NONCOMPLIANT DUE TO UNDERLYING PSYCHIATRIC ILLNESS
6 PATIENT STARTED ANTIRETROVIRAL MEDS DURING HOSPIT
7 WITHDREW CONSENT
8 HE CAN NOT TAKE SO MANY PILLS
9 SUBJECT WITHDREW CONSENT
10 PILL BURDEN
11 PATIENT INCARCERATED (IN JAIL)
12 DIFFICULTY TO SWALLOW PILLS
13 UNABLE TO CONTACT AFTER THE DEATH OF FAMILY MEMBERS/HAS NOT RETURNED FOR VISIT
14 DID NOT WANT TO TAKE ANY MORE STUDY MEDICATION
15 RIFAMPIN
16 PERSONAL DECISION
17 DUE TO PERSONAL PROBLEMS
18 SHE COULDN'T ACCEPT THE CONSISTENCY OF THE TABLETS
19 THE PATIENT IS UNABLE TO SWALLOW THE MEDICATION
20 CHANGED MIND
21 SITE CLOSURE
22 SITE CLOSURE
23 SUBJECT MOVING OUT OF STATE AND NO OTHER SITES ARE LOCAL IN NEW MEXICO
24 PT WITHDREW CONSENT
25 EXPERIENCING LOW PSYCHOLOGICAL MORALE AND HAS THUS DECIDED TO STOP ANTIRETROVIRAL TREATMENT
26 PATIENT CONFUSED DUE TO INTRAVENTRICULAR BLEED, THEREFORE THERE ARE CONSENT ISSUES
27 HOSPITALIZATION
28 PATIENT LEAVING COUNTRY
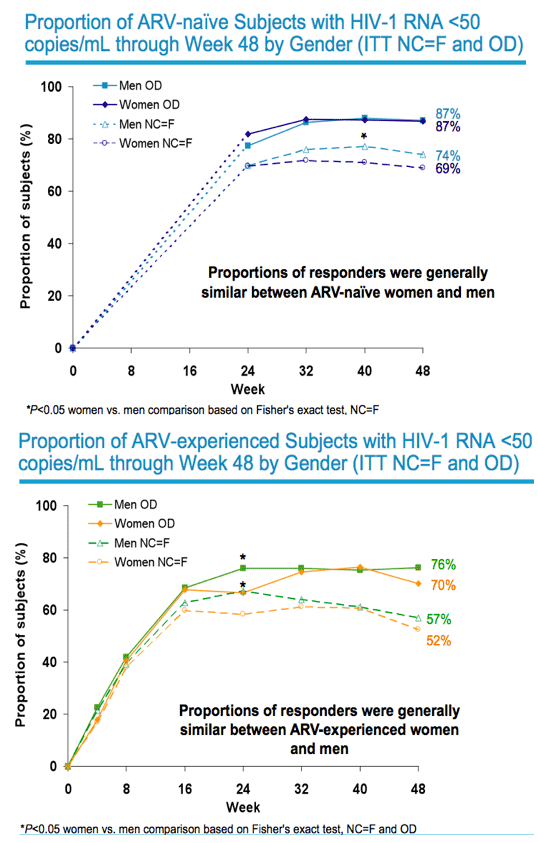
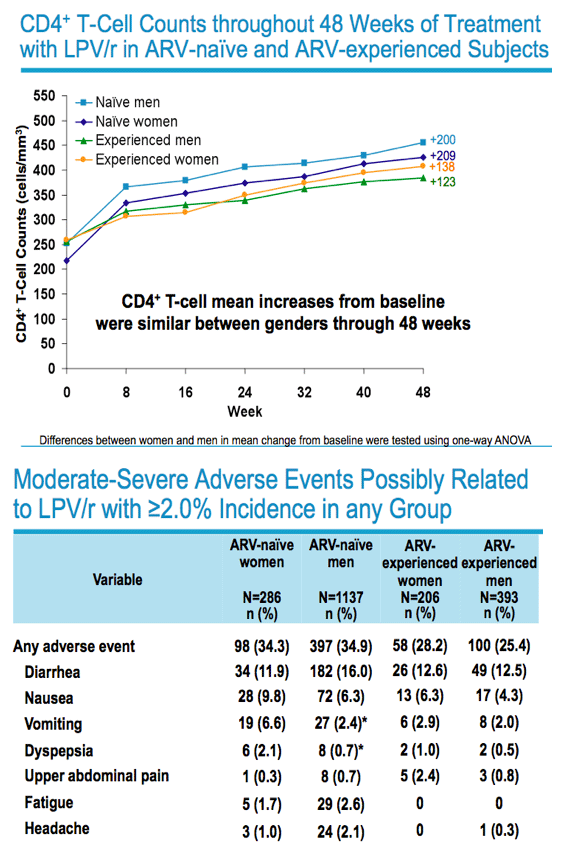
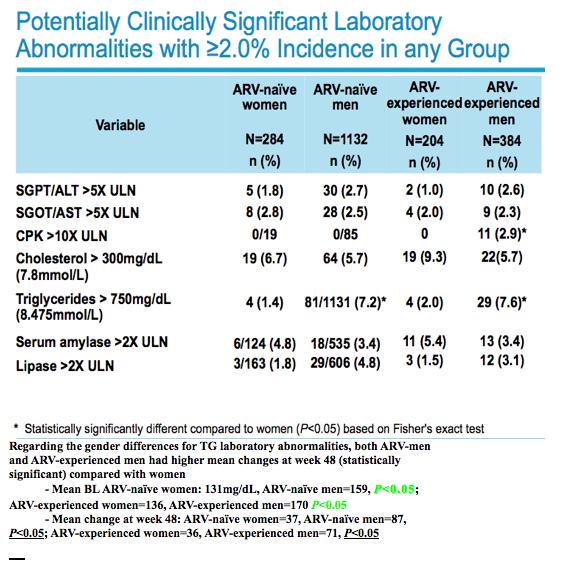
|
| |
|
 |
 |
|
|