 |
 |
 |
| |
Biomarkers of Inflammation and Coagulation and Risk of Non-AIDS Death in HIV/Hepatitis Co-infected Patients in the SMART Study: inflammation associated with advanced liver disease
|
| |
| |
Reported by Jules Levin
CROI 2010 Feb 16-19
Lars Peters and for the INSIGHT SMART Study Group
Copenhagen HIV Prgm, Denmark
AUTHOR CONCLUSIONS
· Baseline levels of IL-6 and D-dimer were significantly higher in HIV/hepatitis co-infected participants with elevated levels of the liver fibrosis marker HA.
· Interruption of ART led to increased levels of IL-6 only in participants who also had elevated HA, but unexpectedly corresponding increases in hsCRP were not observed.
· During follow-up the risk of non-AIDS death in participants with elevated baseline levels of either hsCRP, IL-6 and D-dimer was highest if HA was also elevated.
Author Perspective:
Our data suggest that among HIV-viral hepatitis co-infected persons, those with impaired liver function are particularly in a pro-inflammatory state associated with excess risk of death from causes other than AIDS - and interruption of ART further exacerbates this pro-inflammatory state.
ABSTRACT
Background: In the SMART Study, hepatitis B and C co-infected patients randomized to the drug conservation (DC; interrupt ART until CD4 <250) group were at increased risk of NAD if their baseline plasma level of the fibrosis marker hyaluronic acid (HA) was elevated (>75 ng/mL), while the risk of NAD in the viral suppression (VS; continued use of ART) group was considerably lower and did not depend on baseline HA level. We hypothesized that patients with existing liver impairment (as determined by HA >75) would have higher levels of the coagulation and inflammation markers D-dimer, interleukin-6 (IL-6), and high sensitivity C-reactive protein (hsCRP) relative to patients with normal liver function (HA ≤75) and that the ability of biomarkers to predict NAD would differ by HA level.
Methods: D-dimer, IL-6 and hsCRP levels were measured at Baseline and at Month 6 in all patients (N = 655) positive for HCV-RNA or HBsAg, with baseline HA level and stored plasma samples. Biomarker levels were compared according to randomization group and baseline HA level. Risk of NAD (n = 50) was estimated using Cox regression.
Results: At baseline patients with increased HA (>75 vs ≤75) had higher levels of IL-6 [median (IQR) 4.41 pg/mL (2.68 to 6.42) vs 2.33 (1.59 to 3.69); P <0.001) and D-dimer (0.31 µg/mL (0.17 to 0.76) vs 0.27 (0.17 to 0.44); P =0.01), but not hsCRP. Only D-dimer increased in the DC group vs. the VS group by month 6 (% change (SE) 22.4 (5.5) vs -7.7 (5.3)], but the change in the DC group did not depend on HA baseline level (P =0.10). Higher baseline HA levels predicted risk of NAD before (HR per 100 ng/mL (95% CI) 1.3 (1.1 to 1.4); P <0.001) and after adjustment for other biomarkers and baseline risk factors (HR (95% CI) 1.2 (1.0 to 1.3); P =0.01]. A significant interaction (P =0.005) was found between baseline HA and D-dimer levels for predicting risk of NAD. Patients with elevated levels of both markers had the highest risk of NAD; stratifying patients into four equal sized groups based on median baseline HA and D-dimer levels, 48% overall (50% in DC vs 45% in VS) of the NAD occurred in the subgroup with both biomarkers elevated.
Conclusions: Levels of IL-6 and D-dimer were elevated in HIV/hepatitis coinfected patients with impaired liver function (HA >75 ng/mL). Interruption of ART led to further activation of coagulation but not inflammatory processes. Patients with impaired liver function and already activated coagulation processes are at particularly high risk of NAD.
