 |
 |
 |
| |
The Prevalence of Darunavir Associated Mutations in PI-naive and PI-experienced HIV-1 Infected Children in the UK
|
| |
| |
Reported by Jules Levin
CROI 2010 Feb 16-19 SF
Katherine Boyd1, A. Sarah Walker1, David Dunn1, Ali Judd1, Deenan Pillay2, Esse Menson3, Gareth Tudor-Williams4, and Diana M. Gibb1 on behalf of the Collaborative HIV Paediatric Study (CHIPS) and the UK Collaborative Group on HIV Drug Resistance1 MRC Clinical Trials Unit, London, UK; 2 University College, London, UK; 3 St Thomas' NHS Trust, London, UK; 4 Imperial College, London, UK
AUTHOR SUMMARY & CONCLUSIONS
· DRV RAMs are very rare in PI-naïve children.
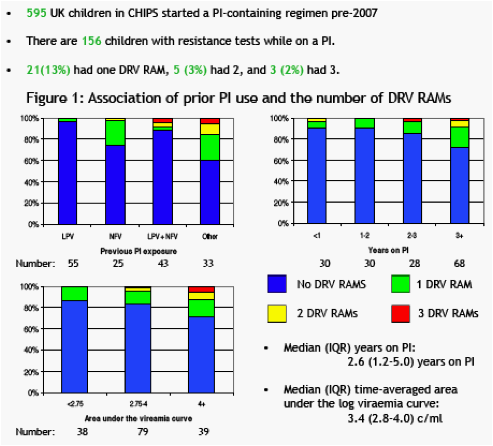
· The prevalence of DRV RAMs is low in those whose only PI exposure is LPV/r. It is higher in those with exposure to other PIs other than LPV (likely due to failure).
· At the last test on PI only 3 children had 3 DRV RAMs.
· There is an increase in resistance with >2 years PI exposure and increase viraemia.
· Overall, results suggest that DRV/r is of use as both a first PI and an alternative second PI as resistance is low.
· We could consider specific analyses looking at the relationship between LPV/rexposure and DRV RAMs using random effects modelling although RAM rates are low.
· ATV/r is only licensed OD for children age 6+ years (weighing ≥25kg if ART-exp). It may also be of interest as a alternative first, or as a second, PI.
ABSTRACT
Background- The protease inhibitor (PI) darunavir, boosted by ritonavir (DRV/r), is virologically effective and well tolerated in adults. Although co-formulated lopinavir (LPV/r) is currently the first-line PI for HIV-1 infected children in the UK, DRV/r has potential utility as once daily first-or second-line PI after previous PI failure. Identifying the prevalence of DRV/r resistance associated mutations (RAMs) in children is important for determining it's clinical utility.
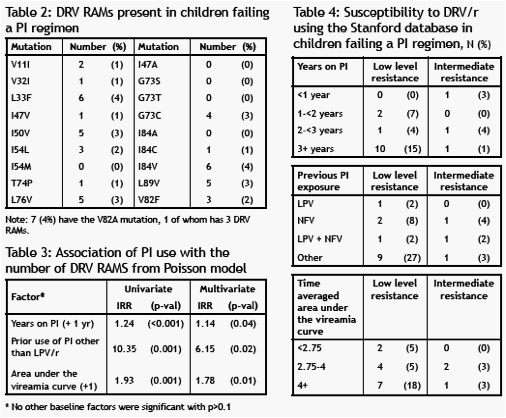
Methods- Data during 1998-2007 combine that from the Collaborative HIV Paediatric Study (CHIPS, a cohort of ~95% reported HIV-1 infected children in UK/Ireland since 1996), and the UK HIV Drug Resistance Database. DRV RAMs were identified from the 2008 IAS mutations list (V11I, V32I, L33F, I47V, I50V, I54L/M, T74P, L76V, I84V, L89V) with additional mutations (I47A, G73S/T/C, I84A/C, V82F) from the Stanford database. The prevalence of RAMs was estimated in (i) PI-naive children, using the first PI-naive test, and (ii) PI-experienced children, using cumulative resistance at the last test on a PI. Associations between the type and the duration of PI exposure and the area under the viraemia curve since the start of PI with the number of RAMs was analysed using multivariate Poisson regression. Susceptibility to DRV/r was defined using the Stanford algorithm.
