 |
 |
 |
| |
Impact of Zidovudine on Hematologic Parameters during Treatment with Peginterferon alfa-2a plus Ribavirin for Chronic Hepatitis C in HIV/HCV Co-infected Patients Enrolled in a Large Randomized Double-blind Study
|
| |
| |
Reported by Jules Levin
CROI 2010 Feb 16-19 SF
M Rodriguez-Torres1, Jihad Slim*2, L Bhatti3, R Sterling4, M Sulkowski5, T Hassanein6, B Clotet7, T Branco8, S Passe9, S Stancic9, and the PARADIGM Study Investigators
1Fndn de Investigación de Diego, Santurce, Puerto Rico; 2St Michael’s Med Ctr, Newark, NJ, US; 3AIDS Hlthcare Fndn, Los Angeles, CA, US; 4Virginia Commonwealth Univ, Richmond, US; 5Johns Hopkins Univ Sch of Med, Baltimore, MD, US; 6Univ of California, San Diego, US; 7Fndn irsiCaixa, Hosp Univ Germans Trias i Pujol, Badalona, Spain; 8Hosp Fernando da Fonseca, Amadora, Portugal; and 9Roche, Nutley, NJ, US
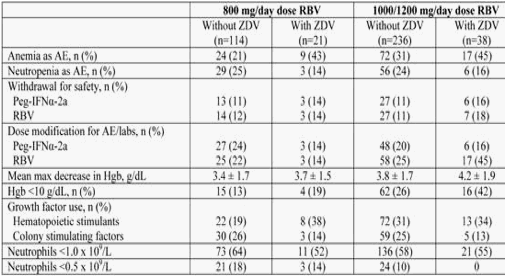
Background: Anemia and neutropenia are reported to be more frequent and severe in HIV-HCV co-infected patients receiving ZDV during Peg-IFN/RBV treatment. However, it is unclear if the hematological safety profile of ZDV with 1000/1200 mg/day RBV is similar to that with 800 mg/day RBV dosing. We explored the safety of Peg-IFN/RBV +/- ZDV in the randomized double-blind international PARADIGM study.
Methods: HIV-HCV (genotype 1) co-infected adults naive to IFN-based therapy for CHC with stable HIV disease (on stable anti-retroviral therapy [ART] or not requiring ART) and ≥100 CD4+ cells/mm3 were eligible. All patients received Peg-IFNα-2a 180 mg/wk and were randomized (1:2) to receive 800 mg/day or 1000/1200 mg/day RBV for 48 wks. The impact of ZDV treatment on hematologic parameters (hemoglobin [hgb], neutrophil count) was examined.
Results: In the intent-to-treat population (n = 410) the mean age was 45 years, the mean HCV RNA level was 6.4 log10, 30% were African American, 24% were Hispanic, 54% had an alanine aminotransferase quotient ≤1.5, 88% were receiving ART at baseline and 6% had <200 CD4+ cells/mm3. Among those receiving 800 mg/day or 1000/1200 mg/day RBV, 16% and 14% were receiving ZDV, respectively. In the safety population (table; n = 409), there was no negative impact of concomitant administration of ZDV with Peg-IFNα-2a/RBV on neutrophils. However, anemia was consistently more common among patients receiving Peg-IFNα-2a and any dose of RBV in combination with ZDV. In particular, the rate of RBV dose modifications among patients receiving 1000/1200 mg/day RBV in combination with ZDV was almost twice as great compared with patients not taking ZDV (45% vs 25%).
Conclusions: This analysis confirms that anemia is more common and severe in co-infected patients on ZDV during Peg-IFN/RBV treatment. It also confirms that ZDV used in conjunction with weight-dosed RBV requires more dose modifications because of anemia. These results should be used to consider ART modifications before therapy for CHC.
|
| |
|
 |
 |
|
|