 |
 |
 |
| |
The Safety, Efficacy, and Steady State Pharmacokinetics of Atazanavir/ritonavir (ATV/r) Once Daily Given in Combination With Twice Daily AZT/3TC During Pregnancy: Results of Study AI424182
|
| |
| |
Reported by Jules Levin
Reported at the Intl 1st Women's & HIV Workshop
H. Hardy; T. Eley; M. Child; S.P. Huang; W. Hu; Y. Wang; M. Gomez; M. Mathew; A. Collins; S. Hilaly; E. Vandeloise; H. Sevinsky; X. Xu; T. Guo; V. Wirtz; A. Farajallah; R. Bertz; D. McGrath
Bristol-Myers Squibb Company, Princeton, NJ, USA

Introduction
There are limited options for the use of protease inhibitors as a component of HAART in HIV-infected pregnant women, and an unmet medical need remains for a once-daily, safe, efficacious, and well tolerated protease inhibitor (PI) for use during pregnancy
Atazanavir (ATV) is a potent, well tolerated, once-daily HIV-1 PI with established efficacy and safety in both treatment-naïve and treatment-experienced, non-pregnant HIV-infected adults
- Safety data are accumulating on the use of ATV in pregnant HIV-infected adults1
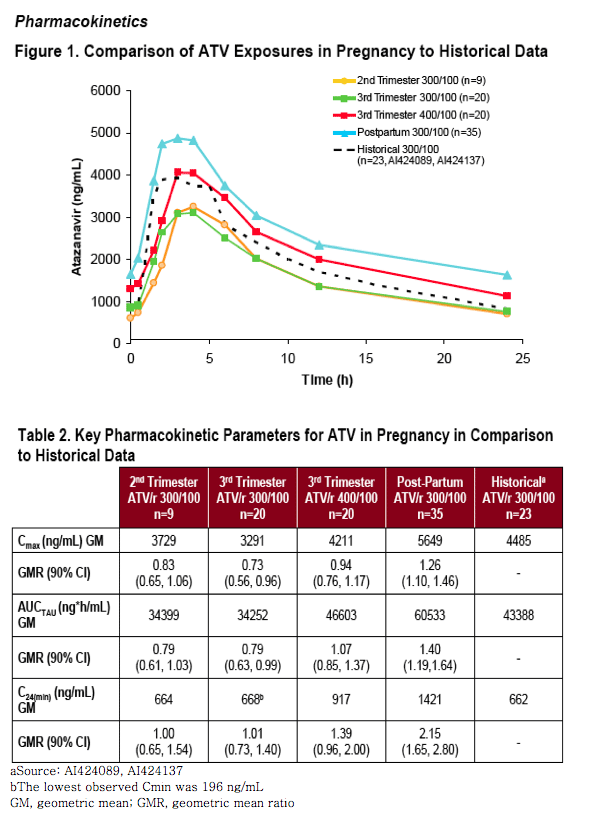
There are some data to suggest that, despite changes in ATV levels during pregnancy (decreased AUCTAU but similar Cmin), they may be adequate to recommend dosing at the usual non-pregnant once-daily dose of ATV 300 mg boosted with low-dose ritonavir 100 mg (ATV/r 300/100 mg)2
However, further data are needed before such recommendation can be made regarding adequate ATV/r dosing
Additionally, as ATV inhibits UGT1A1, there is theoretical concern that using ATV in pregnant women may exacerbate physiologic hyperbilirubinemia in neonates
Objectives
Primary: To determine the appropriate dose of ATV/r that produces adequate drug exposure during pregnancy compared to historical data in HIV-infected patients
Secondary:
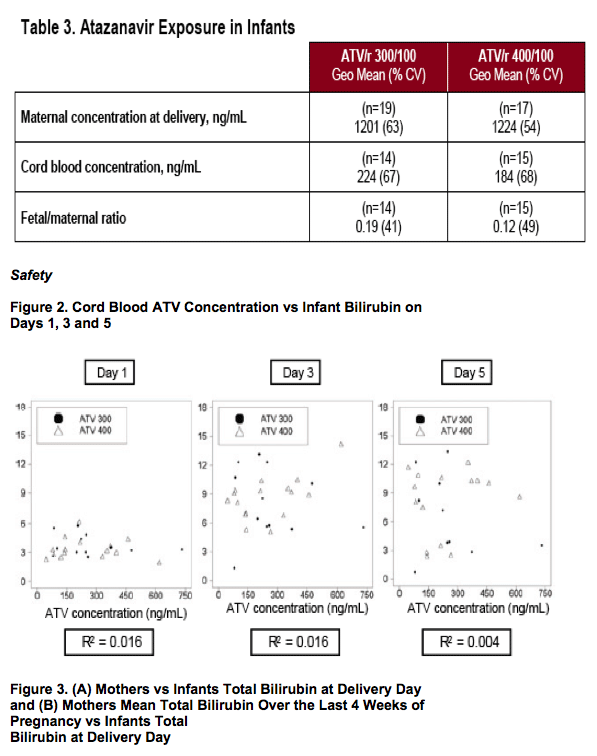
- To measure maternal:infant ATV level ratio
- To assess the safety of ATV/r in pregnant women and in infants born to women