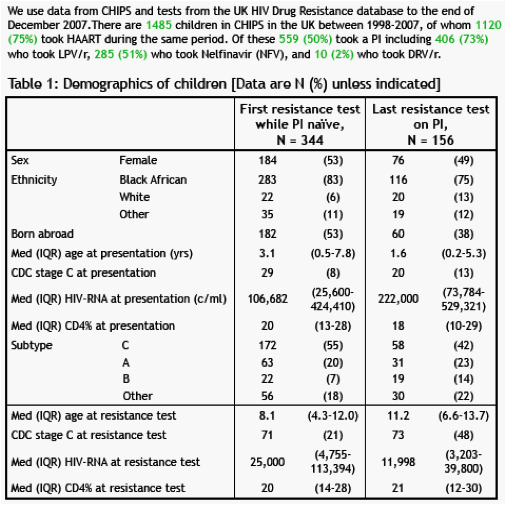
Results- 344 children had a PI-naive resistance test. 14/344 (3%) had a single RAM (2 V11I, 2 V32I, 1 I47A, 7 I50V, 1 G73S, 1 L89V); none had multiple RAMs. 10 (83%) were non-B subtype virus. 156 children had a resistance test on PI at a median (IQR) 2.6 (1.2-5.0) years on PI; 55 (35%) had taken LPV/r as their only PI. 21(13%) had one RAM, 5 (3%) had 2, and 3 (2%) had 3. In a multivariate model, a higher number of DRV RAMs was independently associated with increased time on PI (p=0.04), larger area under the viraemia curve since the start of PI (p=0.01), and any exposure to a PI other than LPV/r (p=0.02 vs. LPV/r only). However, only 3 (2%) PI-experienced children had intermediate level resistance to DRV/r using Stanford.
Conclusions- PI-naive children in the UK, and those whose only prior PI is LPV/r, have little DRV/r resistance, although the number of DRV RAMs increases slightly with an increased time andincreased viraemia on PI. Few PI-experienced children have more than one DRV RAM and susceptibility to DRV/r is high. This suggests that once daily DRV/r may have utility both as asecond PI as well as an alternative first PI, particularly if it can be co-formulated with a PI booster.
INTRODUCTION
Darunavir, boosted by ritonavir (DRV/r), has been shown to have significant activity against both wild-type and multidrug-resistant HIV-1 strains and to be virologically effective and generally well tolerated in children.
· Currently, Lopinavir (LPV/r) is the preferred first PI in UK children and is licensed for allages and weights in both tablet and syrup formulations.
· DRV/r is currently licensed in children aged 6+ years (twice-daily tablet). Trials of asuspension in 3-6 year olds, and once-daily administration across all ages, are ongoing.
Resistance:
· The presence of ≥3 DRV resistance-associated mutations (RAMs) at baseline was associated with a diminished virological response to DRV/r in the POWER [1] and DUET [2] studies in adults.
· In previous studies of resistance in adults, over 90% harboured <3 DRV RAMs and over two-thirds harboured none.
Rationale:
· In the future, DRV/r may be of more use in children both as first PI or as second PI afterprevious failure on PI.
· Therefore, identifying the prevalence of resistance mutations both in PI-naïve and PIexperienced children is important for identifying clinical utility of DRV/r.





References:
1. De Meyer, S. et al. 2008. Resistance profile of darunavir: combined 24-week results from the POWER trials. AIDS Res Hum Retroviruses 24: 379-388.
2. De Meyer, S. et al. 2008. Phenotypic and genotypic determinants of resistance to darunavir: analysis of data from treatment experienced patients in POWER 1, 2, 3 and DUET-1 and 2. Antivir Ther 13: Suppl3:A33.
3. De Meyer, S. et al. 2009. Confirmation of the negative impact of protease mutations I47V, I54M, T74P, and I84V and the positive impact of protease mutation V82A on virological response to darunavir/ritonavir. Poster 126, XVIII International HIV Drug Resistance Workshop.
.
|
| |
|
 |
 |
|
|