 |
 |
 |
| |
No evidence of drug resistance or baseline S282T resistance mutation among GT 1 and GT 4 HCV infected patients on nucleoside polymerase inhibitor RG7128 and Peg-IFN/RBV combination treatment for up to 12 weeks: Interim analysis from the PROPEL study
|
| |
| |
Reported by Jules Levin
AASLD Nov 2 2010 Boston
S. Le Pogam,1 J.M. Yan,1 A. Kosaka,1 Y. Ji,1 D.J. Chin,2 N. Gonzaludo,1 A. Ewing,1 K. Klumpp,1 I. Nájera1
1Roche, Palo Alto, CA, United States; 2Roche, Nutley, NJ, United States
AASLD: High rates of early viral response, promising safety profile and lack of resistance-related breakthrough in HCV GT 1/4 patients treated with RG7128 plus PegIFN alfa-2a (40KD)/RBV: Planned Week 12 interim analysis from the PROPEL study - (11/03/10)

Conclusions
In the population analyzed, no viral load rebound or non-response was observed in any genotype 1 or 4 patient treated with RG7128-based triple therapy in this phase 2b trial.
Eleven patients (six genotype 1b and five genotype 1a) showed partial responses, with viral loads >1000 IU/mL at the end of triple therapy.
No evidence of development of resistance to RG7128 was observed in any of these 11 patients per population and clonal sequencing and phenotypic studies.
Limited replication capacity of the S282T mutant in vitro and the absence of this mutant at baseline may contribute to the lack of clinical resistance.
The potent antiviral activity, high barrier to resistance, and promising safety and tolerability profile are encouraging, and support further development of RG7128 as a potential backbone of future combination therapies, including interferon-free regimens.
No S282T observed in baseline samples
· None of the 406 (out of 408) baseline samples for which the NS5B was successfully amplified and sequenced contained the in vitro identified RG7128 resistance mutation.
INTRODUCTION
RG7128 is a novel nucleoside analog inhibitor of hepatitis C virus (HCV) NS5B polymerase that is active against HCV genotypes 1, 2, 3, and 4.[1-3]
Patients with chronic hepatitis C have experienced rapid and profound reductions in HCV RNA levels after administration of RG7128 alone, in combination with peginterferon alfa-2a plus ribavirin, and in combination with danoprevir.[1-5]
Previous analyses have demonstrated that RG7128 has a high barrier to the development of drug resistance.[6]
One particular amino acid substitution (S282T) has been identified in vitro to confer resistance to RG7128. However, this variant has not been observed in patients treated with RG7128 for up to 2 weeks' monotherapy and 4 weeks in combination with peginterferon and ribavirin.[6]
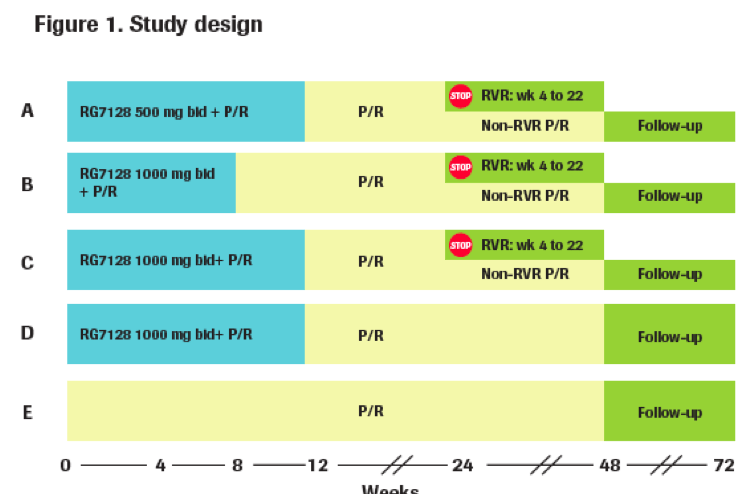
PROPEL is a randomized double-blind, active-controlled, parallel-group study in which treatment-naive HCV genotype 1 or 4 infected patients are treated with RG7128 in combination with peginterferon alfa-2a (40KD) and ribavirin.
