 |
 |
 |
| |
HIV-1 Replication and Immune Dynamics Are Impacted by Raltegravir Intensification of HAART-suppressed Patients
|
| |
| |
Comments by David Margolis on intensification

HIV-1 Replication and Immune Dynamics Are Impacted by Raltegravir Intensification of HAART-suppressed Patients
Reported by Jules Levin
CROI 2010
M J Buzon1, M Massanella1, J Llibre2, A Esteve3, M C Puertas1, S Palmer4, M Stevenson5, B Clotet1,2, J Blanco1, Javier Martinez-Picado*1,6, and Integral Collaborative Group
1Fndn irsiCaixa, Badalona, Spain; 2Fndn Lluita contra la SIDA, Badalona, Spain; 3CEEISCAT, Badalona, Spain; 4Swedish Inst for Infectious Disease Control, Solna; 5Univ of Massachusetts Med Sch, Worcester, US; and 6ICREA, Barcelona, Spain
"The increase in episomal cDNA following raltegravir intensification requires the presence of infectious virus, de novo infection and reverse transcription, which together, strongly suggest that active viral replication persists in some HAART suppressed patients. The observed normalization of immune activation in those patients suggests that active replication under HAART is a cause of aberrant immune activation rather than a consequence of it. The ability of raltegravir intensification to perturb the reservoir has implications for therapeutic strategies aimed at achieving viral eradication."




