 |
 |
 |
| |
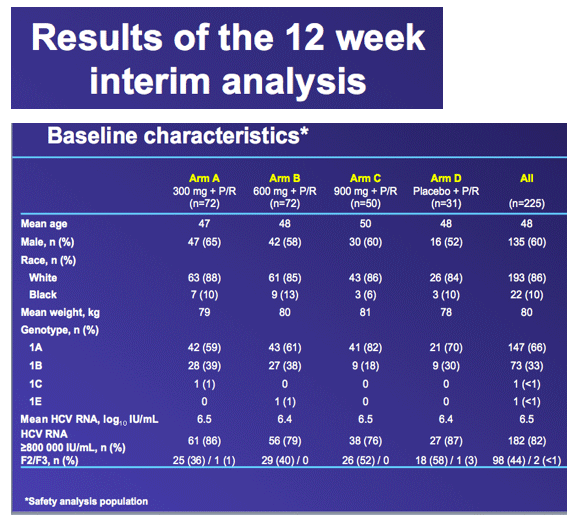
Phase II randomized, partially blind, parallel-group study of oral danoprevir (RG7227) with PegIFN alfa-2a (PEGASYS) plus ribavirin (COPEGUS) in treatment-naive genotype 1 patients with CHC: Results of planned Week 12 interim analysis of the ATLAS study
|
| |
| |
Reported by Jules Levin
AASLD Nov 1 2010 Boston
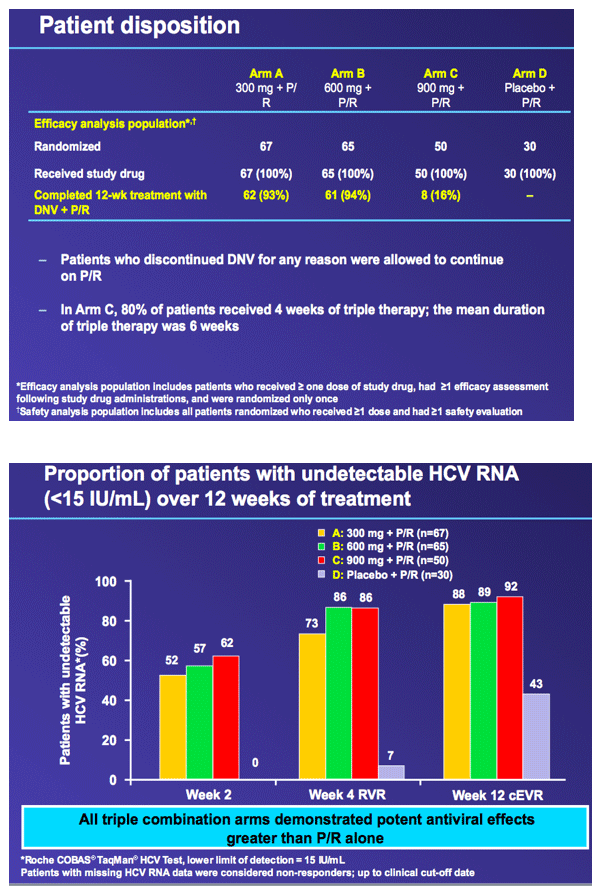
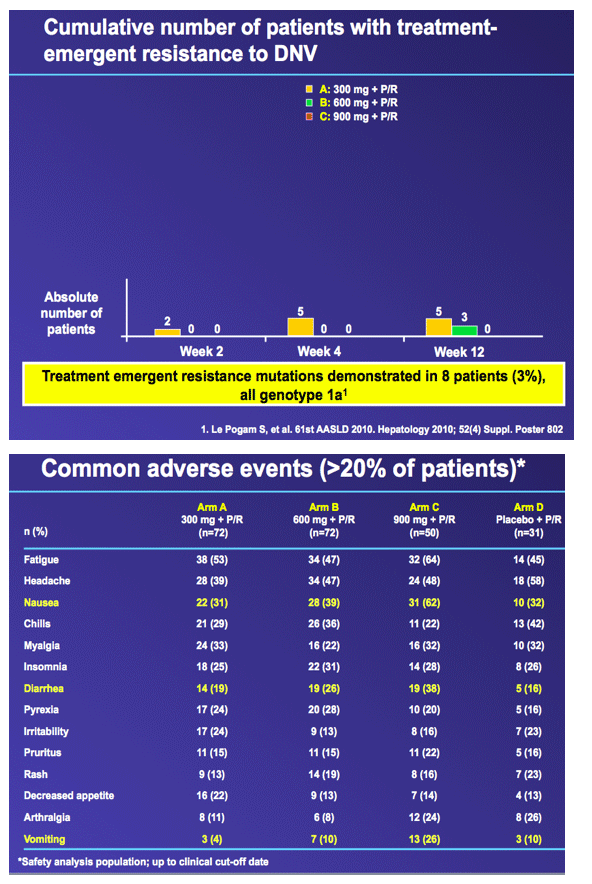
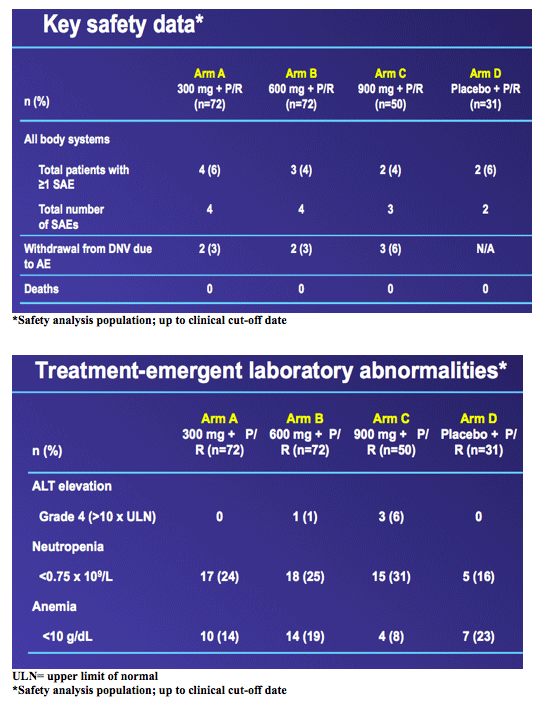
from Jules: this was the Roche HCV protease danoprevir study of 300, 600 and 900 mg where grade 4 ALT elevations emerged in 4 patients, 1 in the 600mg arm (n=60), and 3 in the 900mg arm (n=60). In this slide presentation they said the elevations were asymptomatic, occurred between 6 and 12 weeks of dosing, reversible upon discontinuation, one case was associated with elevated bilirubin, and modeling of available data confirms relationship between DNV exposure (AUC) and likelihood of ALT elevations. Two points, danoprevir displayed potency in this study with 92% in high dose (900mg+PR) achieving cEVR after 12 weeks of treatment, and they announced the new study where danoprevir will be boosted with low dose ritonavir in a once-daily administration of the drug, see the study design below & in attached report of slides presented, and boosting with low-dose ritonavir is intended to address the ALT elevations and perhaps make danoprevir more potent. They said "modeling of available data confirms relationship between DNV exposure (AUC) and likelihood of ALT elevations1.....expected benefits from co-dosing with low-dose ritonavir: maintains antiviral activity with substantially lower DNV AUC and Cmax1".
Norah Terrault, Curtis Cooper, Luis A. Balart,
Dominique G. Larrey, Terry Box, Eric M. Yoshida,
Eric Lawitz, Peter Buggisch, Peter Ferenci,
Martin Weltman, Emily Labriola-Tompkins, Ying Zhang, Mercidita T. Navarro, Chin Yin Lim, Ellen S. Yetzer, Patrick Marcellin




|
| |
|
 |
 |
|
|