Patients were randomized to one of four treatment arms of 8 or 12 weeks of triple therapy followed by peginterferon alfa-2a (40KD) plus
ribavirin to complete a total of 24 or 48 weeks of therapy (Arms A-D), or to 48 weeks of therapy with peginterferon alfa-2a (40KD) plus
ribavirin (Arm E). Treatment of some patients in Arms A-C was RVR-guided (Figure 1). All patients were followed for 24 weeks after the
end of therapy for SVR.
Objective
The aim of this study was to assess the resistance profile of patients enrolled in the PROPEL study who received RG7128, peginterferon alfa-2a (40KD) and ribavirin for 8 or 12 weeks.
Methods
Assessment of HCV kinetics
· Samples for determination of serum HCV RNA concentrations (by Roche COBAS® TaqMan® HCV Test, detection limit 15 IU/mL) and viral resistance monitoring were collected at baseline and at week 1, 2, 4, 6, 8, 10, and 12 of triple therapy for all patients.
Sample selection
· Baseline samples from all patients were analyzed by population sequencing.
· On-treatment samples were selected for resistance monitoring from patients who met the following criteria:
a. viral load rebound, defined as an increase in HCV RNA level of ≥1 log10 IU/mL above nadir that is sustained for at least two consecutive viral load measurements (nadir must be a ≥0.5 log10 IU/mL decrease from baseline);
b. partial response, defined as an initial decrease in HCV RNA of ≥0.5 log10 IU/mL from baseline, followed by stabilization for at least two consecutive viral load measurements, and/or a viral load ≥1000 IU/mL at the end of at least 4 weeks of treatment with RG7128;
c. non-response, defined as a decrease in HCV RNA level of <0.5 log10 IU/mL by the end of treatment in patients who have received at least two weeks of treatment.
· Samples selected for characterization of NS5B coding sequences from patients meeting criteria a-c above include the following (provided the viral load was above 1000 IU/mL):
· Baseline samples in patients with rebound or partial response
· The first sample after nadir in patients with viral load rebound
· Additional samples collected after rebound but before the end of RG7128 dosing
· End-of-RG7128-treatment samples from partial responders
Resistance monitoring procedures
Sequencing
Baseline analysis
· The entire NS5B coding region was amplified and population sequencing was done on baseline samples from all patients.
· Clonal sequencing spanning the NS5B polymerase region was done for baseline samples from patients who experienced rebound, partial response or non response.
On-treatment analysis
· Population and clonal sequencing of the entire NS5B polymerase coding region was performed on samples from patients meeting the criteria described above.
Phenotypic characterization
· Phenotypic characterization was done by:
- Cloning the NS5B genes from selected samples (described above) into transient shuttle replicons
- Transfection of in vitro-transcribed shuttle replicon RNA into Huh7 cells and
-- Calculating their susceptibility to RG7128 and interferon alfa-2a (Roferon) (expressed as the EC50) compared with that of reference strains (H77 for genotype 1a, and Con1 for genotype 1b). EC50 values obtained for treated samples will be compared with those of their respective baseline samples.
Results
HCV kinetics
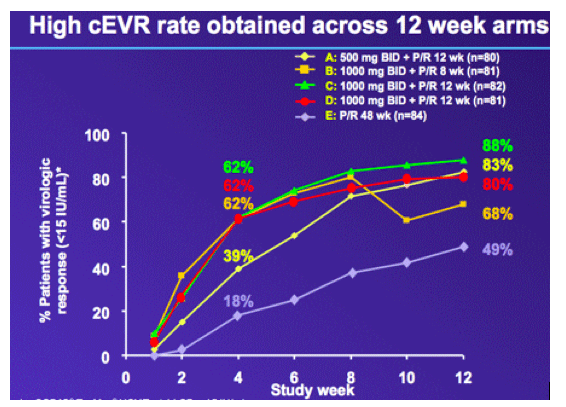
No patients experienced viral load rebound or non-response
· No HCV RNA rebound or non-response has been observed during triple therapy in any patient who was adherent to treatment.
Eleven patients experienced a partial response
· A total of 11 genotype 1 patients (adherent to treatment) had HCV RNA level ≥1000 IU/mL at the end of triple therapy.
- Five patients had received RG7128 500 mg bid for 12 weeks (Group A), and one patient had received 1000 mg bid for 12 weeks (Group D) (Figure 2).