exposed to ATV/r during pregnancy
- To measure antiviral efficacy in mothers and to assess prevention of mother-to-child transmission of HIV-1
- To assess effect of maternal and infant UGT1A1 genotype on maternal and infant bilirubin levels, respectively
-- To assess adherence to treatment regimen and to ATV
Methods
Study Design
Multicenter, open-label, prospective, single-arm, phase I study
- Enrollment in South Africa, Puerto Rico and the United States
Study Population: HIV-1 infected pregnant women between 12-32 weeks of gestation; CD4
≥200 cells/mm3
Treatment: ATV/r 300 or 400/100 mg once-daily and AZT/3TC 300/150 mg twice-daily
Pharmacokinetic Monitoring:
-- Planned First Interim PK Analysis3 was performed after 12 subjects receiving ATV/r 300/100 mg reached steady state during weeks 28-36 of gestation to assess need for increase to ATV/r 400/100 mg in the 3rd trimester
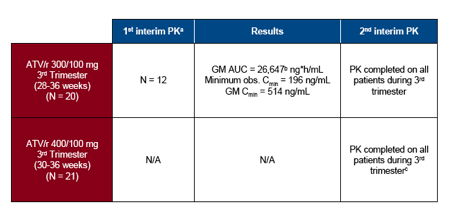
aPrespecified criteria for dose increase - If 2 of 12 subjects with ATV Cmin <150 ng/mL, but 10/12 subjects with ATV Cmin ≥50 ng/mL OR if geometric mean ATV AUC (TAU) for 12 subjects <30,000 but ≥15,000 ng*h/mL
bDecision to increase the ATV dose in the 3rd trimester was based on the AUC results
cAfter decision to increase 3rd trimester dose, subjects enrolled in the study who were in the 2nd trimester underwent blood sampling for PK analysis of ATV/r 300/100 mg in the 2nd trimester
GM = geometric mean

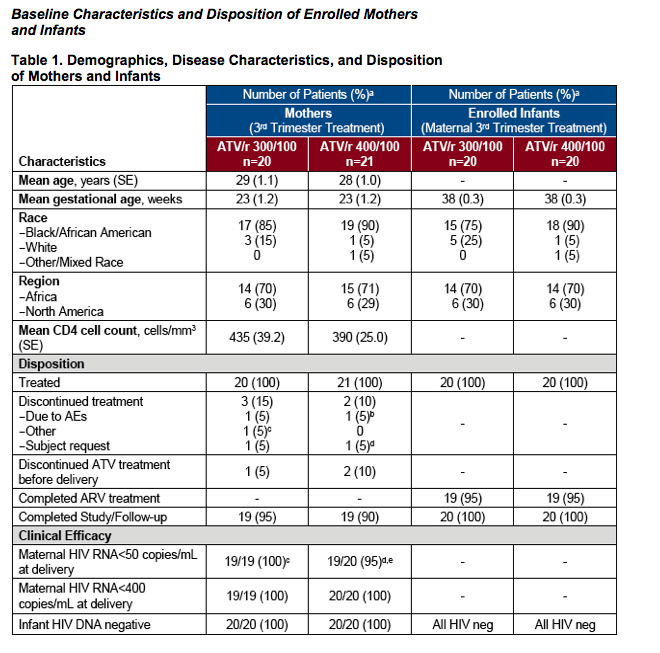
aPercentages are based on patients with measurements
bPatient diagnosed with pre-eclampsia with a grade 3-4 transaminitis; all ARVs held and infant delivered by C-section 4 days later; HIV RNA <50 copies/mL prior to ARV discontinuation and HIV RNA <50 copies/mL on day of delivery
cOne subject was discontinued from study drug when premature labor developed; delivered 12 days later; HIV RNA <50 copies/mL at delivery
dOne subject withdrew from study (withdrew consent) after 3 weeks of therapy, so no HIV RNA was available from time of delivery
