 |
 |
 |
| |
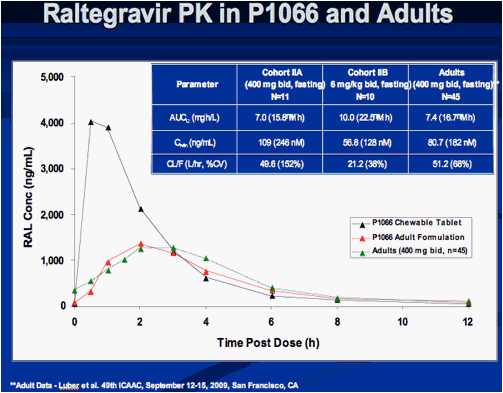
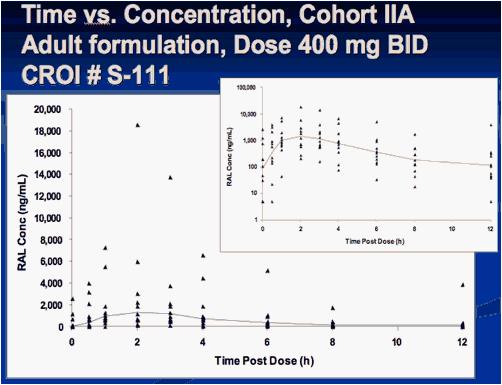
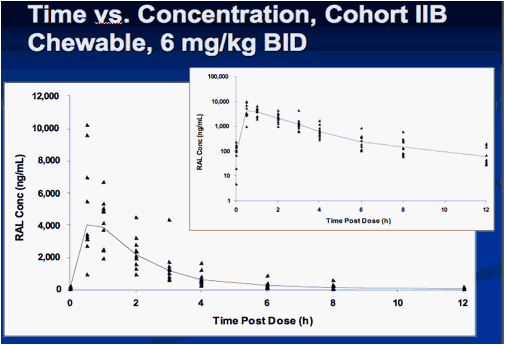
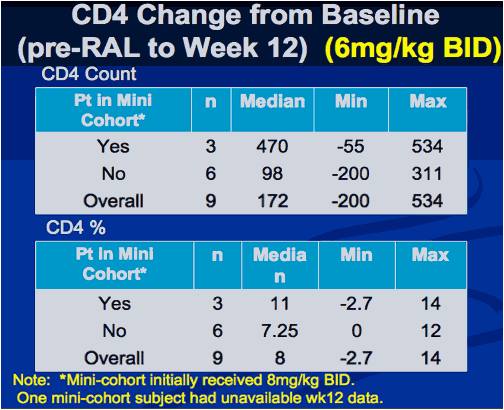
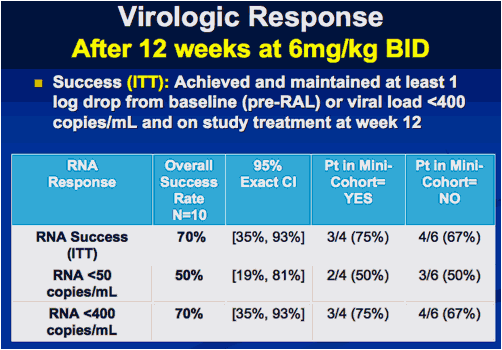
Interim Results from IMPAACT P1066: Raltegravir (RAL) Oral Chewable Tablet (OCT) Formulation in Children 6-11 Years
|
| |
| |
Reported by Jules Levin
CROI 2010 Feb 16-19 SF
Sharon Nachman , Edward Acosta, Pearl Samson, Hedy Teppler, Brenda Homony, Terence Fenton, Edward Handelsman, Carol Worrell, Bobbie Graham, Andrew Wiznia, and The P1066 Group





|
| |
|
 |
 |
|
|