-- Five patients had received RG7128 1000 mg bid for 8 weeks (Group B) (Figure 3).
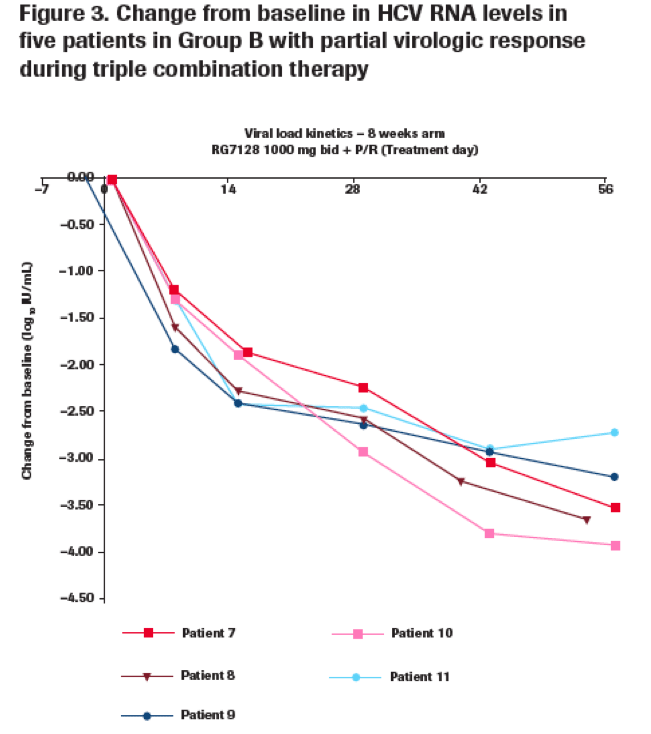
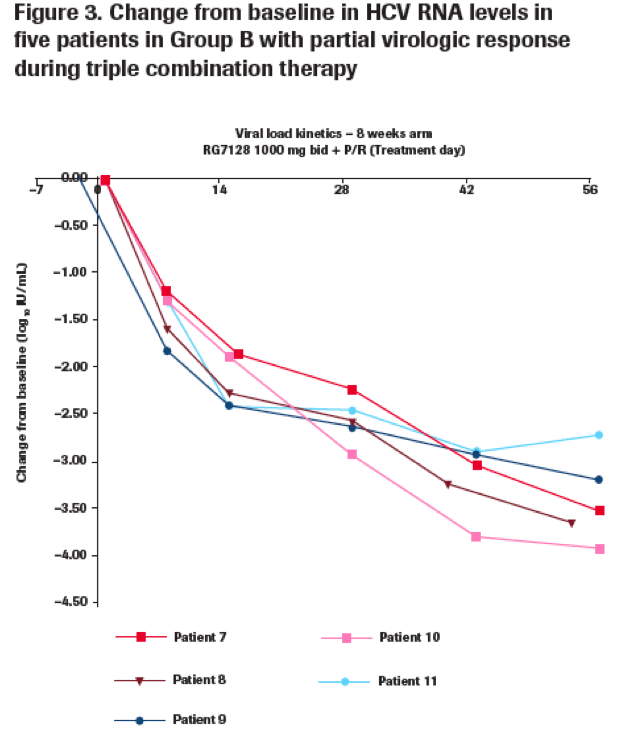
Sequencing results
No S282T observed in baseline samples
· None of the 406 (out of 408) baseline samples for which the NS5B was successfully amplified and sequenced contained the in vitro identified RG7128 resistance mutation.
No S282T observed in on-treatment samples from 11 patients who experienced partial response as assessed through population and clonal analysis
· None of the 11 patients had the RG7128 resistance mutation S282T or any other common mutation in the NS5B coding region before,
during, or at the end of triple therapy.
· Random amino acid changes observed in on-treatment samples were not consistent between patients in this trial (Table 1) or with amino acid changes observed in other trials of RG7128 (not shown).