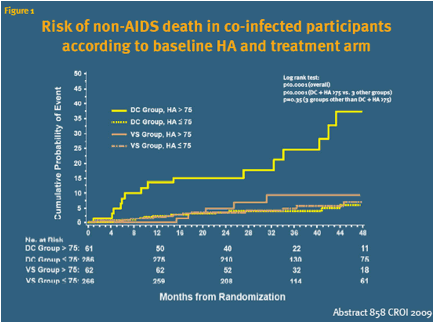

RESULTS
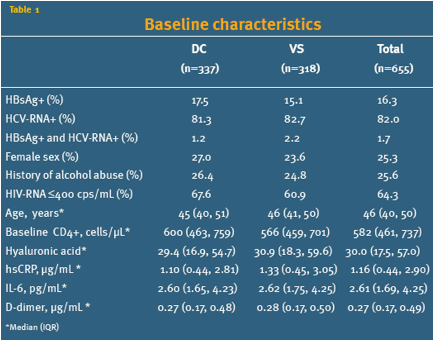
Baseline characteristics
Out of 5,472 participants enrolled in the SMART study from January 2002 - January 2006, 655 (12.0%) were HBV+ or HCV+, had HA measured at baseline and had baseline plasma samples available for biomarker analysis. Table 1 shows the baseline characteristics of the participants according to randomization group. 18.6 % had a HA level above the upper normal range (75 ng/mL). There were no significant differences in baseline variables between the two randomization groups.
Follow-up
At month 6 the median (IQR) CD4+ cell counts in the DC and VS groups were 441 cells/µL (328 - 572) and 596 cells/µL (447 - 758), respectively, while 28.1% and 72.1% had HIV-RNA £400 copies/mL.
50 participants died from non-AIDS causes (30 in DC and 20 in VS). Breakdown of the different causes of death (N;%) was: infection (7; 14%), non-AIDS cancer (6; 12%), substance abuse (6; 12%), hepatic (4; 8%), cardiovascular (4; 8%), renal (4; 8%), accident/violent/suicide (4; 8%), chronic obstructive pulmonary disease (1; 2%), CNS disease (1; 2%), unknown cause (13; 26%). The median (IQR) interval between the
measurement of baseline biomarkers and non-AIDS death was 17 (9 - 34) months.
Biomarker levels at baseline and during follow-up
The baseline levels of the three biomarkers are shown in table 1. The levels of all three biomarkers were higher in participants with elevated baseline HA (>75 ng/mL) compared with participants with HA in the normal range (>75 ng/mL), but the difference was only statistical significant for IL-6 and D-dimer, table 2.
Interruption of ART led to a significantly higher percent change in D-dimer, but not hsCRP and IL-6 levels, from baseline to month 6 (fig. 2).
Participants randomized to the DC group with an elevated HA level had a 47.3% increase in IL-6 from baseline to month 6 compared with a 0.7% decrease in DC group participants with HA within the normal range. The increase in IL-6 was not associated with an increase in hsCRP, which decreased slightly in all groups (fig. 3).
Predictors for non-AIDS death
All participants were stratified into four equal sized groups based on baseline median HA and median biomarker level. Overall the subgroups with both HA and either hsCRP, IL-6 or D-dimer above the median at baseline accounted for 52%, 62% and 48% of all non-AIDS deaths, respectively. On the contrary, only 8% of all non-AIDS deaths were seen in the subgroups having both HA and either biomarker below the median baseline level (fig. 4). The interaction p-values for dichotomous HA x biomarker for non-AIDS death were 0.88, 0.89 and 0.63 for hsCRP, IL-6 and D-dimer, respectively. Adjusted hazard ratios (95% confidence
interval) for non-AIDS death comparing those with both HA and biomarker above the median to those with both HA and biomarker below the median were 6.1 (2.1-17.7), p=0.001; 5.9 (2.0-17.3), p=0.001 and 4.4 (1.5-13.3), p=0.008 for hsCRP, IL-6 and D-dimer, respectively. The results of the univariate analysis were similar.
|
| |
|
 |
 |
|
|