eOne subject had 3 consecutive HIV RNA <50 copies/mL prior to delivery; an HIV RNA <59 copies/mL at delivery, with subsequent re-suppression to <50 copies/mL post-partum SE, standard error; AEs, adverse events
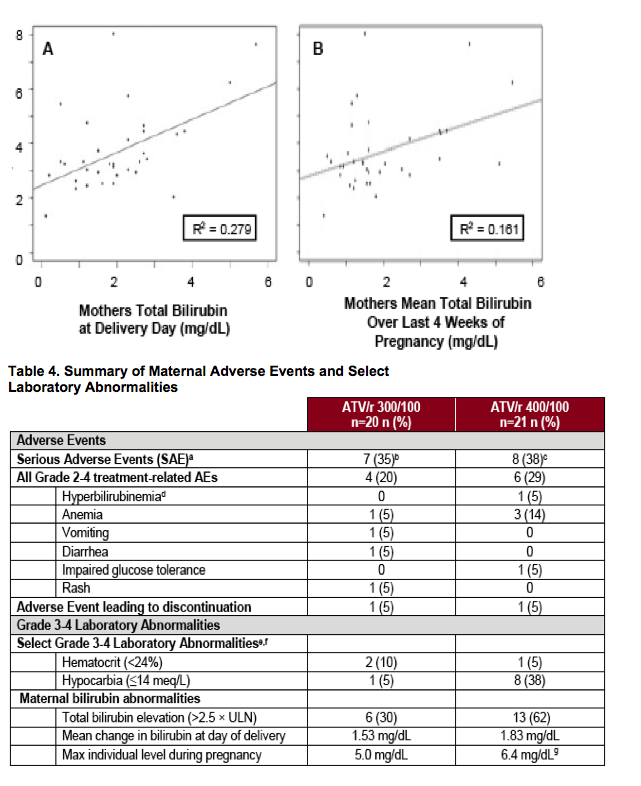
aThe proportion of SAEs are based on enrolled subjects
Drug-related SAEs: bAnemia (n=1); cAnemia (n=2) and hyperbilirubinemia (n=1)
dBased on AE reports not laboratory values
eALT elevation and AST elevation (>5 x ULN) occurred in 0 and 1 (5%) of patients receiving ATV/r 300/100 mg and ATV/r 400/100 mg, respectively
fTotal cholesterol (≥240 mg/dL) occurred in 1/14 (7%) and 3/16 (19%) of patients receiving ATV/r 300/100 mg and ATV/r 400/100 mg, respectively. Fasting lipids were measured at screening and at post-partum Week 4 only
gReported during episode of pre-eclampsia
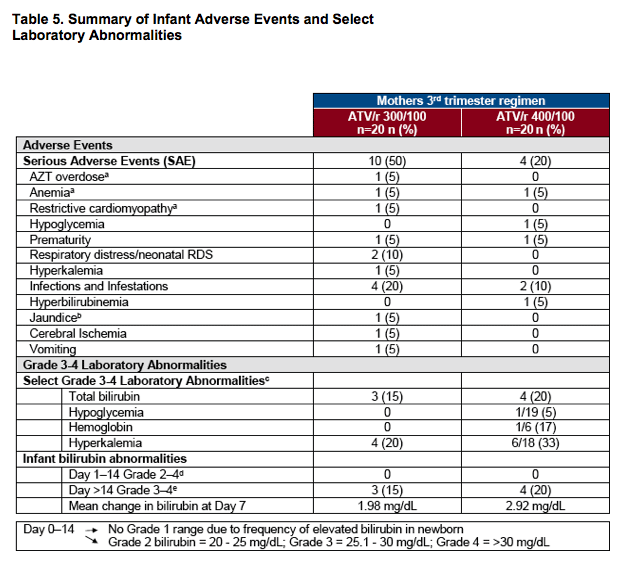
aDrug-related SAEs: three infants (with mothers on ATV/r 300/100 mg) had SAEs considered related to AZT, overdose, anemia and restrictive cardiomyopathy
bInfant born at 35 weeks following premature rupture of membranes; maternal bilirubin 2.3 g/dL on day of delivery; birth weight 1.8 kg; infant bilirubin 13 mg/dL on Day 3; received 3 days of phototherapy; discharged well
cValues for Grade 3-4 abnormalities not provided as thresholds vary with age of newborn
dSix infants received phototherapy
eMaximum individual level: 8.5 mg/dL at Day 15
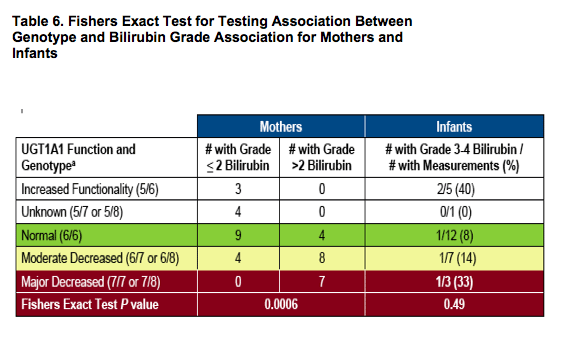
aUGT1A1 polymorphisms are due to varying number of TA repeats and functional groups are based on genotype

F, failure; M, missing; NC, non-completers
aExcludes adherence data from Visit 2 in the 2nd trimester
bNumber Adherent/Number Evaluable (%); Reasons given for non-adherence to regimen included did not want to take pills,
slept through dose time, felt depressed or overwhelmed, had a change in daily routine, had problems taking pills, had too
many pills to take, felt like drug was toxic or harmful

|
| |
|
 |
 |
|
|