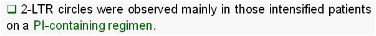
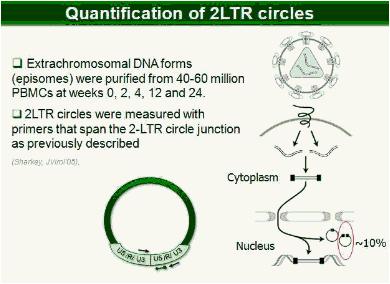
Background: It is generally believed that active HIV replication is halted in patients on HAART, however immune activation/inflammation continues at abnormal levels suggesting continued, low level replication. In the presence of raltegravir, linear HIV-1 cDNA is prevented from integrating into chromatin and is subsequently converted to episomal cDNAs (1 & 2 LTR circles). Therefore, an increase in episomal cDNA occurs when active replication is inhibited by integrase inhibitors. To assess whether active replication might be driving immune activation in HAART-suppressed patients, we examined the impact of intensification with raltegravir on viral cDNA and immune activation parameters.
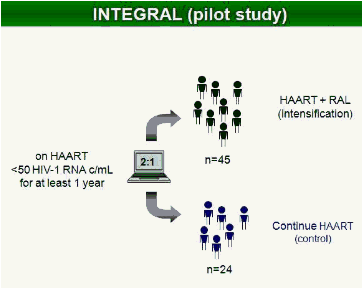
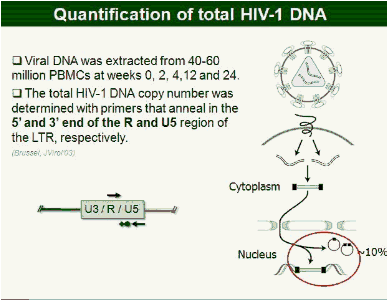
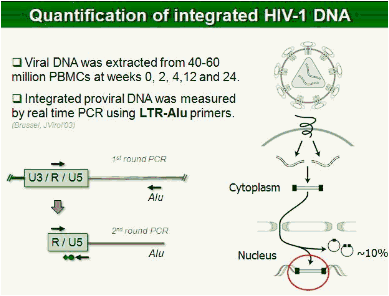
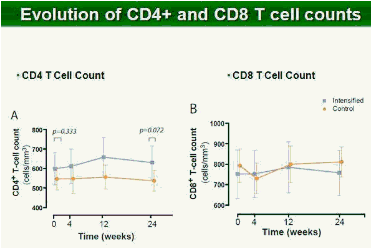
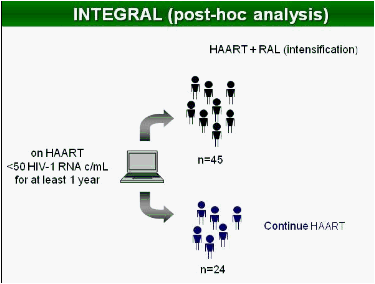
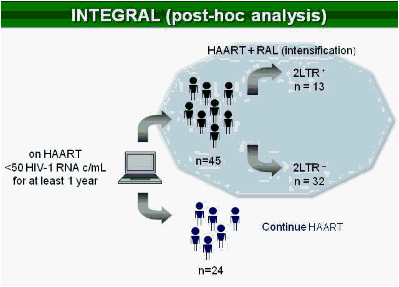
Methods: In this study, 69 patients with <50 HIV-1 RNA copies/mL for >1 year were randomized to intensify their HAART with raltegravir (n = 45), or to continue their HAART (n = 24) for 48 weeks. Integrated, episomal (2LTR circles) and total viral DNA forms were measured by real-time PCR at weeks 0, 2, 4, 12 and 24. Immune activation markers were assessed by flow cytometry in fresh blood samples during follow-up. Ultrasensitive viral load (SCA) and soluble CD14 were measured from plasma samples by real-time PCR and ELISA, respectively.
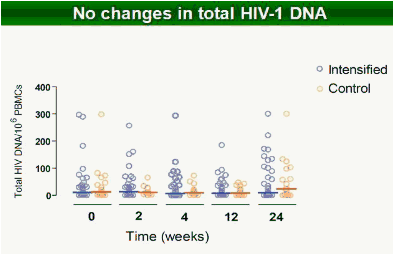
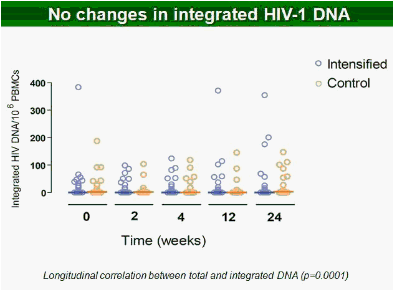
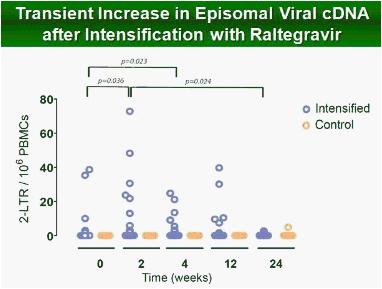
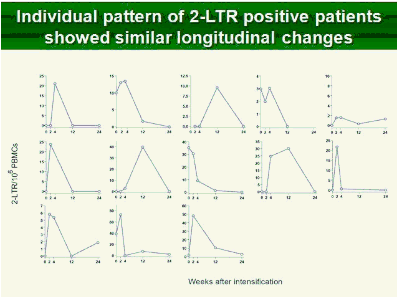
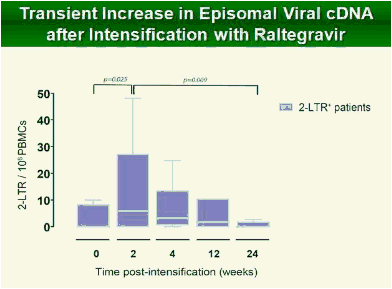
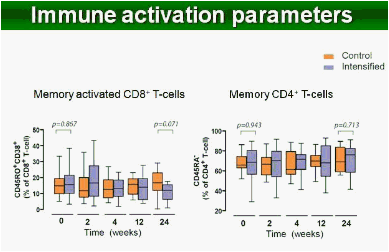
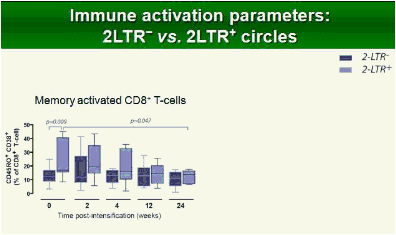
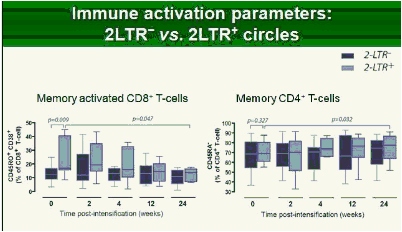
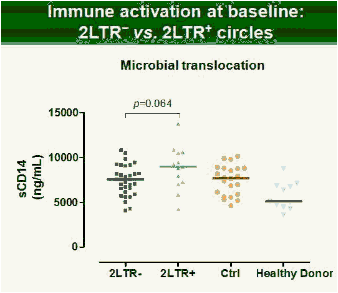
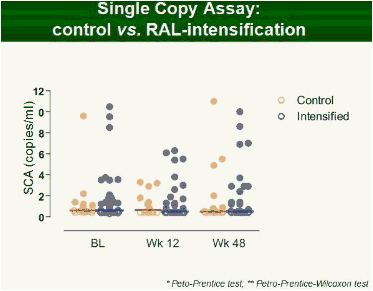
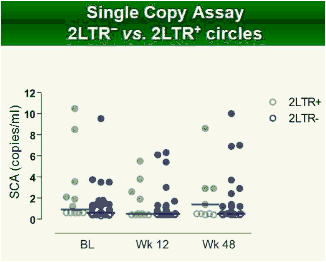
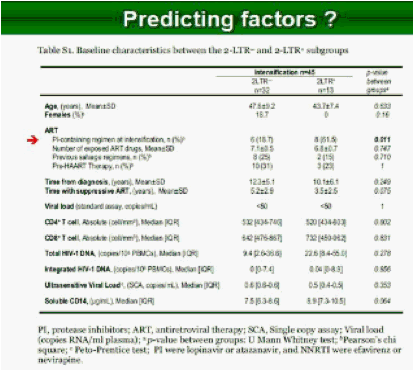
Results: Raltegravir intensification of a 3-drug suppressive HAART regimen resulted in a specific and transient increase in 2-LTR circle DNA in a significant percentage (29%) of HAART-suppressed patients. In contrast, total and proviral DNA levels remained stable during intensification. No significant differences in SCA were found. Interestingly, patients who were 2-LTR+ showed higher levels of immune activation (HLA-DR+CD38+, CD38+CD45RO+, HLA‑DR+CD45RO+ in CD8 T-cells) at baseline and a subsequent normalization after intensification. This subgroup showed also a trend towards higher concentration of plasma soluble CD14.
Conclusions: The increase in episomal cDNA following raltegravir intensification requires the presence of infectious virus, de novo infection and reverse transcription, which together, strongly suggest that active viral replication persists in some HAART suppressed patients. The observed normalization of immune activation in those patients suggests that active replication under HAART is a cause of aberrant immune activation rather than a consequence of it. The ability of raltegravir intensification to perturb the reservoir has implications for therapeutic strategies aimed at achieving viral eradication.






|
| |
|
 |
 |
|
|