· Clonal analysis of ~70 NS5B variants per sample from patients selected for viral resistance monitoring showed:
- No S282T in the 761 NS5B variants from the 11 baseline samples
- No S282T in the 807 NS5B variants from the 11 RG7128-treated analyzed samples
· The observed random amino acid substitutions (or mixtures) are unlikely to confer resistance to RG7128 because all on-treatment samples remained fully susceptible to the drug (see below). Phenotypic characterization of NS5B sequences from the 11 patients who experienced partial response
· No change in susceptibility to RG7128 (Figure 4) or interferon alfa-2a (not shown) was observed between on-treatment and baseline
samples obtained from any patient and all EC50 values were similar to reference controls.

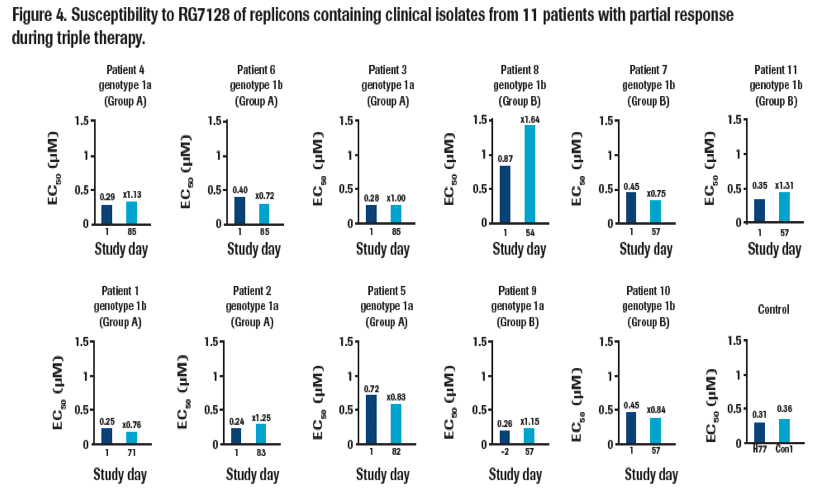
Values are means calculated from 2-5 independent experiments. Actual values are shown for day 1 samples and the fold-change in EC50 relative to day 1 is shown for on-treatment time points. Susceptibility of controls for genotype 1a (H77) and genotype 1b (Con1) are shown for comparison.
References
1. Reddy R, Rodriguez-Torres M, Gane E, et al. Antiviral activity, pharmacokinetics, safety, and tolerability of R7128, a novel nucleoside HCV RNA polymerase inhibitor, following multiple, ascending, oral doses in patients with HCV genotype 1 infection who have failed prior interferon therapy [abstract LB9]. Hepatology 2007; 46 (Suppl. 1): 862A
2. Gane E, Rodriguez-Torres M, Nelson D, et al. Antiviral activity of the HCV nucleoside polymerase inhibitor R7128 in HCV genotype 2 and 3 prior non-responders: interim results of R7128 1500mg BID with PEG-IFN and ribavirin for 28 days [abstract LB10]. Hepatology 2008; 48 (Suppl. 1): 1024A
3. Rodriguez-Torres M, Lalezari J, Gane E, et al. Potent antiviral response to the HCV nucleoside polymerase inhibitor R7128 for 28 days with PEG-IFN and ribavirin: subanalysis by race/ethnicity, weight and HCV genotype [abstract 1899]. Hepatology 2008; 48 (Suppl. 1): 1160A
4. Gane E, Roberts S, Stedman C, et al. Oral combination therapy with a nucleoside polymerase inhibitor (RG7128) and danoprevir for chronic hepatitis C genotype 1 infection (INFORM-1): a randomised, double-blind, placebo-controlled, dose-escalation trial. Lancet 2010; Published online, October 15, 2010
5. Jensen D, Wedemeyer H, Herring R, et al. High rates of early viral response, promising safety profile and lack of resistance-related breakthrough in HCV GT 1/4 patients treated with RG7128 plus PegIFN alfa-2a (40KD)/RBV: planned week 12 interim analysis from the PROPEL study. Oral presentation. The Liver Meeting 2010 [61st Annual Meeting of the American Association for the Study of Liver Diseases (AASLD)], October 29-November 2, 2010, Boston, MA, USA
6. Le Pogam S, Seshaadri A, Ewing A, et al. RG7128 alone or in combination with pegylated interferon-α2a and ribavirin prevents HCV replication and selection of resistant variants in HCV-infected patients. J Infect Dis 2010; 202:1510-19
|
| |
|
 |
 |
|
|